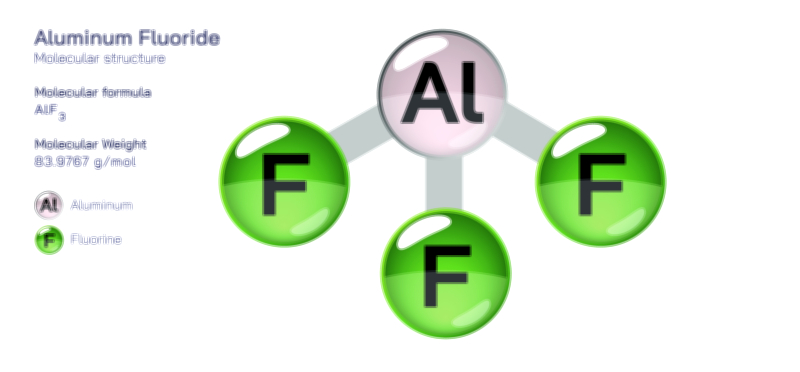
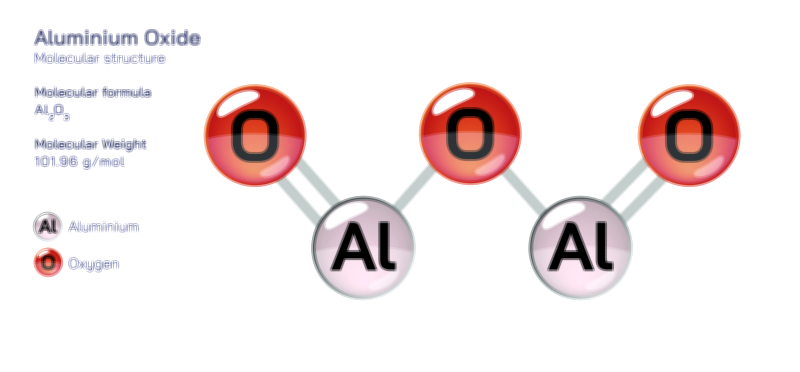
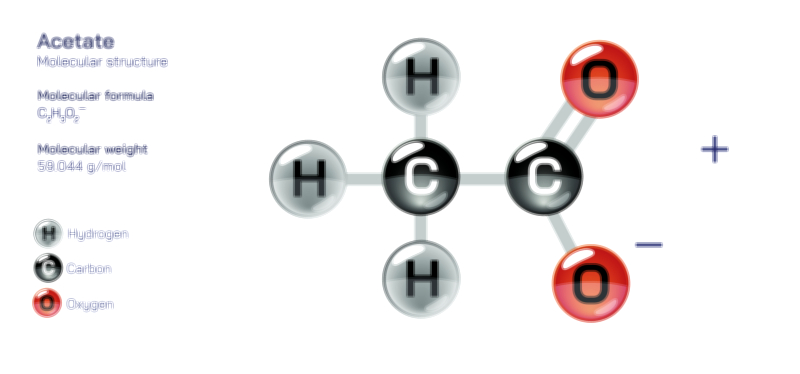
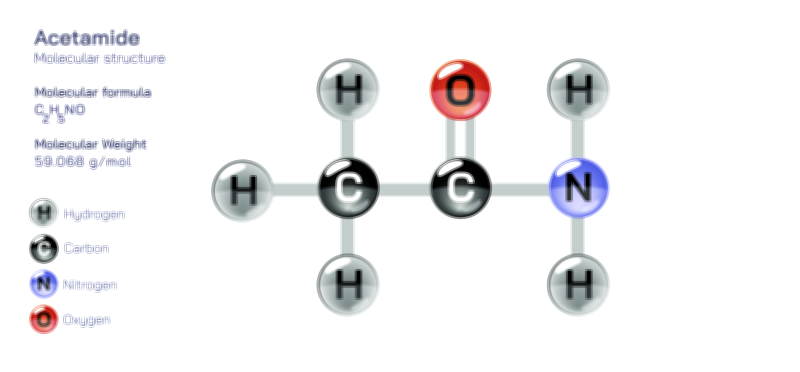
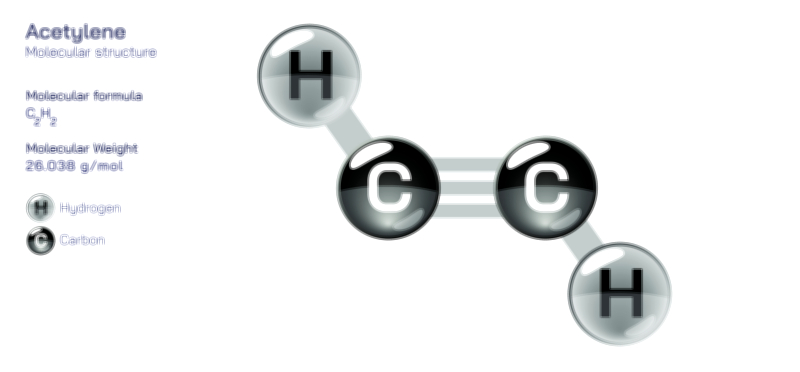
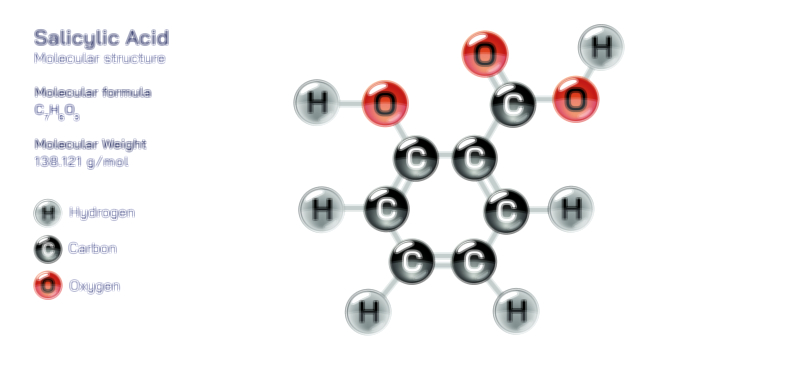
Aluminium Hydroxide — Chemical Structure and Compound Illustration
Fundamental Structure and Identity of Aluminium Hydroxide
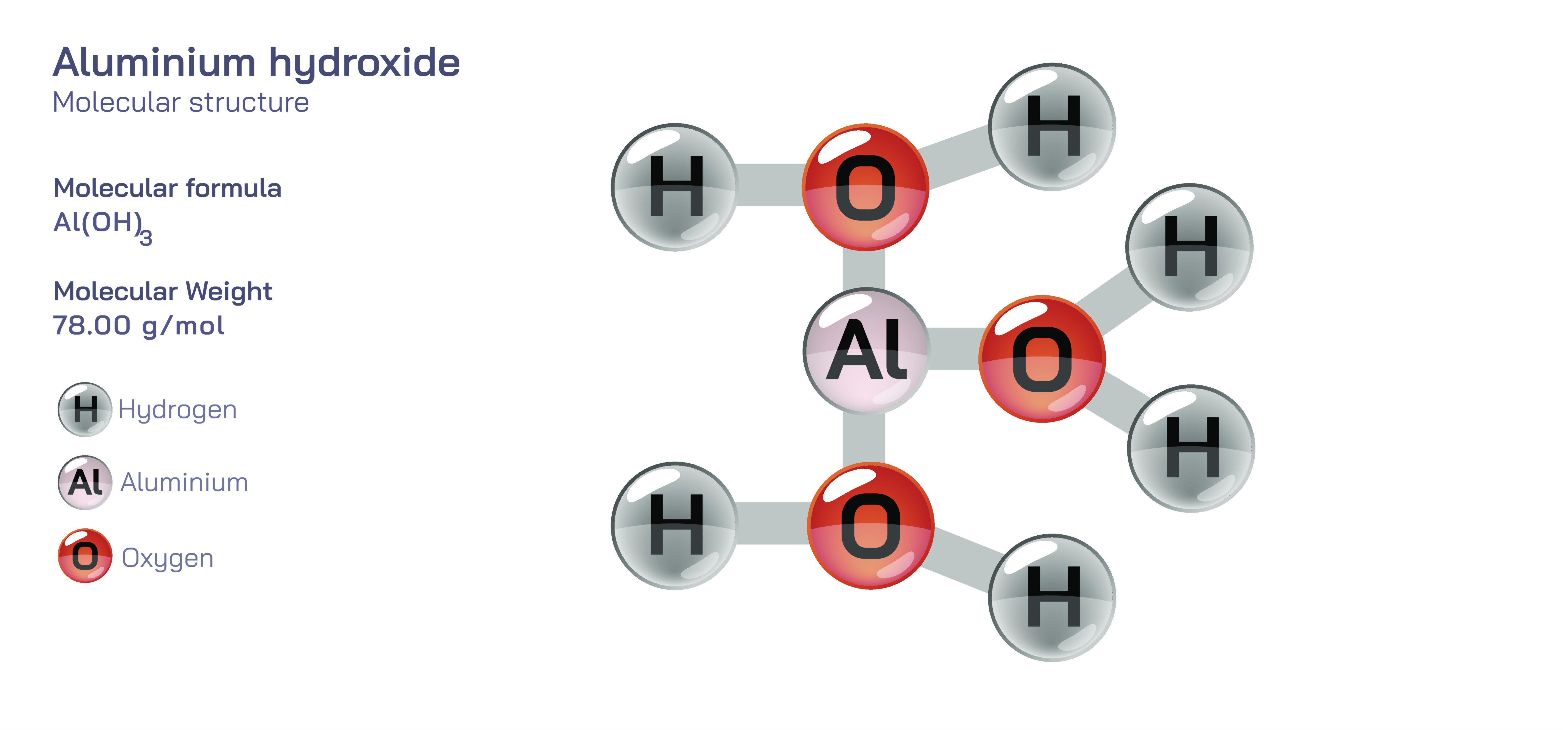
Aluminium hydroxide, often represented by the chemical formula Al(OH)₃, is a vital inorganic compound with wide-ranging significance in chemistry, medicine, industry, and environmental science. It belongs to the class of metal hydroxides and appears naturally as the mineral gibbsite, one of the major components of bauxite ore. Structurally, aluminium hydroxide consists of a central aluminium atom bonded to three hydroxyl groups, forming a network that can exist in various crystalline and hydrated forms. Although its formula appears simple, its