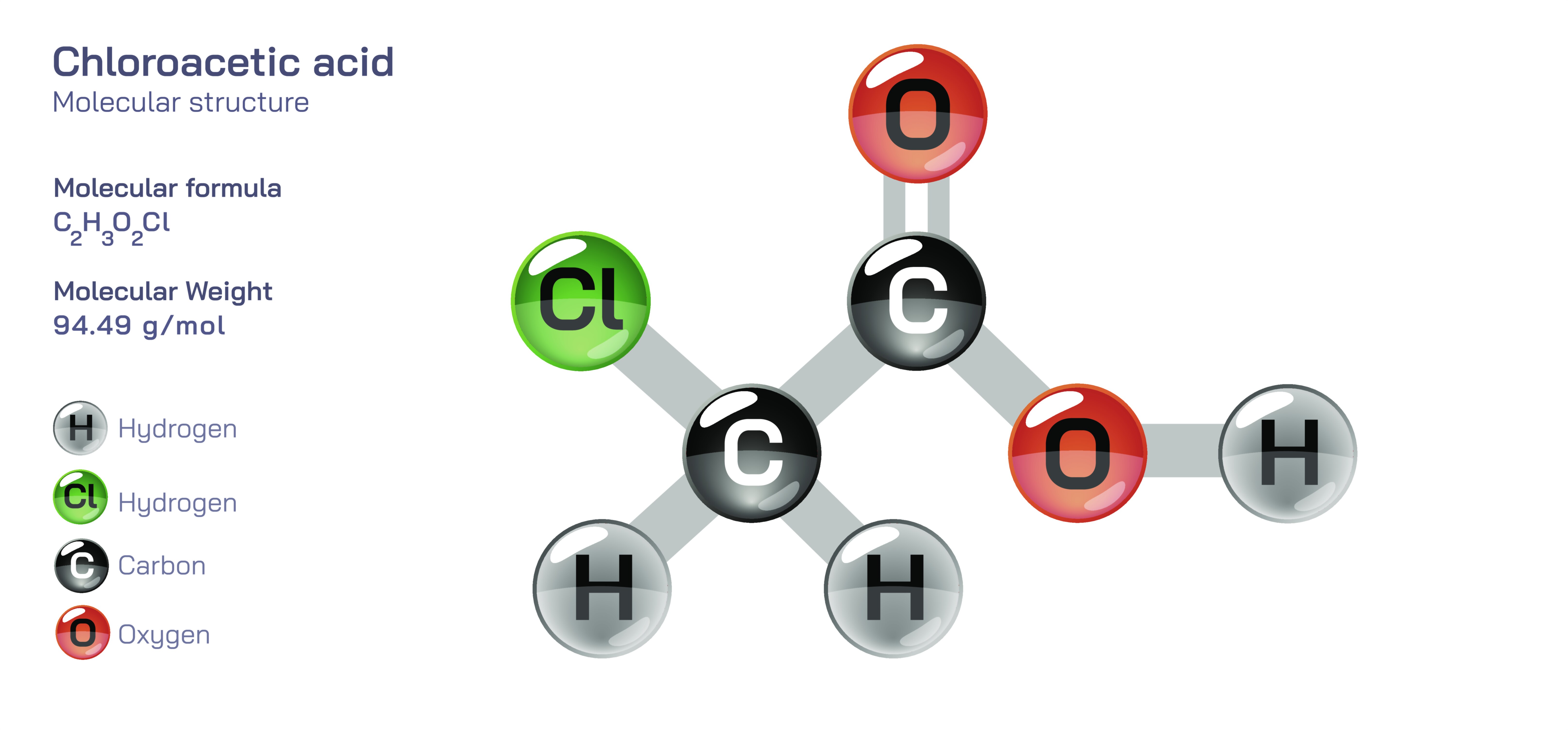
Chloroacetic Acid — Halogenated Organic Compound
Chloroacetic acid is a remarkably influential halogenated organic compound, known for its potent reactivity, its central role as a building block in chemical synthesis, and its profound impact across agriculture, industry, biochemistry, and materials science. Structurally simple yet chemically powerful, chloroacetic acid embodies the transformative effects that halogen substitution can impose on an organic molecule. By replacing one hydrogen atom of acetic acid with a chlorine atom, the compound acquires dramatically enhanced electrophilicity and reactivity, enabling it to participate in a wide range of chemical transformations with a degree of precision and intensity not possible with unsubstituted acetic acid. This amplified behavior makes chloroacetic acid not merely a derivative of a familiar compound but a distinctive molecular tool used to synthesize dyes, pharmaceuticals, surfactants, herbicides, and numerous specialty chemicals that shape modern industry. It is a gateway into understanding how small structural changes radiate through chemical systems, altering interactions, reactivity, and utility in profound ways.
At the molecular level, chloroacetic acid consists of a two-carbon chain bearing a carboxyl functional group and a chlorine substituent on the alpha carbon. This chlorine atom dramatically increases the electrophilic nature of the molecule, rendering the alpha carbon far more susceptible to nucleophilic attack than in ordinary acetic acid. This shift in reactivity stems from chlorine’s strong electron-withdrawing effect, which stabilizes the negative charge that develops during substitution reactions and encourages transformations that form carbon–heteroatom bonds. As a result, chloroacetic acid serves as a highly effective intermediate for introducing a wide variety of functional groups into organic frameworks. The molecule’s dual nature—combining the acidity of the carboxyl group with the reactivity of the chlorine-bearing carbon—allows it to participate in esterification, substitution, condensation, and other key organic reactions, making it an indispensable reagent for chemists seeking versatile, reactive starting materials.
