Henry’s Law — Gas Solubility and Pressure Relationship Explained
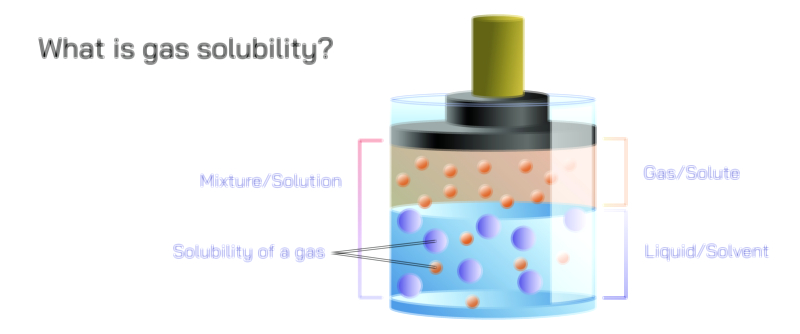
Henry’s Law is one of the most fundamental principles that connects the physical behavior of gases with the way they dissolve in liquids, and its meaning becomes truly clear when explored not as a mathematical rule but as a conceptual description of how gas molecules interact with a liquid surface under different pressures. At its core, Henry’s Law explains something that people observe every day without realizing it: the amount of gas that dissolves in a liquid depends directly on the pressure of that gas above the liquid. When the pressure of the gas increases, more gas dissolves; when the pressure decreases, gas escapes from the liquid. This relationship becomes visible when opening a carbonated drink, when aquatic life relies on dissolved oxygen in water, when divers ascend from deep water, and even when gases circulate between the atmosphere and oceans. Henry’s Law provides the scientific key to understanding all of these events by tying gas solubility to pressure.
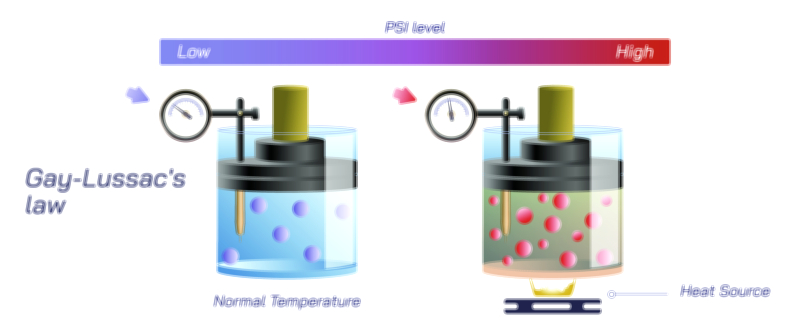
To understand Henry’s Law at a molecular level, imagine a container of liquid in contact with a gas. Gas molecules are constantly moving and colliding with the liquid’s surface. Some of these molecules enter the liquid phase and are temporarily surrounded by liquid molecules. At the same time, some dissolved gas molecules escape back into the gas phase. When the system is left undisturbed, a natural balance forms: the rate at which gas enters the liquid equals the rate at which dissolved gas leaves it. At this point, the gas is said to be in equilibrium with the liquid. Henry’s Law states that the concentration of dissolved gas in this equilibrium increases as the gas pressure above the liquid increases. When the external pressure pushes more gas molecules toward the liquid surface, more of them enter and remain dissolved until a new equilibrium is reached. This microscopic behavior explains why the law is so robust across systems and so essential in understanding gas–liquid interactions.
The principle becomes easy to observe in everyday life when opening a sealed soda bottle. Inside the unopened bottle, carbon dioxide gas is stored under high pressure above the liquid. Because the pressure is high, a large amount of carbon dioxide remains dissolved in the beverage. The moment the bottle is opened, pressure above the liquid suddenly drops. Now the gas is no longer “pushed” into the liquid, so dissolved carbon dioxide begins to escape rapidly, forming bubbles that rise to the surface. Henry’s Law tells us that this happens not because the liquid changed, but because the pressure changed. Once the pressure is released, the solubility threshold drops and the liquid cannot hold as much gas as before. The fizzing that follows is simply the liquid releasing gas until solubility once again matches the lower atmospheric pressure.
