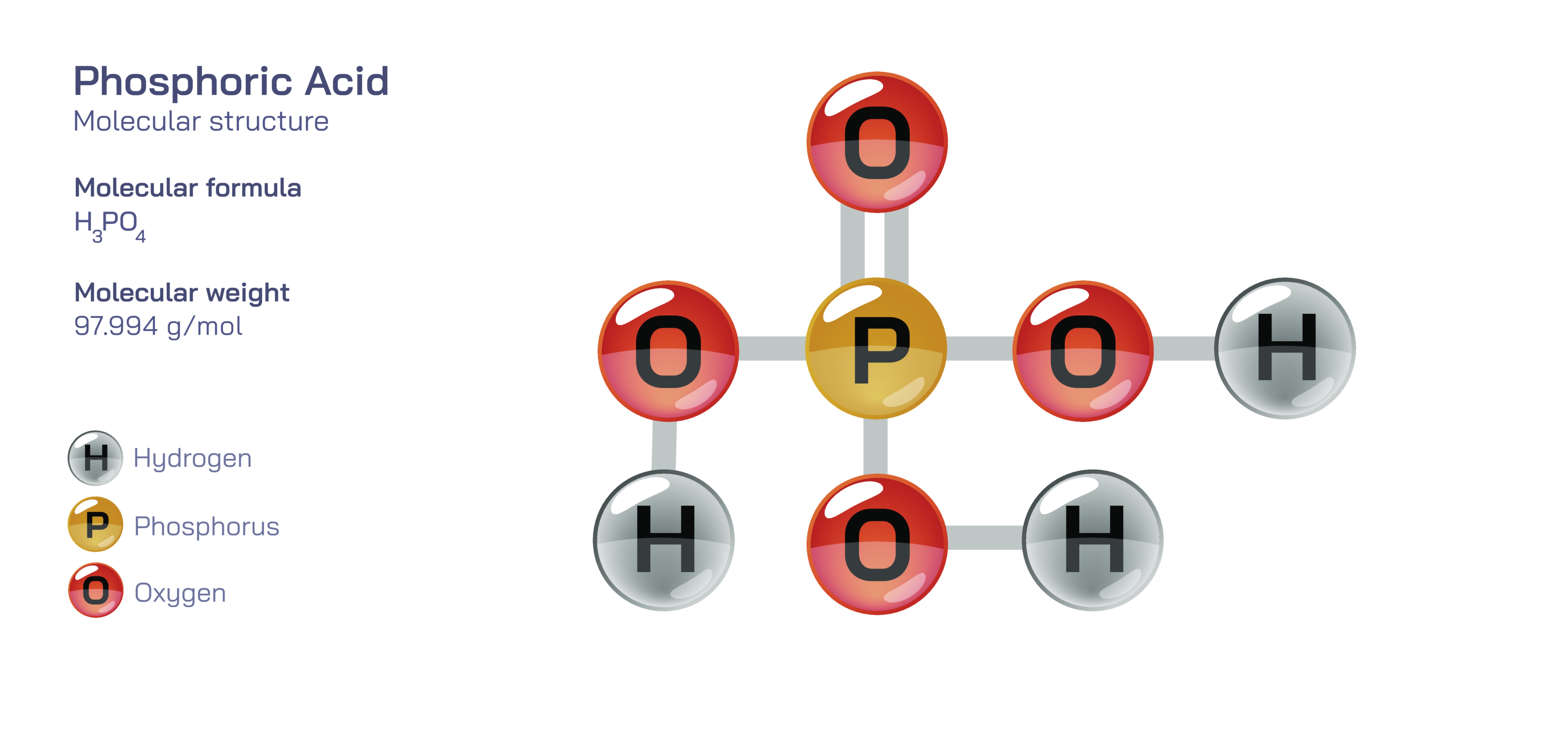
Phosphoric Acid – Structure, Formula, Properties, Industrial Uses, Biological Significance, and Its Role in Modern Chemical Applications
5. Each of the three O–H groups is capable of releasing a hydrogen ion, allowing phosphoric acid to behave as a triprotic acid that dissociates in balanced steps depending on pH and concentration. The stepwise dissociation forms the ions H₂PO₄⁻, HPO₄²⁻, and PO₄³⁻, which play central roles in buffering systems and biological pH regulation. The structure of phosphoric acid therefore links directly to the phosphate family, making it a bridge between molecular chemistry and the essential chemical cycles of living organisms and the environment.
At room temperature, pure phosphoric acid is a clear, odorless, viscous liquid or crystalline solid, depending on temperature and water content. Concentrated industrial phosphoric acid typically contains about 85% H₃PO₄ in water and appears as a thick liquid that mixes readily with water due to extensive hydrogen bonding and high polarity. Even though phosphoric acid is acidic, it is significantly less corrosive than strong mineral acids such as hydrochloric acid, nitric acid, or sulfuric acid. Its moderate reactivity makes it suitable for controlled chemical processes where strong acidity is required without excessive oxidation or aggressive surface attack. The three-step dissociation also means that phosphoric acid produces buffering effects across a wide pH range, which is why phosphate buffering systems are dominant in biological fluids and in laboratory chemistry. In contrast to many acids that exist only in solution, phosphoric acid interacts structurally with minerals, metals, organic molecules, and biological materials, making it versatile in multiple fields.