structure reflects a delicate balance between ionic and covalent bonding characteristics, shaped by the aluminium ion’s tendency to coordinate with oxygen-based groups. The trivalent nature of aluminium means it exerts strong attractive forces on hydroxide ions, creating a compound that is amphoteric—capable of reacting with both acids and bases. This dual behavior makes aluminium hydroxide a uniquely versatile compound that serves critical functions in antacid medications, water purification, flame retardant formulations, and industrial processes where controlled precipitation or neutralization reactions are needed.
Chemical Structure, Bonding, and Amphoteric Behavior
At the atomic level, aluminium hydroxide’s structure reveals aluminium in a +3 oxidation state, coordinating with hydroxide ions arranged around it in a geometry that resembles an octahedral or layered formation depending on the crystalline variation. In gibbsite, the most common form, each aluminium center is surrounded by six hydroxyl groups through shared hydrogen bonding networks and layered sheet-like arrangements. These layers stack together through additional hydrogen bonds, creating a stable yet flexible solid-state architecture. The Al–OH bond represents a hybrid of ionic and covalent character, with aluminium attracting the electron density of the hydroxyl oxygen strongly enough to impart partial charge separation. This bonding arrangement also accounts for its amphoteric nature: in acidic environments, aluminium hydroxide accepts protons and dissolves to form aluminium salts, while in basic environments, it reacts with hydroxide ions to produce aluminate species such as Al(OH)₄⁻. This remarkable ability to behave as both a base and an acid not only defines its chemical reactivity but also underpins its widespread applications in buffering systems, pharmaceutical formulations, and environmental balancing processes.
