Glycerin — Chemical Structure, Organic Composition, and Molecular Behavior Explained
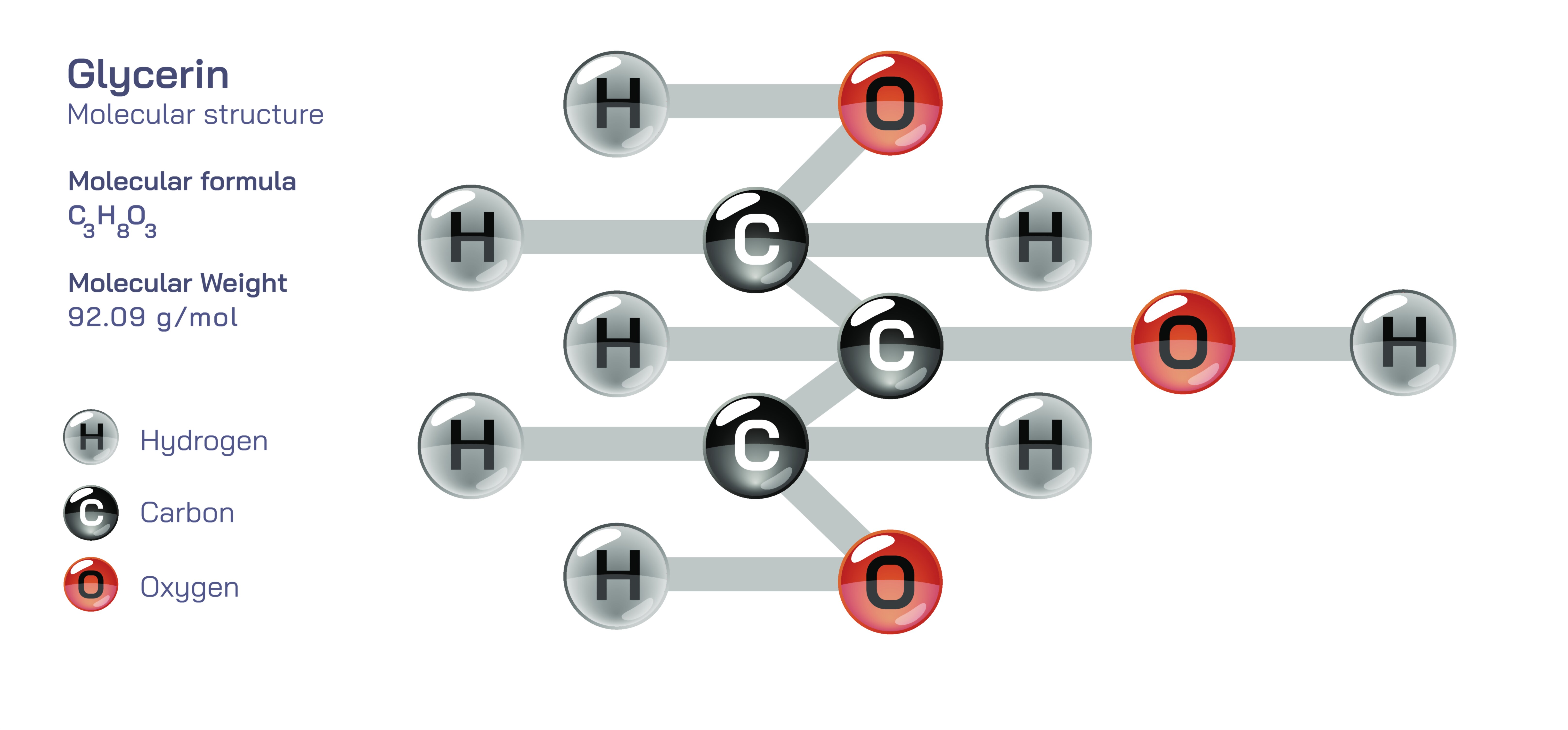
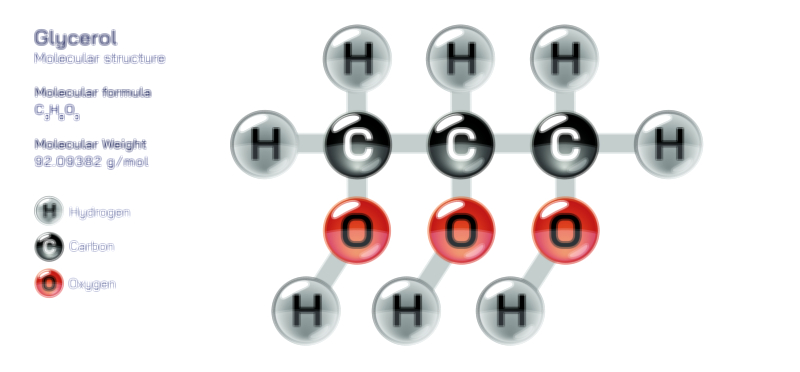
Glycerin, also known as glycerol, is one of the simplest and most important organic compounds in biochemistry and industrial chemistry, yet its true significance becomes clear only when its molecular structure is examined in detail and connected to its physical properties, chemical reactivity, and wide applications. Although diagrams often depict glycerin as a chain of three carbon atoms with attached hydrogen and oxygen groups, understanding the compound conceptually reveals why this small molecule plays such an enormous role in biological systems, pharmaceuticals, cosmetics, food chemistry, and material science. At its core, glycerin is classified as a trihydroxy alcohol, which means it contains three hydroxyl (–OH) groups attached to a carbon skeleton. These three hydroxyl groups shape its identity, making glycerin simultaneously highly polar, strongly hydrophilic, chemically flexible, and exceptionally stable. Every characteristic that makes glycerin useful arises from this structural foundation.
