Glycerol — Chemical Structure, Organic Composition, and Molecular Behavior Explained
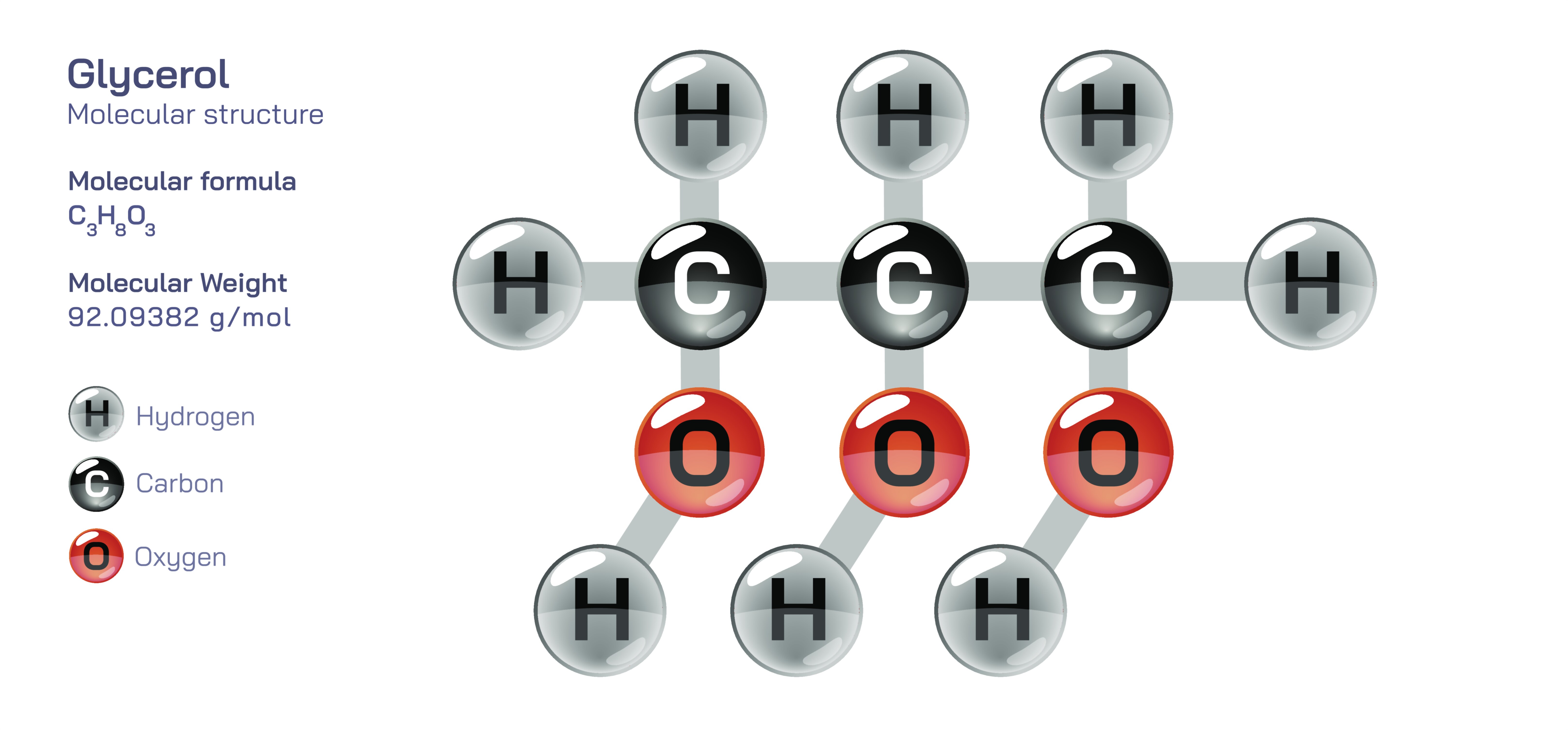
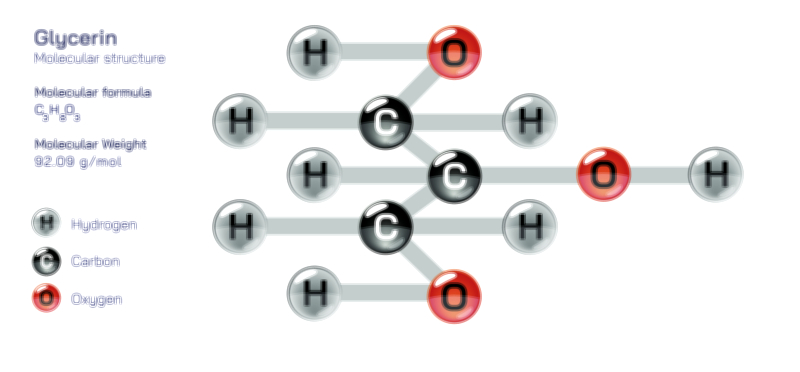
Glycerol, commonly known as glycerin, is one of the simplest yet most influential organic compounds in biological chemistry, industrial manufacturing, and consumer product formulation, and its importance becomes fully clear only when its structure is examined deeply and connected to the many physical and chemical properties that arise from it. Although molecular diagrams often show glycerol as three carbon atoms arranged in a straight chain with oxygen and hydrogen substituents, this simple illustration hides the profound structural logic that explains why glycerol is so soluble in water, why it retains moisture so effectively, why it forms the backbone of fats and oils in living organisms, and why it functions as a stabilizer and humectant in food, pharmaceutical, and cosmetic products. At the molecular level, glycerol belongs to the family of polyhydric alcohols and specifically to the group of trihydroxy alcohols, meaning that its carbon chain contains three hydroxyl groups (–OH), each attached to a carbon atom. This arrangement is not incidental; it is the source of the compound’s polarity, its reactivity, its high boiling point, its viscosity, and nearly every functional role it plays in chemistry and biology.
The structural core of glycerol consists of a three-carbon chain, written conceptually as C–C–C, with each carbon atom bonded to a hydroxyl group. This gives the molecule the general formula C₃H₈O₃, and the spatial arrangement of its atoms creates a compact, almost symmetrical shape. The presence of three hydroxyl groups gives