Salicylic Acid – Structure, Formula, Functional Groups, Mechanism of Action in Skin Treatment, Pharmaceutical Importance, and Therapeutic Uses in Modern Medicine
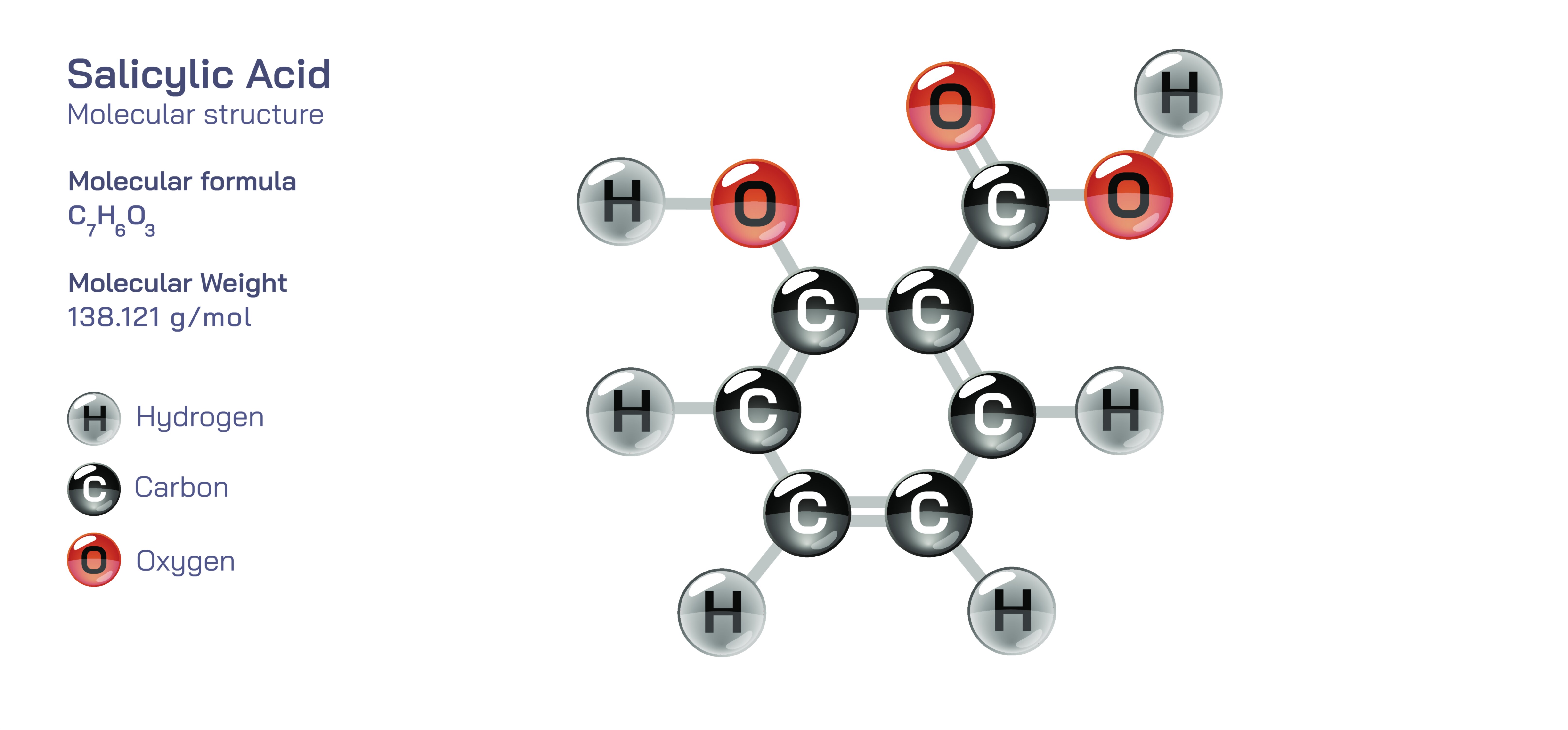
Salicylic acid is one of the most influential compounds in dermatology and pharmaceutical chemistry because its dual functional groups provide both biochemical activity and structural versatility. Its molecular formula is C₇H₆O₃, and its structure consists of a benzene ring substituted with a hydroxyl group (–OH) and a carboxylic acid group (–COOH) positioned ortho to each other (specifically at the 2-position on the ring). This arrangement makes salicylic acid an ortho-hydroxybenzoic acid, combining the aromatic stability of the benzene ring with the acidic reactivity of the carboxyl group and the hydrogen-bonding and phenolic properties of the hydroxyl group. The close proximity of the –OH and –COOH groups enables intramolecular hydrogen bonding, which stabilizes the molecule and influences its solubility, melting behavior, and reactivity. This structural framework not only determines physical properties but also shapes its biological functions, especially its impact on skin and inflammation.
