Types of Instability in Emulsification — Separation and Breakdown Process
Emulsions, though remarkably useful and versatile, exist in a delicately balanced state. They are formed by dispersing tiny droplets of one immiscible liquid within another and stabilizing these droplets through surface-active agents. Yet despite careful formulation, emulsions naturally tend toward instability over time. This is because the energetic preference of the system is to reduce the interfacial area between the two liquids, returning to a separated state. The different types of instability observed in emulsions arise from a range of physical processes, each governed by droplet interactions, density relationships, interfacial forces, gravity, and the mechanical or chemical environment surrounding the emulsion. Exploring these forms of instability reveals how emulsions weaken, how breakdown begins, and what mechanisms govern the journey from a stable, uniform mixture to separated layers. The processes that drive instability illustrate the subtle interplay between microscopic droplet behavior and macroscopic separation, making emulsification a vivid example of how material systems seek equilibrium.
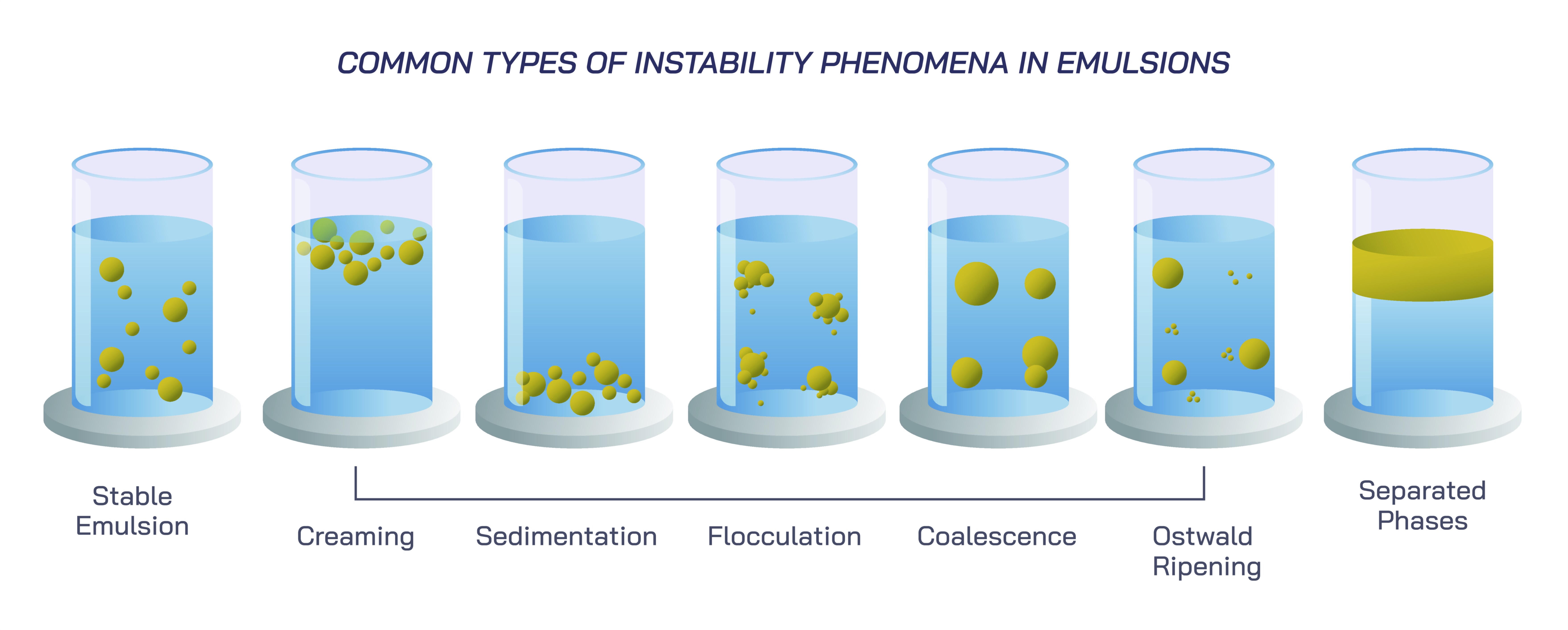
One major path of instability emerges when droplets begin to migrate within the emulsion because of density differences between the dispersed and continuous phases. This leads to the phenomenon known as creaming or sedimentation, depending on whether the droplets rise or sink. In an oil-in-water emulsion, where oil is usually less dense than water, droplets tend to rise toward the surface, forming a layer near the top. In a water-in-oil emulsion, heavier water droplets sink if the oil is lighter. Although creaming and sedimentation do not immediately break the emulsion’s internal structure, they change its visual appearance and physical uniformity. The droplets remain intact and separate from one another, but their migration creates distinct layers that can eventually encourage deeper destabilization. The gravitational pull acting upon each droplet makes this process inevitable unless the droplets are extremely small or the continuous phase is thickened enough to resist their movement.
Another form of instability involves the subtle clustering of droplets without fully merging. This phenomenon, called flocculation, causes droplets to come together into loose, reversible aggregates held by weak attractive forces. The droplets do not blend into one another; instead, they form clusters that can easily be redispersed with gentle agitation. Flocculation changes the texture, appearance, and flow behavior of an emulsion, making it appear thicker, grainier, or cloudier. While not catastrophic on its own, flocculation often marks the early stages of deeper structural weakening, because clustered droplets have a greater likelihood of eventually merging into larger droplets. This increased contact frequency sets the stage for more severe instabilities. The delicate balance between attractive and repulsive forces around each droplet determines whether flocculation remains a mild disturbance or becomes a precursor to full separation.
