Phosphate – Structure, Function, and Applications: Molecular Architecture, Biological Importance, Environmental Role, Industrial Value, and the Global Significance of One of the Most Essential Chemical Species
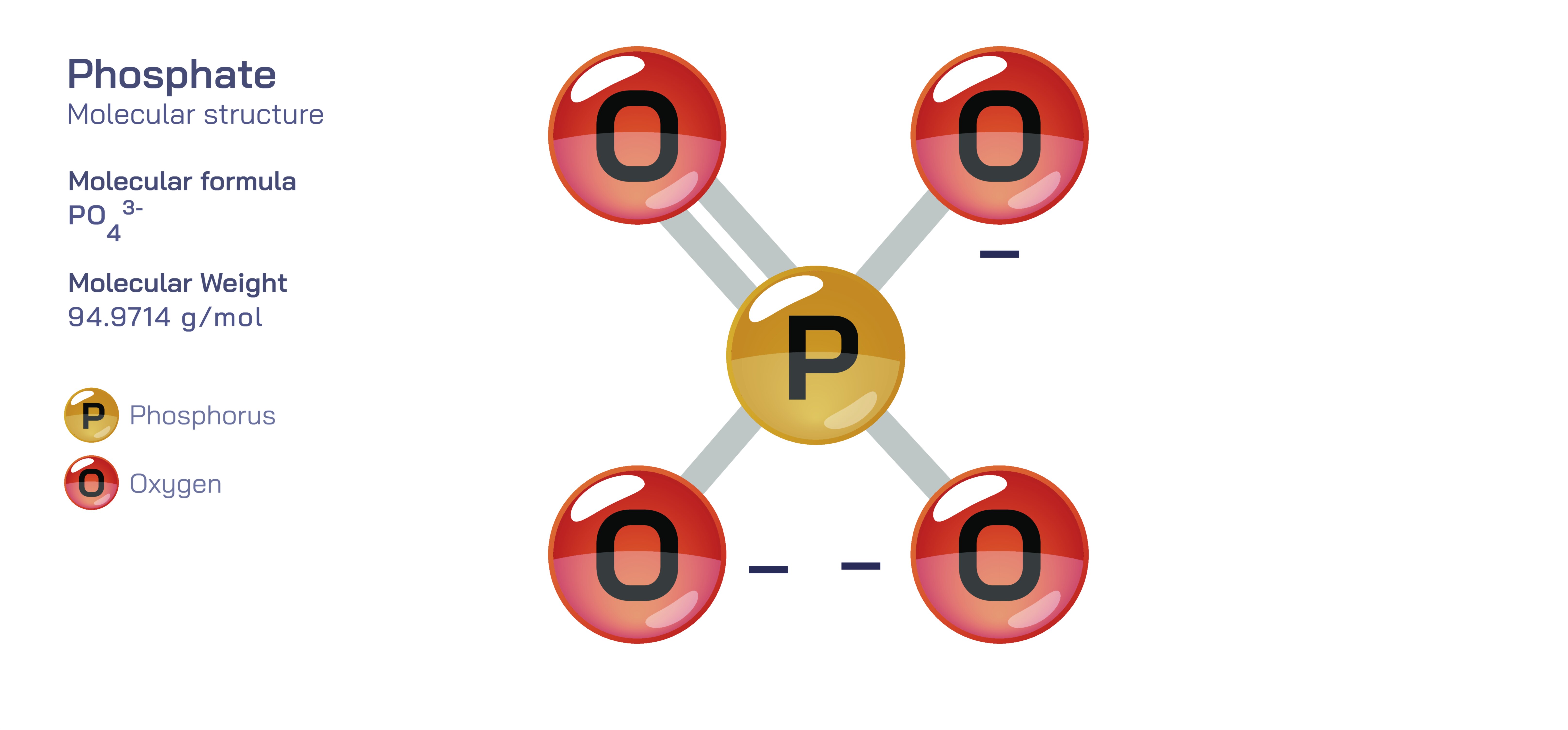
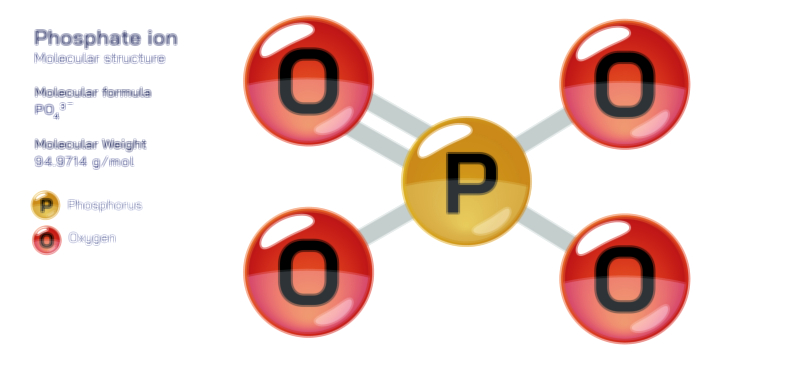
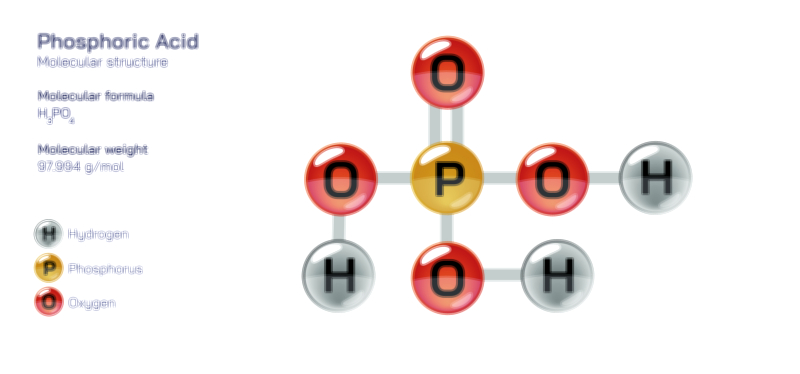
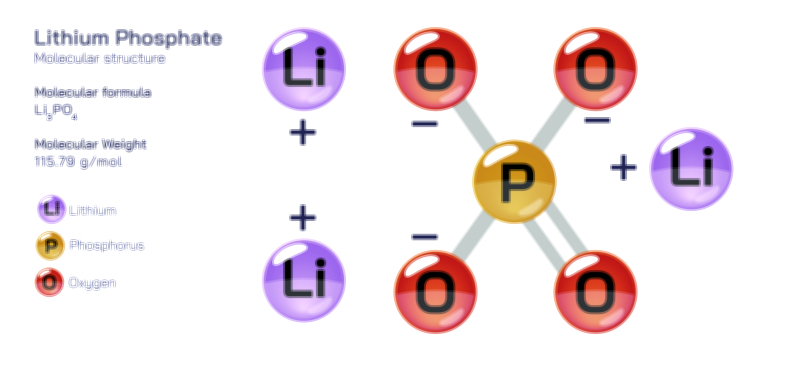
Phosphate stands at the center of chemistry, biology, agriculture, and industrial development because its molecular structure enables a combination of stability and reactivity that is rare among inorganic ions. Represented by the chemical formula PO₄³⁻, the phosphate ion consists of a phosphorus atom surrounded by four oxygen atoms in a tetrahedral arrangement. This geometric structure arises from covalent bonding between phosphorus and oxygen, reinforced by extensive resonance that distributes electron density evenly across the ion. The overall −3 charge results from the deprotonation of phosphoric acid, and this strong negative charge makes phosphate highly effective at forming ionic bonds, coordinating metal ions, participating in acid–base reactions, and linking to organic molecules. The tetrahedral structure not only creates symmetry but also forms the basis for many of phosphate’s functions in biochemistry and geochemical processes. Because the phosphate ion exists at the intersection of structural rigidity and chemical adaptability, it helps organize molecular systems ranging from DNA strands to mineral deposits and industrial materials.
Structural Characteristics and Chemical Behavior
The tetrahedral arrangement of the phosphate ion plays a crucial role in its stability and reactivity. One of the key chemical features of phosphate is its resonance stabilization. Instead of a single P=O double bond and three P–O single bonds existing independently, electrons in the phosphate ion are delocalized, allowing each oxygen atom to share partial double-bond character. This delocalization spreads the negative charge evenly and lowers the energy of the ion, giving phosphate exceptional chemical durability. Chemically, phosphate acts as a base and a nucleophile and can bind strongly to positively charged metal cations such as calcium, magnesium, and iron. Because phosphate is the fully deprotonated form of phosphoric acid, it participates in a series of acid–base equilibria represented by H₃PO₄ → H₂PO₄⁻ → HPO₄²⁻ → PO₄³⁻. These transformations allow phosphate to act as a buffer in biological and laboratory solutions, resisting rapid shifts in pH and stabilizing chemical reactions. Its charge distribution and geometric compatibility with other ions enable phosphate to form crystals, stabilize proteins, coordinate with surfaces, and participate in polymer structures.
