Phenol – Chemical Structure, Bonding Nature, Molecular Behavior, Physical and Chemical Properties, Biological Relevance, and Industrial Importance of This Fundamental Organic Compound
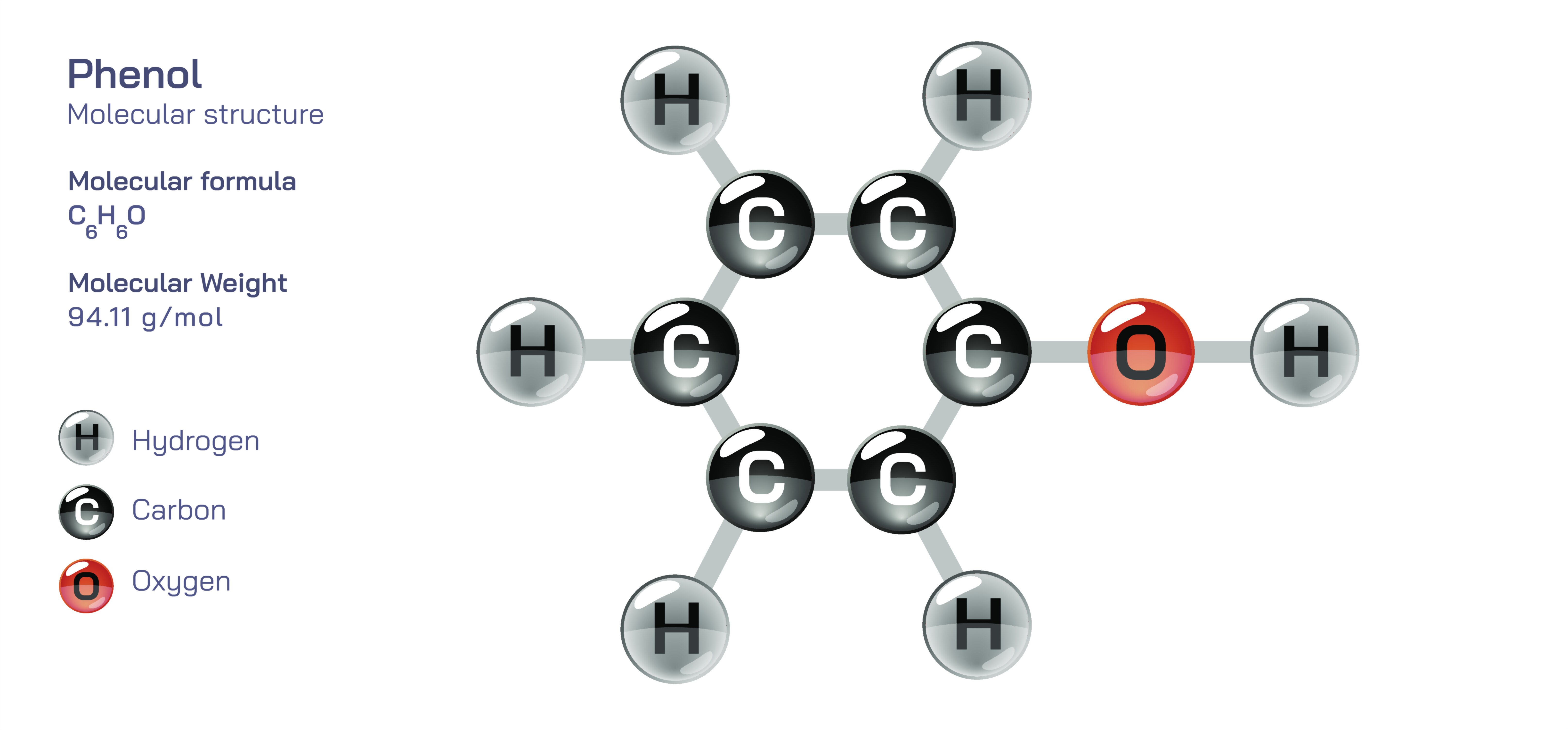
Phenol stands as one of the most influential and foundational molecules in organic chemistry because its structure unites the aromatic stability of a benzene ring with the strong polarity and reactivity of a hydroxyl group. Written chemically as C₆H₅OH, phenol consists of a six-carbon aromatic ring directly bonded to an –OH group, forming a molecule that belongs to a special class of organic compounds known as aromatic alcohols or phenolic compounds. Unlike ordinary alcohols such as ethanol or propanol, phenol behaves differently because the hydroxyl group interacts with the π-electron system of the benzene ring, modifying the molecule’s acidity, solubility, resonance, reactivity, and intermolecular forces. This dual identity — part aromatic ring and part polar functional group — gives phenol a unique chemical personality that influences almost every property it exhibits, making it central to the study of organic reactivity, industrial chemistry, polymer science, pharmaceuticals, and even biological processes.
Chemical Structure and the Influence of Aromaticity on the –OH Group
The structure of phenol contains a planar benzene ring in which carbon atoms are arranged in a perfect hexagon, each connected by alternating single and double bonds. However, due to resonance, these double bonds are not fixed; rather, electrons are delocalized across the entire ring, producing equivalent C–C bonds and reinforcing the molecule’s stability. The hydroxyl group attaches directly to one of the carbon atoms and becomes part of the electron network. When the –OH group donates electron density through resonance, it increases the electron richness of the aromatic ring, particularly at the ortho and para positions. This effect explains why phenol undergoes electrophilic aromatic substitution far faster than benzene. The resonance interaction also spreads across the oxygen atom, making its lone pair partially delocalized into the aromatic ring — a phenomenon that directly contributes to phenol’s acidity and reactivity profile.
