Combined Gas Law — Pressure, Volume, and Temperature Relationship
The Combined Gas Law explains one of the most fundamental and universal relationships in thermal physics by unifying how pressure, volume, and temperature of a gas change in response to one another. It emerges from the integration of three classical gas laws—Boyle’s law, Charles’s law, and Gay-Lussac’s law—and presents them not as separate ideas but as one cohesive understanding of gaseous behavior. Even without mathematical expressions, the essence of the Combined Gas Law lies in recognizing that gases are dynamic, constantly moving collections of particles whose behavior is tightly governed by changes in their surroundings. As pressure varies, as temperature increases or decreases, and as the volume of the container expands or contracts, the gas responds through shifts in energy, motion, spacing, and internal interactions. This interdependent relationship makes the Combined Gas Law one of the foundational principles used not only in physics and chemistry but also in engineering, meteorology, aviation, environmental science, and countless real-world processes where gases play an essential role.
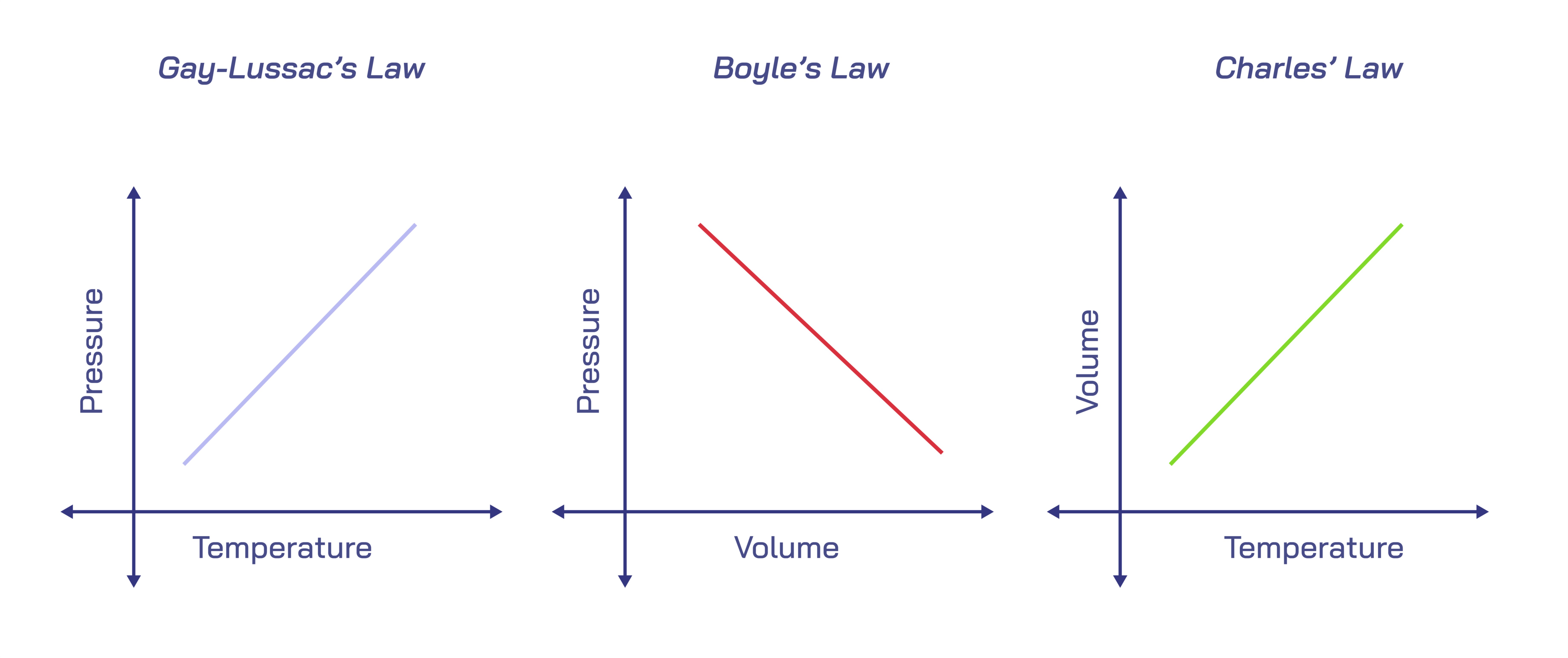
At its core, the Combined Gas Law reveals that when the amount of gas remains constant, any change in one variable necessitates a proportional adjustment in the others. This interconnectedness can be understood by considering the microscopic world of gas particles. As temperature rises, the particles gain kinetic energy, move more vigorously, and collide with greater force against the walls of their container. These energetic collisions naturally lead to higher pressure if the volume is held constant or force the gas to expand if the container is flexible. Conversely, when temperature drops, the particles lose energy, slow down, and exert less outward force, which results in reduced pressure or contraction in volume depending on the constraints of the system. In this way, the Combined Gas Law captures the energetic sensitivity of gases and shows how the simplest environmental change can cascade into measurable physical effects. Its explanatory power lies not in complex equations but in this deeply intuitive understanding of particle behavior.