Biological Functions – The Foundation of Life Processes
Phosphate plays a role so fundamental in biology that life as we know it could not exist without it. In DNA and RNA, phosphate groups connect sugar molecules through phosphodiester bonds, creating the sugar-phosphate backbone that maintains the structure of genetic material. This backbone is both chemically stable and flexible enough to permit twisting into the double helix or forming messenger RNA strands. The negative charges on the phosphate groups prevent DNA strands from collapsing into each other, ensuring separation during replication and transcription. Phosphate is equally vital in energy management. Adenosine triphosphate (ATP) — the primary energy currency of cells — stores energy in the high-energy bonds between phosphate groups. When ATP releases a phosphate to become ADP (adenosine diphosphate), usable energy powers processes such as muscle contraction, nerve signaling, active membrane transport, biosynthesis, and enzyme regulation. Phosphate appears again in phospholipids, essential building blocks of cell membranes, where phosphate-containing head groups make membranes amphiphilic, enabling them to self-assemble into bilayers that protect and organize cellular life. In proteins and enzyme regulation, phosphorylation acts as a switch that activates or deactivates biological pathways. This ability to control biochemical reactions demonstrates phosphate’s function as both a structural component and a signaling participant in living systems.
Geochemical and Environmental Importance
Phosphate cycles through ecosystems and geological deposits, linking soils, oceans, organisms, and minerals. In nature, phosphate occurs chiefly in minerals such as apatite, formed from calcium phosphate in rocks. Weathering releases phosphate into soil and water, where it becomes an essential nutrient for plant growth, playing a central role in photosynthesis, root development, and metabolic energy transfer. However, phosphate availability in soil is often limited because it binds strongly to minerals, especially in acidic or alkaline conditions, preventing plants from absorbing it efficiently. This limitation makes phosphate one of the most critical nutrients controlling global crop production. Human consumption of phosphate fertilizers has increased agricultural yields dramatically, but excessive use creates environmental challenges. Phosphate runoff from agricultural fields and detergents can accumulate in lakes and rivers, stimulating excessive algal growth and triggering eutrophication — a process that depletes oxygen and harms aquatic ecosystems. Thus, phosphate is essential for global food production but must be managed responsibly to maintain ecological balance and water quality.
Industrial and Technological Applications
Beyond biology and agriculture, phosphate is deeply embedded in manufacturing, materials science, and consumer technology. Fertilizers that contain monoammonium phosphate, diammonium phosphate, or superphosphate are fundamental to sustaining global agriculture and feeding billions of people. In water treatment, phosphate prevents scale formation in pipes and boilers by binding calcium and magnesium ions, while in detergents, polyphosphates improve cleaning by softening water and sequestering metals. Phosphate-based flame retardants protect fabrics, plastics, and construction materials by reducing flammability and limiting the spread of fire. In metallurgy, phosphate coatings protect steel from corrosion and improve the adhesion of paint and polymers. In the field of energy storage, lithium iron phosphate (LiFePO₄) is a highly stable and safe cathode material used in rechargeable batteries for electric vehicles and power backup systems. In pharmaceuticals, phosphate salts stabilize formulations, buffer pH, and serve as counterions in drug molecules. Across all these industrial sectors, phosphate’s chemical stability, buffering capacity, metal-binding strength, and structural compatibility make it an invaluable raw material.
Role in Food, Medicine, and Human Health
Phosphate also influences nutrition and metabolic health. In the human body, phosphate is essential for bone and teeth mineralization, where it joins calcium to form hydroxyapatite — the rigid mineral that strengthens skeletal structures. Phosphate also plays a role in maintaining blood pH balance, supporting muscle and nerve function, and forming metabolic intermediates in carbohydrate and fat processing. Dietary phosphate occurs naturally in protein-rich foods such as meat, dairy products, nuts, legumes, and grains, and is also present in processed foods as additives for preservation, flavor enhancement, and moisture retention. While deficiency can lead to bone weakness and metabolic dysfunction, excessive phosphate intake — especially from processed foods — may contribute to kidney stress and cardiovascular disorders in vulnerable individuals, underscoring the importance of balanced intake and medical monitoring.
Scientific and Educational Significance
The study of phosphate brings together concepts from general chemistry, biochemistry, environmental science, and industrial technology. Its tetrahedral molecular structure illustrates covalent and ionic bonding, resonance, and charge distribution. Its buffering function teaches acid–base equilibrium and pH regulation. Its biological role reveals the molecular mechanics of heredity, energy, and membrane architecture. Its interaction with minerals links chemistry with geology, agriculture, and ecology. Its participation in industrial formulations connects laboratory science with real-world applications. From a scientific perspective, phosphate is a model system that demonstrates how molecular structure leads directly to function — a core idea that underpins all of chemistry and biochemistry.
Ultimately, phosphate represents far more than its PO₄³⁻ chemical formula. It is a molecular cornerstone of life, a driver of agriculture, a component of modern technology, and a regulator of ecological health. Whether working inside DNA, fueling metabolism through ATP, nourishing crops through fertilizer, supporting manufacturing materials, buffering industrial processes, or storing energy in lithium-phosphate batteries, phosphate continues to shape the biological and technological world. Understanding its structure, function, and applications offers insight not only into molecular science but into the sustainability and advancement of human civilization.