Phenol is far more acidic than typical alcohols, even though the molecular structure suggests they share similar functional groups. This acidity emerges from resonance stabilization of the phenoxide ion (C₆H₅O⁻), which forms when phenol loses a proton. In ordinary alcohols, deprotonation yields an alkoxide ion that lacks appreciable resonance stability, making the reaction unfavorable. In phenol, however, the negative charge of the phenoxide ion becomes partially distributed over the aromatic ring through resonance. This stabilization reduces the energy barrier for losing a proton, making phenol a weak acid that can react with strong bases like NaOH but not with carbonates or weaker bases. As a result, phenol occupies a conceptual middle ground between alcohols and carboxylic acids — less acidic than carboxylic acids but far more acidic than saturated alcohols, highlighting how chemical structure affects proton transfer behavior.
Physical Properties Arising from Polarity, Hydrogen Bonding, and Aromaticity
Phenol exhibits physical properties that reflect the combination of its polar and aromatic features. In pure form, phenol appears as white, needle-like crystals or a colorless to pale pink solid at room temperature, with a melting point of around 40–43°C. Its melting point is unusually high for an aromatic compound because hydrogen bonding between phenol molecules forms a strong cohesive lattice. This same hydrogen bonding gives phenol a boiling point of around 181–182°C, considerably higher than that of benzene or toluene. Phenol dissolves moderately in water due to its ability to form hydrogen bonds with water molecules, and its solubility increases significantly with temperature. The molecule exhibits a distinctive medicinal odor that is easily recognizable and historically contributed to its early medical and antiseptic applications.
Chemical Reactivity — From Electrophilic Substitution to Oxidation and Ester Formation
Phenol’s reactivity is one of its defining features, and its interaction with the aromatic ring makes chemical transformation both selective and predictable. The electron-rich ring favors electrophilic aromatic substitution at the ortho and para positions. As a result, nitration, halogenation, sulfonation, and Friedel–Crafts type reactions occur more readily than in benzene and often without the need for strong catalysts. This high reactivity enables phenol to act as a building block for dyes, polymers, pharmaceuticals, and fragrances. The hydroxyl group itself also participates in key reactions. Phenol reacts with acyl chlorides or acid anhydrides to form esters, with alkyl halides to form ethers, and with formaldehyde to initiate polymerization — leading to the formation of phenol-formaldehyde resins (Bakelite), one of the first synthetic plastics ever produced. These diverse reactions show how phenol functions as a versatile reagent in organic synthesis.
Oxidation represents another important aspect of phenol chemistry. In the presence of oxidizing agents, phenol can form quinones, colored complexes, or polymeric oxidation products depending on reaction conditions. These oxidation behaviors play crucial roles not only in organic laboratories but in biological systems. Many plant-derived phenolic compounds, including flavonoids and tannins, undergo oxidation during ripening, browning, and defensive biochemical reactions, showing how phenol-based structures contribute to ecological and metabolic functions. Phenolic antioxidants in food and plant tissues, in contrast, work by donating hydrogen atoms and stabilizing free radicals through resonance — a mechanism that also stems from the phenolic chemical architecture.
Biological Activity and Toxicity — A Dual Identity
Phenol holds a complex position in biological and medical history. It was one of the first antiseptics used in surgery, introduced by Joseph Lister in the 1800s to disinfect instruments and wounds. Its strong antimicrobial action results from protein denaturation and disruption of cell membranes. However, this same destructive capability makes concentrated phenol corrosive and toxic to human tissues. Skin exposure can cause burns, systemic absorption, cardiac disturbances, and neurological effects if medical precautions are not taken. In controlled pharmaceutical settings, phenolic derivatives are widely used in analgesics (aspirin originates from salicylic acid, a phenol derivative), antiseptics, disinfectants, and topical anesthetics. The balance between therapeutic value and toxicity demonstrates the importance of dosage, formulation, and chemical modification in drug development.
Industrial and Commercial Applications — From Plastics to Energy Production
Phenol is a major industrial feedstock. A large percentage of global phenol production goes into manufacturing bisphenol A (BPA), which is used to produce polycarbonate plastics and epoxy resins used in electronics, automotive components, kitchenware, medical devices, adhesives, and coatings. Another substantial portion is used in phenolic resins, flame-retardant materials, insulation foams, rubber chemicals, and synthetic fibers. In petroleum refining, phenolic compounds help control oxidation in lubricants and fuels. In agriculture, phenolic derivatives appear in herbicides and plant-growth regulators. The extensive industrial relevance of phenol illustrates how a simple molecule can become central to modern material science and everyday manufactured goods.
Phenol as a Conceptual Bridge in Organic Chemistry
Phenol is more than a single compound; it is a foundation for an entire class of organic chemistry. Compounds in which one or more hydroxyl groups are bonded directly to an aromatic ring are collectively called phenols, and they appear throughout nature and human-designed chemistry. Natural phenols contribute to flavors, pigments, plant defense molecules, and antioxidants. Synthetic phenols play roles in polymers, pharmaceuticals, dyes, stabilizers, and biotechnology. Learning phenol at the structural level therefore opens the door to understanding vast applications in medicinal chemistry, materials science, biochemistry, and industrial design.
In the end, phenol represents an elegant demonstration of how small changes in molecular structure reshape the entire chemical behavior of a compound. The attachment of a single hydroxyl group to an aromatic ring transforms non-polar benzene into a polar, reactive, weakly acidic, hydrogen-bonding solid that participates in vital biological and industrial processes. The molecule unites structure and function so clearly that it has become a cornerstone of organic chemistry education, illustrating to students how atomic arrangement defines everything — from acidity and reactivity to medicinal action and material innovation. Phenol’s chemical structure and properties therefore remain a powerful example of how molecules shape both science and life.