Gas Solubility — Factors, Temperature Influence, and Henry’s Law Explained
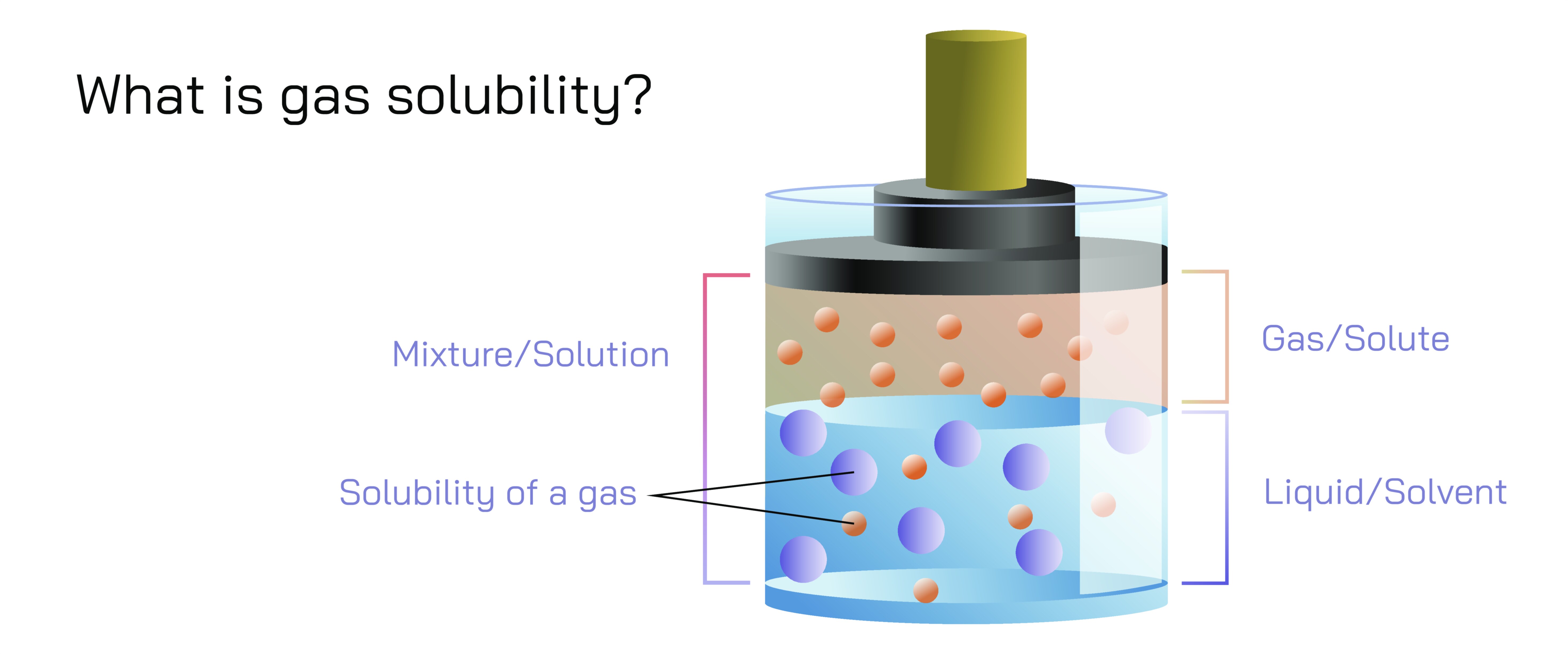
Gas solubility describes how much of a gas can dissolve in a liquid, and although it is often summarized through formulas or short definitions, its true meaning becomes clearer through a detailed conceptual understanding of the molecular processes involved. When a gas dissolves in a liquid, its molecules escape the free, fast-moving state of the gas phase and enter the more structured environment of the liquid phase, where they become surrounded by liquid molecules. The ease with which this transition happens depends on how strongly gas molecules interact with liquid molecules, how frequently gas particles collide with the liquid surface, and how physical conditions such as pressure and temperature influence molecular motion. Solubility is not just a chemical property; it is a key mechanism underlying numerous natural and industrial phenomena, including the oxygenation of blood, aeration of water bodies, carbonation of beverages, deep-sea diving physiology, and the behavior of atmospheric gases dissolved in oceans that contribute to climate regulation.
