Homogeneous and Heterogeneous — Types of Mixtures
In chemistry, the distinction between homogeneous and heterogeneous mixtures forms one of the essential foundations for understanding how substances combine, interact, and behave when brought together. These two categories—though seemingly simple—reveal profound insights into the microscopic arrangement of matter, the forces that hold substances together, and the principles governing physical and chemical behavior across natural systems, industrial processes, and everyday phenomena. A mixture, unlike a pure substance, contains two or more components that coexist without undergoing chemical bonding or transformation. Instead, each component retains its own identity, interacting physically rather than chemically. The way these components distribute themselves determines whether the mixture is homogeneous or heterogeneous. This classification highlights the deep relationship between molecular dispersion, uniformity, phase behavior, and the perceptual or analytical distinctions that arise when substances coexist in shared space.
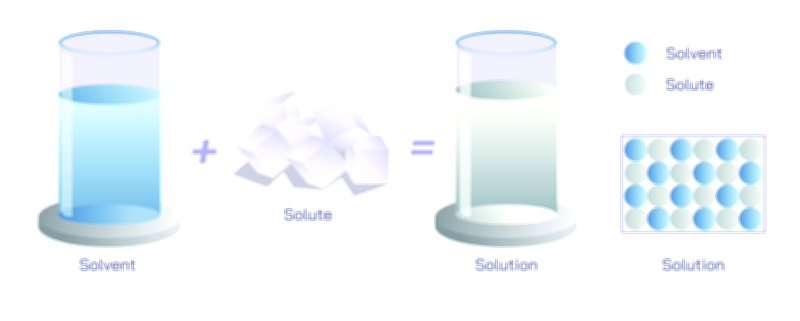
A homogeneous mixture is characterized by uniform composition throughout, meaning that the components are evenly distributed at the molecular or ionic level. No matter which portion of the mixture is sampled, it contains the same proportion of ingredients as any other part. This uniformity results from the intimate mixing of particles, often facilitated by dissolution, diffusion, or continuous blending. Solutions represent the most common type of homogeneous mixture. When salt dissolves in water, for instance, the sodium and chloride ions disperse uniformly among water molecules, forming a single transparent phase. This microscopically even distribution ensures that no visible boundaries or layers form, and the mixture behaves as one continuous medium. Homogeneous mixtures appear consistent because the particles are so small—often molecular or ionic in size—that they do not scatter light, rendering the mixture optically clear in many cases. The concept extends beyond liquids: metal alloys such as brass or steel are solid homogeneous mixtures, where atoms of different elements intermix uniformly within a shared crystalline structure. Gaseous mixtures, such as air, also belong to this category because nitrogen, oxygen, carbon dioxide, and trace gases disperse evenly throughout the atmosphere.
The uniformity of homogeneous mixtures has significant implications for their physical behavior and practical applications. Because the mixture has consistent composition, its properties—such as density, boiling point, or conductivity—remain the same throughout the entire sample. This predictability is crucial in chemical reactions, where reactants must be consistently distributed to ensure controlled and reproducible outcomes. In pharmaceuticals, homogeneous mixing ensures accurate drug dosing, preventing uneven distribution of active ingredients. In food science, beverages such as milk processed through homogenization achieve consistent texture and taste by mechanically dispersing fat droplets so they no longer separate. Even in industrial processes, the creation of homogeneous mixtures supports reliable material performance, whether in synthetic polymers, fuel blending, or coatings. The stability and uniformity of these mixtures reflect the success of achieving microscopic mixing across all components.
In contrast, a heterogeneous mixture contains components that are not uniformly distributed, leading to the presence of distinct regions or phases. The composition varies from one part of the mixture to another, and the individual substances often remain visibly or physically separate. The particles in heterogeneous mixtures may range from microscopic to macroscopic, but they do not intermix at the molecular level. Classic examples include sand and water, oil and water, granite, soil, and mixtures of cereals or powders with different grain sizes. In heterogeneous mixtures, the phases are distinguishable because the components differ in density, solubility, state of matter, or particle size. Some heterogeneous mixtures settle over time, such as muddy water, where heavier particles sink to the bottom under the influence of gravity. Others maintain distinct phases indefinitely, such as oil floating on water due to differences in density and intermolecular attraction.