Salicylic acid is classified chemically as a beta-hydroxy acid (BHA) because its hydroxyl group is bonded to the beta carbon relative to the carboxyl group, although structurally it differs from the aliphatic BHAs commonly found in fruit extracts. This aromatic BHA has strong keratolytic, comedolytic, anti-inflammatory, and antimicrobial properties, which explain its remarkable prominence in dermatological medicine. Its ability to break down intercellular connections between dead skin cells allows it to exfoliate the outer layer of the skin and unclog pores, making it exceptionally effective in acne and oily-skin treatment. Unlike water-soluble alpha-hydroxy acids (AHAs) that work mainly on the skin surface, salicylic acid is oil-soluble, enabling it to penetrate deep inside sebum-rich pores and dissolve accumulated oil, debris, and keratin plugs. This deep penetration prevents pore blockage, reduces comedones (blackheads and whiteheads), controls acne formation, and restores a smoother skin texture.
Salicylic acid’s benefits extend beyond exfoliation. It exhibits anti-inflammatory activity by inhibiting prostaglandin synthesis, reducing swelling and irritation in inflamed tissues. This mechanism is related to its historical significance — salicylic acid is the biochemical precursor to acetylsalicylic acid (aspirin). In the body, certain metabolites of salicylic acid modulate inflammatory signaling by interfering with cyclooxygenase (COX) pathways, giving the compound a dual role in both topical and systemic medicine. For skin conditions such as acne, psoriasis, seborrheic dermatitis, warts, corns, calluses, and dandruff, salicylic acid helps soften and dislodge excess keratinized skin, decrease scaling, and reduce itch or discomfort. In wart and callus treatments, higher concentrations of salicylic acid break down thickened keratin layers until the unwanted tissue gradually peels away. This keratolytic power comes directly from the molecule’s ability to loosen cellular adhesion in the stratum corneum, the skin’s outermost layer.
In cosmetic and dermatologic formulations, salicylic acid is present in cleansers, serums, gels, acne patches, peels, exfoliating toners, medicated face washes, dandruff shampoos, and scalp treatments. Concentrations vary depending on the purpose: low strengths (0.5–2%) are common in over-the-counter skincare for daily acne management, while higher strengths are used under professional supervision for chemical peels or wart removal. The oil-soluble, pore-penetrating nature of salicylic acid also helps reduce excess sebum production and bacterial growth inside follicles, making breakouts shorter and less inflammatory. Its mild numbing and soothing effects also reduce redness and irritation associated with acne. Over time, consistent use can reduce blackheads, prevent recurrent acne formation, and improve overall skin clarity and tone.
Salicylic acid also has value beyond dermatology. In oral medicine, it acts indirectly through its derivative aspirin, which is synthesized by acetylation of salicylic acid. Aspirin revolutionized medicine as an analgesic (pain reliever), antipyretic (fever reducer), and anti-inflammatory medication. This transformation shows how modifying functional groups in salicylic acid can shift its physiological outcomes while preserving its anti-inflammatory capabilities. In agriculture, salicylic acid and salicylates act as plant immunity regulators, triggering defense pathways that enhance resistance to pathogens and environmental stress. In food technology and preservatives (historically), salicylic acid prevented microbial contamination before modern safety regulations favored alternative preservatives.
Chemically, salicylic acid participates in esterification, salt formation, and aromatic substitution reactions because of its dual reactive groups. It forms salts known as salicylates, which appear in medicines, muscle-relief balms, lotions, liniments, and medicated oils to provide localized anti-inflammatory effects. It also acts as a precursor for dyes, fragrance intermediates, polymers, and flavoring agents, further demonstrating its versatility. Laboratory modifications of salicylic acid help generate functional ingredients for ointments, hair-care products, sunscreen stabilizers, and pharmaceutical active compounds.
Although salicylic acid is extremely valuable, its usage must be matched with safe concentration and frequency. High levels or excessive application can cause irritation, dryness, peeling, or sensitivity, especially in people with dry or sensitive skin. When applied appropriately — especially in leave-on acne care or fumigation therapy for scalp and hair conditions — it provides visible improvement without damaging deeper skin layers. Because salicylic acid can be absorbed through the skin, medical guidelines ensure safe dosages for children, people with diabetes, and people using other medications. This responsible tolerance-driven approach makes salicylic acid both safe and highly effective when used under proper supervision.
In essence, salicylic acid demonstrates how a precise molecular structure determines biological performance. The benzene ring gives stability and lipophilicity, the carboxyl group contributes acidity and reactivity, and the phenolic hydroxyl group helps mediate hydrogen bonding and anti-inflammatory behavior. Together, these features enable exfoliation, pore cleansing, keratolysis, and inflammation control — a combination unmatched by most other skincare actives. From acne treatment to callus removal, from dandruff therapy to pharmaceutical synthesis, salicylic acid remains one of the most important multifunctional compounds in medicine and cosmetic science. Understanding its structure, formula, and medicinal uses provides insight into how chemical architecture translates into therapeutic value and how a single molecule can shape the future of both pharmaceuticals and skincare.