glycerol the ability to form extensive hydrogen bonds with surrounding molecules. The oxygens within the hydroxyl groups carry partial negative charge, while the hydrogens bonded to them carry partial positive charge, allowing constant attraction between neighboring molecules. When glycerol interacts with water, this hydrogen-bonding system becomes even more extensive, producing complete miscibility in any proportion. The same interaction explains glycerol’s role as a moisture-tempering agent: its hydroxyl groups bind water molecules and prevent them from evaporating quickly. This single molecular feature allows glycerol to act as a humectant, a compound that attracts and holds water—one reason it is widely used in lotions, soaps, shampoos, toothpaste, and skincare products where long-lasting hydration and softness are desired.
The physical texture of glycerol also comes from its molecular structure. Because the hydroxyl groups can form hydrogen bonds not only with water molecules but also with adjoining glycerol molecules, the liquid has high internal cohesion and resistance to flow, resulting in high viscosity. Unlike oils, which feel greasy because their nonpolar fatty chains repel water, glycerol feels smooth and soft because its polar structure encourages interaction with skin moisture. At the same time, hydrogen bonding gives glycerol a high boiling point and low volatility, explaining why it stays liquid at room temperature and does not evaporate the way lighter organic compounds do. Even its sweetness arises from the hydroxyl groups: these structural elements interact with receptors on the tongue and give glycerol its mildly sweet taste, even though it is not a sugar in the biochemical sense.