A similar concept applies in aquatic environments. Lakes, rivers, and oceans contain dissolved oxygen that fish and other organisms rely upon for survival. When water is exposed to air under normal pressure, oxygen molecules continuously dissolve into it. However, if atmospheric pressure changes or water temperature rises—because warm water holds less gas—the amount of dissolved oxygen can drop. Henry’s Law therefore helps explain seasonal variations in aquatic life and why oxygen levels fall during warm periods. Cold water at high pressure, such as deep ocean water, contains much more dissolved gas than warm shallow water. When deep water rises toward the surface, pressure decreases, allowing some of the dissolved gases to escape. These natural processes are powerful examples of Henry’s Law shaping environmental chemistry.
The law also plays a crucial role in deep-sea diving and hyperbaric medicine. When a diver descends, the pressure of the surrounding water increases significantly. As pressure rises, more nitrogen gas from the breathing mixture dissolves into the bloodstream and tissues. While a diver remains at depth, the nitrogen remains dissolved without causing harm. However, when a diver ascends too quickly, the external pressure drops faster than the dissolved gas can be safely released. Large amounts of nitrogen suddenly come out of solution and form bubbles in the bloodstream, causing decompression sickness, commonly known as “the bends.” This condition is not due to nitrogen entering the body, but to nitrogen leaving too rapidly. Divers avoid this by ascending slowly, giving the dissolved gas time to leave the body gradually, showing how Henry’s Law governs safety in high-pressure environments.
In industrial and scientific settings, Henry’s Law explains why gases are absorbed more effectively under pressure. In water treatment, oxygen is dissolved under pressure to purify wastewater more efficiently. In chemical engineering, carbon dioxide is dissolved into liquids under pressure to produce carbonated beverages. In hyperbaric chambers, higher gas pressures are used to increase dissolved oxygen in the blood for medical treatment. In each case, the principle remains the same: increasing pressure increases solubility; decreasing pressure decreases solubility.
From a conceptual standpoint, Henry’s Law highlights a fundamental interplay between physical conditions and molecular behavior. Gas solubility does not arise from chemical bonding alone; it arises because molecules distribute themselves in proportion to the forces and conditions acting upon them. Pressure determines how frequently gas molecules encounter the liquid surface, and that encounter rate determines how much of the gas dissolves. In a sealed high-pressure system, more gas molecules collide with the liquid than can escape, resulting in high solubility. When pressure falls, molecular escape becomes dominant and dissolved gas leaves the liquid. This relationship emphasizes that solubility is not a fixed property but a dynamic balance controlled by external conditions.
Henry’s Law also interacts with temperature, even though the law itself focuses on pressure rather than heat. In general, gases dissolve better at high pressure and low temperature. Higher temperatures give dissolved gas molecules more kinetic energy, enabling them to escape into the gas phase more easily. This is why warm soda loses fizz faster than cold soda and why cold water holds more oxygen than warm water. Temperature therefore does not contradict Henry’s Law — it changes the equilibrium point at which the pressure–solubility relationship settles.
Another important implication of Henry’s Law appears in atmospheric chemistry. Earth’s oceans absorb large amounts of carbon dioxide from the atmosphere under pressure. When atmospheric CO₂ concentrations rise, the oceans naturally dissolve more of it. While this provides buffering for atmospheric changes, it also contributes to ocean acidification as dissolved CO₂ reacts to form carbonic acid. The equilibrium between CO₂ in air and CO₂ in seawater remains governed by Henry’s Law, showing how a principle of physical chemistry influences climate biology and environmental balance on a planetary scale.
Ultimately, Henry’s Law teaches that the solubility of a gas in a liquid is not random, and it does not depend merely on the chemical nature of the gas or the solvent. Instead, it depends on the thermodynamic condition of pressure, which shifts the balance of molecular movement and determines whether gas molecules remain dissolved or escape. From fizzing drinks to diver safety, from aquatic respiration to atmospheric CO₂ balance, from industrial gas absorption to medical hyperbaric therapy, the principle of pressure-driven solubility influences countless natural and engineered systems.
Through this deeply conceptual view, Henry’s Law emerges not just as a statement about gases, but as a unifying idea connecting microscopic molecular behavior to macroscopic environmental, technological, and biological outcomes. It reveals how the invisible movement of gas particles creates visible effects in everyday life and how controlling pressure enables science and engineering to shape gas solubility to meet human needs.