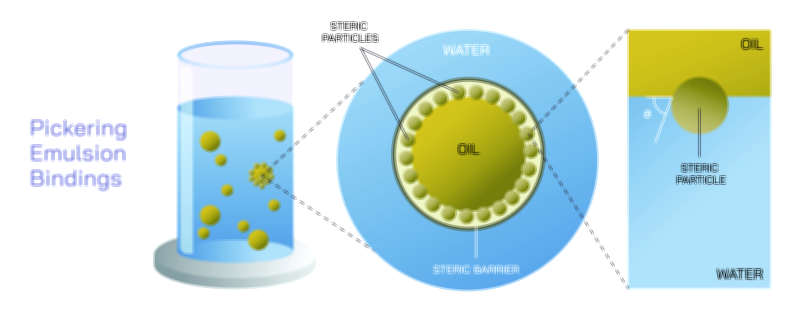
Heterogeneous mixtures exhibit a wide range of structural complexity, from simple two-phase systems to elaborate combinations of solids, liquids, and gases. Suspensions represent one type of heterogeneous mixture where solid particles are dispersed in a liquid but are large enough to settle over time. These particles scatter light, making the mixture cloudy or opaque. Emulsions, while often appearing uniform to the naked eye, are technically heterogeneous because they consist of tiny droplets of one liquid dispersed within another. Milk, mayonnaise, and lotions fall into this category. Although emulsions may seem stable, they contain two immiscible phases held together by emulsifying agents that prevent separation. Colloids, another type of heterogeneous mixture, contain particles that are larger than those in true solutions but smaller than those in suspensions. These particles remain dispersed without settling, creating mixtures such as fog, gelatin, whipped cream, and paints. The Tyndall effect—a scattering of light caused by colloidal particles—reveals their heterogeneous nature even when visual separation is not apparent.
The behavior of heterogeneous mixtures is influenced by the properties of their components and the interactions between them. Differences in solubility determine whether substances will mix or remain separate. Density differences govern layering in mixtures such as oil and water. Particle size influences stability, with smaller particles requiring surfactants or stabilizing agents to prevent aggregation. Temperature and agitation can temporarily alter the degree of mixing, but unless a chemical reaction or dissolution occurs, these mixtures usually revert to their distinct phases. The structural diversity of heterogeneous mixtures makes them essential in countless natural and engineered systems. Soil, for example, is a highly complex heterogeneous mixture of minerals, organic matter, water, gases, and microorganisms. Its ability to support plant life derives from this diverse composition. In medicine, heterogeneous mixtures such as suspensions are used to deliver drugs that cannot dissolve fully. Industrial processes use heterogeneous catalysts, where solid catalysts interact with gaseous or liquid reactants at surface interfaces to accelerate reactions.
The contrast between homogeneous and heterogeneous mixtures reveals much about how matter behaves on molecular and macroscopic scales. A homogeneous mixture represents a high level of molecular dispersion, where substances interact closely enough to create uniformity. A heterogeneous mixture reflects limited molecular interaction, allowing components to retain distinct identities. These differences underscore the role of intermolecular forces, solubility, polarity, and physical properties in determining mixture type. For example, polarity differences explain why oil and water form heterogeneous mixtures: water molecules strongly attract each other through hydrogen bonding, excluding nonpolar oil molecules. Conversely, ethanol and water form homogeneous mixtures because their polarities and ability to hydrogen bond create compatibility at the molecular level.
Mixtures, whether homogeneous or heterogeneous, serve vital roles across science and daily life. In biology, blood behaves partly as a homogeneous solution (dissolved gases and ions) and partly as a heterogeneous mixture (cells suspended in plasma). In environmental science, air pollution represents a heterogeneous mixture of particulate matter within the atmosphere. In food science, the texture, stability, and flavor of products depend on the balance between homogeneous solutions and heterogeneous dispersions. Industrial materials—from reinforced concrete to polymer composites—derive strength and durability from heterogeneous structures. Meanwhile, homogeneous mixtures underpin processes such as metal alloy formation, electrolyte preparation, and solution-based chemical synthesis.
Ultimately, the classification of mixtures into homogeneous and heterogeneous forms is not merely a visual distinction but a reflection of deeper chemical and physical principles. It demonstrates how the size, nature, and interactions of particles determine the structure of matter. Homogeneous mixtures embody complete molecular-level blending, leading to uniform properties and predictable behavior. Heterogeneous mixtures retain their multi-phase identity, offering structural diversity, complexity, and the ability to separate components physically. Together, these categories illuminate the versatility of mixture behavior, showing how substances combine in ways that shape natural environments, technological systems, and chemical processes. Understanding these mixture types provides a fundamental perspective on matter’s organization and enriches the broader study of chemistry, materials science, biology, and environmental science.