Understanding the relationship between pressure and volume forms a key element of the Combined Gas Law. When the temperature remains the same and volume decreases—such as compressing air in a bicycle pump—the gas particles have less space to move. Their collisions with the container walls increase in frequency, resulting in higher pressure. Alternatively, when the volume expands—such as airing up a balloon—the particles spread out, collide less frequently, and the internal pressure falls. These transformations occur every time gases are compressed in industrial cylinders, when deep-sea divers descend and feel pressure changes, or when weather balloons rise and expand as atmospheric pressure decreases. Such examples demonstrate that volume and pressure are intimately linked and that their connection remains predictable when part of the larger Combined Gas Law framework.
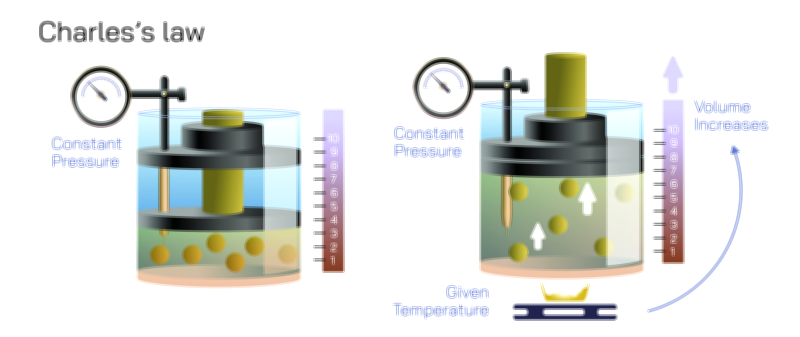
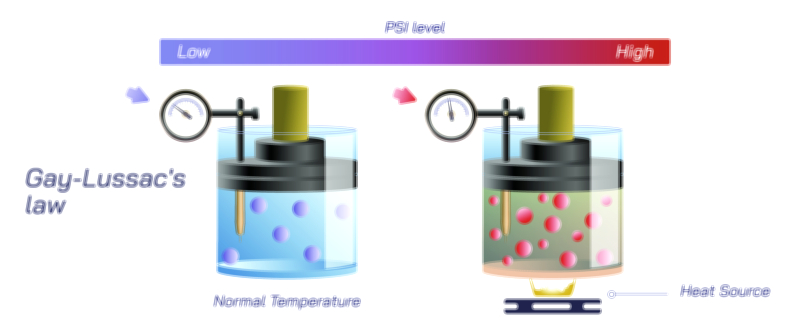
Temperature plays an equally crucial role and reveals much about how gases behave in natural and engineered systems. Warmer gases expand or exert more pressure because their particles possess greater kinetic energy. This principle explains why heated air rises, why hot air balloons float, and why tire pressure increases on hot days. Similarly, cold gases contract or decrease in pressure, which is why car tires appear slightly deflated during winter mornings or why refrigerated containers require careful monitoring of gas pressure. The Combined Gas Law ties these behaviors together by showing that temperature does not act independently—it directly influences pressure and volume in a symbiotic relationship. As temperature shifts, the gas responds holistically, adjusting its physical state to maintain consistency with the surrounding conditions.