One of the most important industrial applications of chloroacetic acid lies in the production of carboxymethyl cellulose (CMC), a widely used thickener, stabilizer, and binding agent. By reacting chloroacetic acid with cellulose under carefully controlled conditions, hydroxyl groups in the cellulose backbone are substituted with carboxymethyl groups. The resulting polymer is water-soluble and forms stable gels and solutions used in food products, pharmaceuticals, cosmetics, detergents, adhesives, paper coatings, and oil drilling fluids. CMC’s ability to adjust viscosity, retain moisture, stabilize emulsions, and form smooth textures demonstrates how a single chemical transformation driven by chloroacetic acid can lead to materials with broad technological significance. Without chloroacetic acid’s unique reactivity, the consistent and efficient production of CMC would be far more challenging, underscoring the compound’s foundational role in modern materials science.
Chloroacetic acid is also essential in synthesizing various herbicides, particularly those that disrupt plant growth by interfering with metabolic or physiological pathways. Compounds such as 2,4-dichlorophenoxyacetic acid (2,4-D), one of the most widely used herbicides in the world, are prepared through pathways that involve chloroacetic acid as a key precursor. This role situates the compound within global agriculture, where its reactivity helps create products that manage weeds, support crop yields, and sustain large-scale food production. Although herbicides derived from chloroacetic acid have generated debate over environmental impact and responsible use, their fundamental chemistry depends heavily on this small halogenated acid’s capacity to form essential synthetic intermediates. In this way, chloroacetic acid acts as a molecular bridge between organic synthesis and agricultural technology.
In pharmaceuticals, chloroacetic acid provides pathways for generating active ingredients, intermediates, and functionalized molecules with therapeutic potential. Its ability to introduce carboxymethyl or other substituted groups makes it useful in modifying molecular backbones, enhancing solubility, adjusting metabolic stability, and preparing compounds that interact with biological targets. Some sedatives, anesthetics, and anti-inflammatory agents trace their origins to synthetic processes involving chloroacetic acid. Because medicinal chemistry depends heavily on precise structural modification, chloroacetic acid’s predictable reactivity allows chemists to design molecules with properties that support absorption, targeted distribution, and biological activity. This process reflects how even hazardous reagents, when handled responsibly, contribute to life-saving advancements in healthcare.
Chloroacetic acid also finds widespread use in surfactant production. Surfactants rely on molecular structures that balance hydrophobic and hydrophilic components, and chloroacetic acid enables the introduction of polar, water-loving groups into larger organic molecules. Through nucleophilic substitution, alcohols, amines, and other substrates can be transformed into functionalized compounds that reduce surface tension, emulsify oils, and support cleaning formulations. These surfactants appear in detergents, shampoos, cosmetics, industrial cleaners, and agricultural sprays. The conversion of simple organic molecules into complex surfactants through the involvement of chloroacetic acid showcases how fundamental organic reactions give rise to substances that permeate nearly every aspect of daily life.
Despite its extensive industrial value, chloroacetic acid is a highly hazardous compound that demands careful handling. It is corrosive to skin and mucous membranes, capable of causing severe burns, and dangerous if inhaled or ingested. Its strong reactivity allows it to penetrate tissues rapidly, producing systemic toxicity that affects cellular metabolism and organ function. This danger highlights the tension between chemical utility and chemical risk, demonstrating why responsible storage, protective equipment, ventilation, and training are essential when working with chloroacetic acid. Industrial systems designed to manufacture or transport the compound employ strict safeguards, including closed reactors, controlled temperature conditions, and continuous monitoring. These precautions ensure that its valuable chemical reactivity is applied safely without exposing workers or the environment to unnecessary harm.
Environmentally, chloroacetic acid and related haloacetic acids can appear as byproducts of chlorine-based water disinfection, leading to concerns about their persistence and potential toxicity to aquatic life. Water treatment facilities monitor levels of haloacetic acids to ensure compliance with safety standards and to minimize formation during purification. Although chloroacetic acid readily degrades under certain environmental conditions, its presence in water sources requires careful oversight. These considerations have prompted research into alternative disinfection techniques and improved monitoring technologies. At the same time, chemists have studied pathways for environmentally friendly decomposition of chloroacetic acid, increasing the understanding of how halogenated compounds interact with natural systems.
From a chemical perspective, chloroacetic acid exemplifies the effect that halogenation can have on molecular behavior. The introduction of a halogen atom reshapes acidity, electrophilicity, and reactivity, providing a clear demonstration of structure–function relationships at the molecular level. Students of chemistry often encounter chloroacetic acid early in their study of reaction mechanisms because it illustrates how inductive effects influence reaction rates, how leaving groups determine substitution outcomes, and how halogenation can be exploited in synthesis. Its reactivity becomes a kind of teaching tool, bridging abstract concepts with tangible laboratory results.
Chloroacetic acid also reminds us that the distinction between helpful and harmful chemicals lies not in their intrinsic nature but in how humans manage and apply them. While it can cause serious injury or environmental damage if mishandled, it also serves as a keystone reagent enabling the creation of safe pharmaceuticals, agricultural tools, sustainable materials, and countless consumer products that improve daily life. Its presence in global industries highlights the importance of chemical literacy, safety engineering, and regulatory frameworks that ensure its benefits outweigh its risks.
Ultimately, chloroacetic acid stands as a powerful example of halogenated organic chemistry at its most influential. With its simple carbon framework enhanced by the presence of chlorine, it becomes a versatile synthetic intermediate that fuels modern manufacturing, supports agricultural productivity, advances materials science, and contributes to medical innovation. Its reactivity serves as both a tool and a challenge—one that demands respect, precision, and scientific insight. By understanding chloroacetic acid, we gain not only a deeper appreciation for organic reactivity but also a clearer view of how fundamental chemical principles shape the technologies and products that define contemporary life.