A large part of phosphoric acid’s global importance is its use in the fertilizer industry. When phosphoric acid reacts with ammonia, it forms monoammonium phosphate (MAP) and diammonium phosphate (DAP), two of the most widely used agricultural fertilizers. These compounds release phosphate ions into soil, enabling plant root development, enzymatic function, photosynthesis, and energy transfer through ATP. Without phosphoric acid–based fertilizers, global crop yields would decline dramatically, threatening food security. However, responsible use is necessary because uncontrolled fertilizer runoff can carry phosphate into rivers and lakes, causing eutrophication and disturbing ecosystems. This dual nature — essential for agriculture yet potentially harmful to the environment — requires careful management and highlights the significant influence of phosphoric acid on ecological systems.
In food and beverages, phosphoric acid serves a very different purpose. When used in small amounts, it acts as a food acidulant that enhances tartness, balances sweetness, controls microbial growth, and stabilizes color and texture. It is widely used in carbonated soft drinks, processed cheeses, bakery products, and meat curing formulations. Because phosphoric acid adds sharp acidity without lemon-like flavor notes, it became one of the signature ingredients that shape the taste of popular cola beverages. In processed food manufacturing, the acid also functions as a preservative that controls pH to inhibit spoilage. Although safe in regulated amounts, continuous consumption of high-phosphate soft drinks has raised concerns about dietary imbalance, especially when intake exceeds the buffering capacity of the body or when calcium consumption is low, linking phosphoric acid indirectly to bone health and mineral balance in vulnerable individuals.
Phosphoric acid also plays a major role in pharmaceuticals and healthcare. It appears in certain dental and orthodontic procedures where buffered phosphoric acid gels are used to etch tooth enamel prior to bonding, creating a micro-roughened surface that improves adhesion of composite resins and brackets. In medicinal formulations, phosphoric acid is used both as an acidifying agent and as a component of phosphate buffers that stabilize liquid medicines. It is found in some anti-nausea remedies, oral solutions, and vitamin supplements where controlled acidity improves absorption or preservation. The importance of phosphoric acid in dental and pharmaceutical applications arises from its controlled corrosive action, water compatibility, and predictable dissociation behavior.
Industrial surface treatment and metal processing are two additional areas where phosphoric acid is indispensable. When applied to metal surfaces, phosphoric acid reacts with rust, converting iron oxide into a layer of iron phosphate that adheres strongly and prevents further corrosion. This reaction simultaneously dissolves loose rust and passivates the surface, making phosphoric acid a widely used ingredient in rust removers, automotive metal cleaners, and surface preparation treatments prior to painting or electroplating. The resulting phosphate coating improves paint adhesion, enhances durability, and reduces corrosion formation. This application illustrates how an acid can serve as a protective agent rather than only a destructive one when its molecular properties are properly harnessed.
In laboratory chemistry and analytical science, phosphoric acid is valued for its predictable acid–base equilibria. Phosphate buffer systems derived from phosphoric acid maintain stable pH in biological experiments, enzyme reactions, chromatography processes, and molecular biology protocols. Because living cells and enzymes function only in narrow pH ranges, phosphate buffers provide an essential environment for preserving biological activity. Phosphoric acid also participates in organic synthesis as a catalyst for dehydration reactions, esterification, and polymerization. In polymer chemistry, it plays a role in the creation of flame retardants, plasticizers, and specialty materials that require thermal stability.
Energy and material technology represent expanding fields of application for phosphoric acid derivatives. In high-efficiency lithium iron phosphate batteries (LiFePO₄), phosphate provides a stable lattice structure that improves battery safety and longevity compared to older cathode materials. Phosphate-based electrolytes are also being researched for fuel cell systems operating at high temperatures. In flame-retardant materials used in textiles, construction, and electronics, phosphate compounds inhibit combustion by promoting char formation and reducing flammable gas release. These modern uses reflect how the chemistry of phosphoric acid continues to align with emerging technological needs.
In nature and biological systems, phosphoric acid exists mainly in the form of its conjugate bases — dihydrogen phosphate and hydrogen phosphate — which regulate pH in blood, control enzyme activation, and participate in metabolic pathways. In skeletal systems, reaction of phosphate with calcium forms hydroxyapatite, the mineral responsible for the hardness of bone and teeth. In energy metabolism, bonding and breaking of phosphate groups in ATP govern cellular activity. In genetic molecules, phosphate groups connect nucleotides to create the sugar–phosphate backbone of DNA and RNA. These biological roles illustrate that phosphoric acid supports both the structure and function of life.
Across all these scientific and technological areas, phosphoric acid demonstrates the core idea that a molecule’s structure determines its behavior, and that small variations in molecular function can impact the world on scales ranging from microscopic cells to global industry. The tetrahedral arrangement, stepwise acidity, strong affinity for metals, compatibility with water, and ability to form stable phosphate derivatives give phosphoric acid a unique identity that few chemical compounds match. Through its functions in agriculture, materials science, industrial chemistry, medicine, energy storage, food production, and biochemistry, phosphoric acid has become a molecule that quietly supports modern civilization. Understanding its structure, formula, and uses means understanding a chemical foundation that links the inorganic world to life, technology, and the sustainable future of our planet.