Physical Properties and Their Chemical Implications
Aluminium hydroxide typically appears as a white, gelatinous, or powdery solid with a relatively high melting and decomposition point. Its low solubility in water, except under strongly acidic or basic conditions, contributes to its usefulness as a slow-reacting neutralizing agent. The compound’s layered crystalline arrangement creates a structure that can trap water molecules or release them depending on temperature, giving it dehydrating properties that are crucial in industrial uses. When heated, aluminium hydroxide decomposes to form aluminium oxide while releasing water vapor—a behavior that makes it extremely valuable as a flame retardant. By emitting water when exposed to heat, the compound absorbs energy and reduces surface temperatures, slowing the spread of fire. Its physical stability and chemical gentleness explain why it is widely used in pharmaceuticals, especially as an antacid ingredient where its slow dissolution rate allows it to neutralize stomach acid without causing abrupt chemical shifts that could irritate sensitive tissues. Its overall physical and chemical characteristics combine to form a compound that reacts predictably, tolerates environmental variations, and integrates easily into numerous technical processes.
Reactivity and Transformation Pathways
The reactivity of aluminium hydroxide arises from its amphoteric nature and its ability to participate in acid-base, precipitation, and thermal decomposition reactions. When introduced to strong acids, it dissolves to produce soluble aluminium ions, forming salts such as aluminium chloride or aluminium sulfate. In basic solutions, the compound reacts with excess hydroxide to yield aluminate ions, creating complexes that often remain soluble in high-pH environments. Heating aluminium hydroxide causes it to undergo endothermic decomposition, releasing water and converting into aluminium oxide, a transformation that underpins its fire-resistant uses. It also participates in controlled precipitation reactions, especially in water treatment plants where it helps remove suspended impurities through coagulation and flocculation. The precipitated aluminium hydroxide traps fine particles, organic material, and contaminants through adsorption and entanglement, allowing them to settle out of the water. This behavior makes it indispensable for producing clean drinking water and treating wastewater. In biological contexts, its reactivity is intentionally minimized by controlling particle size and solubility so that it can function gently as an antacid or as a vaccine adjuvant, slowly releasing ions that stimulate targeted immune responses without overwhelming the body.
Industrial, Medical, and Environmental Applications
Aluminium hydroxide’s wide applicability stems from its chemical versatility. In medicine, it is one of the most commonly used active ingredients in antacid formulations, neutralizing stomach acidity while forming protective coatings along the gastrointestinal tract. It is also used as an adjuvant in vaccines, where its ability to adsorb antigens and release them gradually increases the body’s immune response. In industrial settings, aluminium hydroxide serves as a precursor for producing alumina, which is essential for manufacturing ceramics, refractories, abrasives, and aluminum metal through the Hall–Héroult process. Its flame-retardant properties make it a core ingredient in plastics, rubber, insulation materials, and textiles where fire resistance is required. Environmentally, it plays a critical role in purification systems, particularly in water treatment where it helps remove pollutants and improve clarity. Because it is non-toxic and biodegradable, it is widely adopted in eco-conscious industrial systems. Across these diverse fields, aluminium hydroxide continues to function as a compound whose stability, reactivity, and adaptability support essential processes with reliability and safety.
Illustrating the Structure and Functional Behavior of Aluminium Hydroxide
Illustrations depicting aluminium hydroxide typically highlight the central aluminium atom bonded to hydroxide groups arranged in layered or octahedral environments. These visual representations often show how hydrogen bonding stabilizes the layers and how the structure responds to acidic or basic surroundings. Illustrations may also depict its decomposition pathway, showing the release of water and the formation of aluminium oxide under heat. In medical or industrial diagrams, aluminium hydroxide may be shown interacting with acids or participating in adsorption processes for purification. Such representations help clarify how the molecular structure governs its amphoteric reactivity, solid-state behavior, and functional applications. By visualizing its bonding patterns and transformations, one gains a deeper appreciation of how this compound embodies the balance between chemical simplicity and practical complexity across multiple scientific and technological domains.