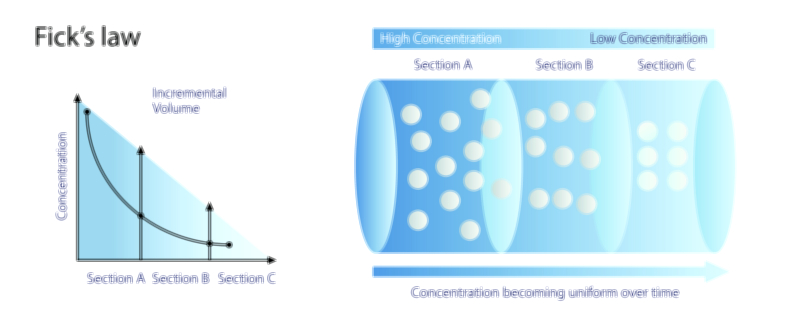
To understand gas solubility deeply, one must appreciate the relationship between intermolecular forces and the structure of liquids. Liquids consist of molecules packed much more closely than those in gases. When gas molecules collide with the surface of a liquid, some enter the liquid phase and become temporarily trapped by intermolecular interactions. If these interactions are strong enough, the gas molecules remain in the liquid; if too weak, they escape back into the gas phase. Non-polar gases such as nitrogen dissolve poorly in water because water molecules form strong hydrogen-bonded networks that do not interact well with non-polar molecules. In contrast, gases like carbon dioxide, sulfur dioxide, and ammonia form partial or full bonds with water molecules, making them more soluble. This principle is crucial for numerous biological and environmental processes. For example, aquatic organisms depend on the solubility of oxygen in water for cellular respiration, while carbon dioxide’s solubility helps regulate ocean acidity and global climate.
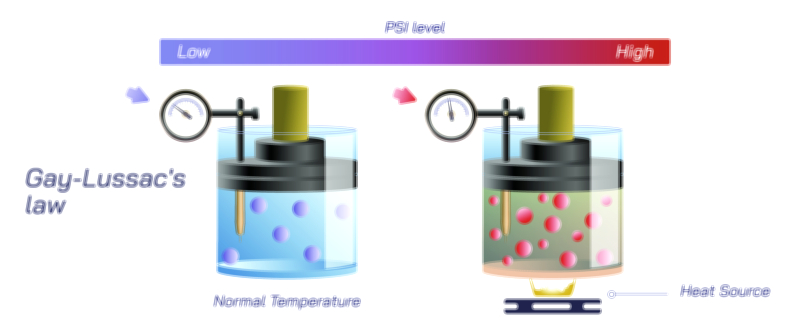
Temperature plays a powerful and somewhat counterintuitive role in gas solubility. In almost all gas-liquid systems, solubility decreases as temperature increases. The reason becomes clear when examining molecular motion: at higher temperatures, both liquid and gas molecules move more vigorously, increasing the tendency of dissolved gas molecules to escape back into the gas phase. This explains why cold water can hold more dissolved oxygen than warm water and why carbonated drinks lose their fizz much faster when left at room temperature. Warm beverages allow gas to escape readily because thermal energy disrupts the interactions that keep gas molecules dissolved. In nature, this temperature-solubility relationship leads to seasonal variations in aquatic oxygen levels, affecting ecosystems profoundly. During warm months, lakes and rivers may lose dissolved oxygen, stressing fish and other aquatic life. Conversely, cold polar waters absorb large amounts of atmospheric gases—including carbon dioxide—contributing substantially to global gas exchange cycles.
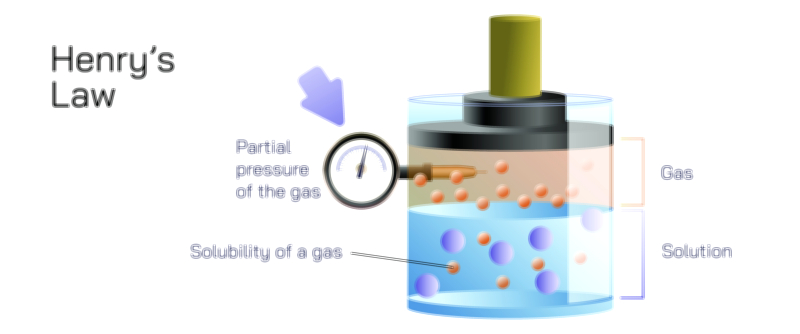
Pressure also strongly influences gas solubility. As the pressure of a gas above a liquid increases, more gas molecules collide with the liquid surface and dissolve. This direct relationship between gas pressure and solubility is captured conceptually by Henry’s Law, which states that the amount of gas dissolved in a liquid is proportional to the partial pressure of the gas above the liquid. Although Henry’s Law can be represented mathematically, the principle can be understood without formulas by recognizing that when gas pressure increases, more gas molecules are forced into close contact with the liquid surface, increasing the likelihood that they will enter and remain in the solution. This is why carbonated beverages are packaged under high pressure: the elevated pressure forces large amounts of carbon dioxide into the liquid. Once the container is opened, the pressure suddenly drops, and the dissolved carbon dioxide escapes as bubbles until equilibrium is reached again. Henry’s Law thus provides a conceptual link between gas pressure and the concentration of dissolved gas in a liquid.