The basic molecular structure of glycerin consists of three carbon atoms arranged in a straight chain, each bonded to hydrogen atoms and hydroxyl groups. The central carbon links symmetrically between the two terminal carbons, producing a compact and balanced geometry. Each carbon carries one hydroxyl group, giving rises to the formula C₃H₈O₃. The oxygen atoms in the hydroxyl groups possess strong electronegativity, creating regions of partial negative charge and enabling the molecule to form extensive hydrogen bonding networks with water and other polar molecules. This makes glycerin extremely soluble in water and allows it to attract moisture from its environment, which is why glycerin remains syrupy, thick, and non-crystalline rather than drying out or evaporating easily. The dense web of intermolecular hydrogen bonds also contributes to its high boiling point, low volatility, and viscous liquid nature—properties that become difficult to understand without a clear picture of its chemical structure.
The three hydroxyl groups also make glycerin chemically reactive in controlled and useful ways. They enable glycerin to participate in condensation and esterification reactions, forming ester bonds with fatty acids. This single structural capability is the reason glycerin is a fundamental component of triglycerides, the molecules that store energy in living organisms. A triglyceride forms when glycerin binds with three fatty acid molecules, each hydroxyl group linking to the carboxyl group of a fatty acid. The resulting molecule becomes the major energy reserve in plants and animals. When living organisms need energy, enzymes break down triglycerides and release glycerin, which is then converted into metabolic intermediates that participate in energy production pathways. This direct chemical connection between glycerin and biological energy storage demonstrates how a simple molecular design becomes a foundation of life chemistry.
Outside biological systems, the same structure gives glycerin remarkable industrial and commercial versatility. Its affinity for water makes it a powerful humectant, a substance that draws and retains moisture. This property is why glycerin appears in cosmetics, creams, lotions, soaps, shaving gels, and hair care products to maintain softness and hydration. In pharmaceuticals, glycerin provides smooth texture, improves solubility for active ingredients, prevents tablets from drying and cracking, and acts as a mild preservative by reducing microbial growth through water-binding effects. In food science, glycerin is used as a sweetener, stabilizer, and moisture-retention agent in products such as baked goods, candies, icings, and nutritional bars. The molecule does not crystallize easily because its hydrogen bonding makes it resistant to forming rigid solid structures, resulting in a constant smoothness valued in many formulations.
The structure of glycerin also determines its thermal and chemical stability, allowing it to withstand high temperatures without decomposition under normal conditions. This stability, combined with its reactivity toward acids, makes glycerin a valuable raw material in chemical synthesis. When reacted with strong oxidizing agents or nitrating mixtures, two of its hydroxyl groups can undergo modification to form nitroglycerin, an energetic compound with explosive potential that also serves as a key cardiovascular drug when administered in controlled doses. This dramatic contrast—between the gentle humectant used in skincare and the energetic compound in explosives and medicine—illustrates how the transformation of specific structural features changes the coarse behavior of a molecule without altering its carbon backbone.
Another significant implication of glycerin’s molecular structure lies in its role in polymer science and materials chemistry. Because of its three hydroxyl groups, glycerin functions as a crosslinking agent that can bind with polymers to adjust flexibility, elasticity, and moisture retention. It is widely used in biodegradable plastics, alkyd resins, water-based paints, and coatings. In personal care and pharmaceutical industries, glycerin acts as a plasticizer—a compound that increases flexibility and reduces brittleness in films and gels. Once again, the molecule's ability to develop multiple hydrogen bonds influences how surrounding molecules arrange themselves, reshaping physical properties on a macroscopic scale.
The structural symmetry and compact nature of glycerin also explain its non-toxicity, biodegradability, and compatibility with biological systems. It is neutral, non-irritating in dilute form, and naturally metabolized in the human body. These qualities make glycerin an ideal component for oral and topical pharmaceuticals, baby products, food ingredients, and artificial tears. Its ability to blend with both water and alcohol also creates a medium in which otherwise insoluble ingredients can be stabilized and dispersed—an effect that simplifies formulation chemistry across many industries.
Even glycerin’s physical feel—smooth, viscous, and non-greasy—can be understood through its molecular shape. The compact arrangement of hydroxyl groups allows glycerin to slip between water molecules without forming surfactant-like films, giving it lubrication without oily residue. The same hydrogen-bonding interactions that hold water molecules together now incorporate glycerin molecules, creating a fluid network that resists crystallization but supports viscosity.
Thus, the value and diversity of glycerin arise not from its size but from the precise placement of three hydroxyl groups along a three-carbon chain. That structure creates a cascade of chemical consequences: hydrophilicity, strong hydrogen bonding, high boiling point, viscosity, reactivity with fatty acids, energy-storage roles in living organisms, humectant behavior in skincare and food, plasticizing and crosslinking ability in materials science, and metabolic compatibility in human physiology. Every property that makes glycerin useful traces back to its molecular architecture.
Understanding glycerin through this deeply conceptual lens—rather than through formula alone—reveals the elegance of organic chemistry: how the arrangement of atoms controls function, how microscopic structure produces macroscopic properties, and how a single molecule can bridge the worlds of biology, medicine, consumer industry, and materials science. Glycerin stands as a clear example of how the discipline of chemistry turns simple molecular frameworks into powerful agents of physical, biological, and industrial transformation.