The Combined Gas Law becomes most meaningful when applied to real-life systems where pressure, volume, and temperature change simultaneously. Consider the environment inside an airplane cabin: as the aircraft ascends, outside air pressure decreases dramatically, but the air within the cabin must be maintained at safe and comfortable levels. Temperature also drops in the upper atmosphere. Engineers rely on the Combined Gas Law to design pressurization systems that compensate for those changes, ensuring that the internal environment remains stable. The same principle governs the design of pressurized tanks, scuba gear, heating systems, and even aerosol cans that respond to temperature fluctuations. Fire extinguishers, gas pipelines, refrigeration cycles, and industrial boilers all depend on predictable gaseous behavior based on these relationships.
In meteorology, the Combined Gas Law helps explain large-scale atmospheric motions and daily weather phenomena. As air masses warm, they expand, become less dense, and rise, creating regions of low pressure that influence wind patterns and precipitation. As air cools, it contracts and sinks, increasing pressure and shaping high-pressure systems that bring clear skies. The behavior of air during the formation of storms, the movement of cold and warm fronts, and the development of pressure gradients all reflect the interplay described by the Combined Gas Law. Without an understanding of how temperature affects pressure and volume, meteorologists could not predict weather patterns or model atmospheric systems with accuracy.
The Combined Gas Law is also vital in understanding natural processes such as volcanic eruptions, oceanic gas release, and the behavior of gases trapped in glaciers. In a volcano, for example, gases trapped within magma expand dramatically when heated, increasing pressure until the surrounding rock fractures, releasing the stored energy in a powerful eruption. Likewise, the expansion of dissolved gases in carbonated beverages when exposed to warm temperatures or reduced pressure is a simple everyday example of the same principle. Even the way organisms breathe reflects the Combined Gas Law: lungs expand and contract as pressure changes allow air to flow in and out, driven by temperature and volume adjustments during inhalation and exhalation.
In engineering design, the Combined Gas Law helps determine how materials must behave under fluctuating pressures and temperatures. Storage cylinders for medical oxygen, industrial gases, or liquefied fuels must account for temperature-induced pressure changes to avoid bursts or structural failure. Engineers designing heating, ventilation, and air conditioning systems must consider how heated or cooled air expands, contracts, and exerts pressure on ducts and chambers. Mechanical systems such as internal combustion engines rely on the expansion of gases under high temperature to push pistons and generate motion. The law’s principles guide the safe handling of gases in laboratories, manufacturing plants, chemical reactors, and energy facilities where precise control of conditions is essential.
One of the most profound insights offered by the Combined Gas Law lies in its connection to human perception of energy. Through everyday experiences—such as a balloon shrinking in cold air, a pressure cooker working faster under high pressure, or a tire expanding on a hot day—people observe how gases continually respond to environmental changes. The law captures these intuitive observations and unifies them under a single conceptual umbrella. Because gases are highly sensitive to temperature and pressure fluctuations, their behavior can be easily observed and understood even without specialized scientific knowledge. The Combined Gas Law transforms these simple experiences into a deeper understanding of the physical world.
It also forms the stepping stone toward more advanced concepts in thermodynamics such as the ideal gas behavior, the kinetic molecular theory, and the relationship between energy, entropy, and molecular motion. By appreciating the Combined Gas Law, one begins to see how matter responds to energy input, how microscopic behavior influences macroscopic effects, and how universal principles govern even the simplest systems. It teaches that materials, when subjected to external conditions, adapt in predictable ways, providing the foundation for scientific reasoning across multiple disciplines.
Ultimately, the Combined Gas Law demonstrates that pressure, volume, and temperature are not isolated variables but parts of an interconnected system governed by the energetic behavior of gas particles. This relationship shapes everything from climate and weather to engines and everyday objects. It defines how gases expand and contract, how they store and release energy, and how their responsiveness enables technologies and natural processes to function. Through its universal applicability and conceptual clarity, the Combined Gas Law stands as one of the most important insights in the physical sciences, illustrating how changes in the environment translate directly into changes in the behavior of matter.