Beyond its physical attributes, the structure of glycerol gives it substantial chemical reactivity, especially with organic acids. Each hydroxyl group can participate in esterification, a reaction that produces esters when an alcohol reacts with a carboxylic acid. In biological systems, this property allows glycerol to bind with long-chain fatty acids to create triglycerides, the primary storage form of energy in animals and plants. In this biochemical role, glycerol forms the central scaffold to which three fatty acid chains attach. The resulting molecule becomes hydrophobic due to the long hydrocarbon tails, even though the glycerol backbone itself is hydrophilic and polar. The remarkable part of this transformation is that the polarity of glycerol gives way to the nonpolar nature of the triglyceride not because of a change in carbon arrangement, but because the hydroxyl groups become part of ester bonds that alter how the new compound interacts with water. Thus, the glycerol backbone supports life’s energy storage strategy—something that cannot be deduced from a simple formula but becomes clear when the structure is examined deeply.
The biological journey of glycerol does not end with its role in fat formation; it also plays a key role when triglycerides break down. When the body needs energy, enzymes release glycerol from triglycerides, and the glycerol molecule enters metabolic pathways, where it is converted into intermediates that help fuel cellular respiration. These transformations highlight how glycerol occupies a central position in metabolism, bridging energy storage in fats and energy extraction in glucose-dependent pathways. Its compatibility with biological systems is rooted in the neutrality of its structure: it carries no ionic charge and dissolves easily in water, allowing it to travel efficiently in the bloodstream without requiring special carriers.
In industrial contexts, glycerol demonstrates a similar structural versatility. The three hydroxyl groups allow it to function as a plasticizer, a compound that increases flexibility and moisture retention in materials such as gelatin capsules, polymer films, adhesives, and coatings. When glycerol is incorporated into plastics or gels, its hydrogen bonding prevents rigid intermolecular packing, making the resulting material softer and more flexible. This same mechanism improves the texture of food products such as protein bars, chewing gum, icing, and baked goods, helping them resist dryness by retaining internal moisture. In pharmaceuticals, glycerol is used as a stabilizing agent in liquid medications and syrups, preventing crystallization and helping dissolved active ingredients remain evenly distributed. Its role as a solvent arises from its polarity and compatibility with both water and alcohols, making it an ideal medium for dissolving compounds that must remain chemically stable over long storage periods.
The chemical structure of glycerol also enables more dramatic transformations in synthetic chemistry. When exposed to nitrating agents under controlled conditions, the hydroxyl groups form nitro esters, producing nitroglycerin, a compound with explosive energetic properties and medical use in vasodilation therapy. This contrast—between glycerol’s gentle moisturizing behavior and nitroglycerin’s powerful reactivity—illustrates how changing just a few functional groups can drastically reshape molecular behavior. It also shows that glycerol acts as a chemical scaffold capable of wide modification without losing its core structural identity.
Because glycerol is biodegradable, non-toxic, and metabolically compatible with the human body, it has become a preferred ingredient in personal care products, medicinal formulations, and food processing. It does not irritate skin in normal concentrations and has mild antimicrobial effects due to its ability to bind water and reduce free-water availability for microbial growth. For similar reasons, glycerol is used in cryopreservation solutions to protect biological cells during freezing, where hydrogen bonding helps prevent ice crystal formation inside cell membranes. This protective capability demonstrates that glycerol’s molecular structure not only influences chemical reactions but also shapes biological survival techniques in sensitive environments.
Ultimately, the behavior of glycerol across biology, industry, and materials science can be traced directly to its three-carbon, three-hydroxyl molecular design. That structure determines how the compound interacts with water, how it reacts with acids, how it forms the backbone of fat molecules, how it remains liquid and viscous at room temperature, how it retains moisture in foods and cosmetics, how it alters the flexibility of polymers, and how it can be chemically transformed into derivatives with vastly different properties. Glycerol demonstrates how organic chemistry grows from a simple foundation: small atomic arrangements generate enormous functional diversity, and the precise positioning of a few key functional groups can determine whether a molecule nourishes biological tissues, moisturizes skin, stabilizes food, or becomes part of an energetic compound with medical or industrial significance.
By examining glycerol beyond formula symbols—through the lens of molecular structure, intermolecular interaction, biochemical function, and practical application—one gains a holistic appreciation for how chemistry underlies everyday materials and living systems. Glycerol stands as a clear example of how structure dictates behavior and how a small, seemingly simple organic compound can influence biological life, industrial manufacturing, and applied science in deeply interconnected ways.