Henry’s Law has deep practical significance across medicine, engineering, diving physiology, and environmental science. In clinical settings, it helps explain how oxygen dissolves into blood based on oxygen pressure in the lungs; higher partial pressure during inhalation increases oxygen solubility in the bloodstream. For divers, rapid changes in underwater pressure can produce dangerous effects if nitrogen dissolved under high pressure suddenly comes out of solution when pressure drops, forming gas bubbles in the bloodstream—a condition known as decompression sickness. Industrial chemists use Henry’s Law to design absorption towers that remove pollutants from emissions by dissolving gases into liquids under pressurized conditions. On a planetary scale, Henry’s Law governs how the atmosphere exchanges gases with Earth’s oceans, shaping patterns of carbon sequestration and influencing climate regulation.
Another important factor influencing gas solubility is the chemical reactivity of the gas in the liquid. Gases that chemically react with the solvent exhibit much higher solubility than would be predicted from physical interactions alone. For example, carbon dioxide reacts with water to form carbonic acid, which further dissociates into bicarbonate and carbonate ions. This reaction removes carbon dioxide from the dissolved state and drives more gas into the solution, increasing effective solubility. Similarly, ammonia dissolves readily in water because it reacts to form ammonium ions. These reactive pathways explain why some gases remain dissolved even after pressure drops and why others escape readily. The interplay between physical dissolution and chemical reaction contributes to complex environmental behavior, such as ocean buffering systems that stabilize acidity through carbonate chemistry.
Agitation, particle size of gas bubbles, and liquid flow also influence gas solubility by increasing the rate of contact between the liquid and gas. Stirring or shaking enhances solubility temporarily by allowing more gas to meet the liquid surface. Fine bubble diffusers used in aquariums and wastewater treatment systems increase oxygen transfer efficiency by dispersing gas into tiny bubbles, dramatically increasing the surface area available for dissolution. In natural waters, turbulence from waves and waterfalls enhances gas exchange with the atmosphere, improving aeration for aquatic life.
The polarity of the liquid also plays a major role. Polar liquids like water dissolve polar or reactive gases far more effectively than non-polar gases. Conversely, non-polar liquids such as oil dissolve gases like nitrogen or oxygen more readily than they dissolve polar molecules. This relationship between solvent characteristics and solubility explains why oxygen dissolves into membrane lipids in living cells and why gases behave differently in different chemical environments. It also influences applications such as food preservation, polymer processing, and petrochemical separations.
Salinity is an additional factor influencing solubility. Dissolved salts reduce the ability of water to hold gases, meaning that seawater contains less dissolved oxygen than freshwater at the same temperature and pressure. This “salting-out” effect is important in marine biology and ocean circulation. Lower gas solubility in saltwater constrains oxygen availability for marine organisms and affects global biogeochemical cycles.
Ultimately, gas solubility emerges from the interplay of pressure, temperature, solvent composition, molecular interactions, and chemical reactivity. Henry’s Law serves as a cornerstone principle that ties pressure and solubility together, revealing why gases dissolve more readily under high pressure and escape when pressure decreases. Temperature shapes solubility by influencing molecular motion, while chemical reactions and solvent polarity determine how strongly dissolved molecules are held in the liquid phase. When viewed as a complete conceptual system rather than a collection of formulaic rules, gas solubility reveals its importance across countless natural and human-made systems: maintaining life in aquatic habitats, enabling respiration in humans, influencing carbonation in beverages, affecting industrial gas scrubbing, guiding deep-sea safety protocols, and regulating Earth’s climate. Appreciating these interconnected dynamics provides a deeper understanding of both the beauty and the complexity of gas-liquid interactions in the world around us.