As instability progresses, droplets may begin to merge into larger ones through a process known as coalescence. Unlike flocculation, coalescence involves the breakdown of the thin protective layer around each droplet, allowing their internal contents to fuse. When droplets coalesce, the number of droplets decreases while their average size increases, reducing the total interfacial area within the emulsion. This shift is energetically favorable for the system, which naturally tries to minimize interfacial energy. The loss of emulsifier effectiveness, changes in temperature, mechanical disruption, or chemical imbalance can all weaken the protective layers surrounding droplets. Once coalescence begins, the emulsion increasingly loses stability, as larger droplets rise or sink more quickly and are harder to keep suspended. The texture becomes uneven, the consistency weakens, and visual separation accelerates. Coalescence represents one of the most significant steps toward complete breakdown, because it destroys the dispersed structure that defines an emulsion.
When coalescence continues unchecked, the process may lead to a full collapse known as breaking. Breaking occurs when the dispersed droplets reunite into a continuous phase, completely separating from the other liquid. The interface between the two phases reforms, producing distinct layers—usually oil gathering as a unified mass apart from the water, or vice versa. Unlike flocculation or mild creaming, breaking is not reversible through simple shaking or mixing, because the protective layers separating the droplets have been irreversibly compromised. The system has returned to a lower-energy state where the two liquids no longer share a dispersed structure. Breaking marks the end stage of emulsion instability, signaling that the delicate balance of emulsification has been fully destroyed.
A related but distinct type of instability occurs when droplets grow not by merging but through an exchange of molecules across the continuous phase. This phenomenon, known as Ostwald ripening, develops when smaller droplets shrink and larger ones expand due to differences in internal pressure and solubility. Smaller droplets have higher internal pressure, making their molecules more likely to migrate into the continuous phase and reattach to larger droplets, which have lower internal pressure. Over time, this microscopic material exchange causes the overall droplet size distribution to shift toward fewer, larger droplets, destabilizing the emulsion even if no coalescence occurs. Ostwald ripening is especially common when the dispersed liquid has partial solubility in the continuous phase. The process may be slow and silent, but its cumulative effect significantly undermines stability, contributing to long-term breakdown in food products, pharmaceuticals, and cosmetic formulations.
Temperature fluctuations introduce yet another pathway to instability. Heating an emulsion increases molecular motion, potentially weakening the protective layers around droplets. This speeds coalescence, encourages droplet migration, and may distort the interfacial structure that keeps the mixture stable. Cooling, on the other hand, may cause components to crystallize or solidify, disturbing the arrangement of droplets and creating irregularities that lead to separation upon reheating. Many emulsions, particularly culinary or cosmetic ones, are highly sensitive to temperature changes, transforming smooth textures into grainy or separated mixtures when conditions fluctuate beyond safe limits. Temperature-induced instability demonstrates how delicate the balance of emulsification is and how environmental conditions can undermine its structural foundation.
The breakdown of emulsions also depends heavily on the concentration and effectiveness of the emulsifying agent. If insufficient emulsifier is present to coat all droplets, unprotected droplets quickly merge or cluster, accelerating separation. Over time, emulsifiers themselves may degrade chemically, lose their structural orientation, or become displaced at the interface by other molecules. The loss of emulsifier integrity directly weakens the droplet barrier, allowing instability mechanisms to advance more rapidly. The emulsion’s longevity therefore hinges not only on initial formulation but also on the continued viability of its stabilizing agents.
Instability can also arise from mechanical disturbances. Vigorous shaking, vibration, or repeated shear forces can disrupt the thin films surrounding droplets, leading to merging or restructuring of the emulsion. While some emulsions withstand mechanical manipulation, others break quickly if their interfacial layers are fragile. This sensitivity influences how emulsions must be stored, transported, or packaged, especially in industrial and pharmaceutical contexts where consistent texture and performance are essential.
All these forms of instability—creaming, sedimentation, flocculation, coalescence, Ostwald ripening, and breaking—reveal the multiple pathways through which emulsions gradually deteriorate. They demonstrate that emulsification is not a permanent achievement but an ongoing balance between competing forces: interfacial tension versus emulsifier stability, droplet size versus gravitational pull, molecular diffusion versus structural resistance, and environmental conditions versus compositional integrity. Each type of instability reflects a different aspect of this struggle for balance, with some processes reversible and others irreversible.
Understanding these mechanisms allows scientists, cooks, manufacturers, and formulators to design more stable emulsions by controlling droplet size, adjusting emulsifier concentration, manipulating viscosity, and regulating storage conditions. By mastering the forces that drive instability, people can create emulsions that maintain their consistency, appearance, and performance over time.
Emulsification therefore becomes a vivid illustration of how microscopic events determine macroscopic outcomes. The smallest shifts—whether a droplet growing slightly larger, a film thinning at its boundary, or molecules diffusing across phases—can reshape the entire structure of the emulsion. Instability is not a single event but a sequence of physical changes that gradually transform a unified mixture back toward separation. Through understanding these processes, the breakdown of emulsions becomes not merely a practical concern but a window into the dynamic nature of mixed systems and the delicate balance required to keep incompatible liquids harmoniously combined.