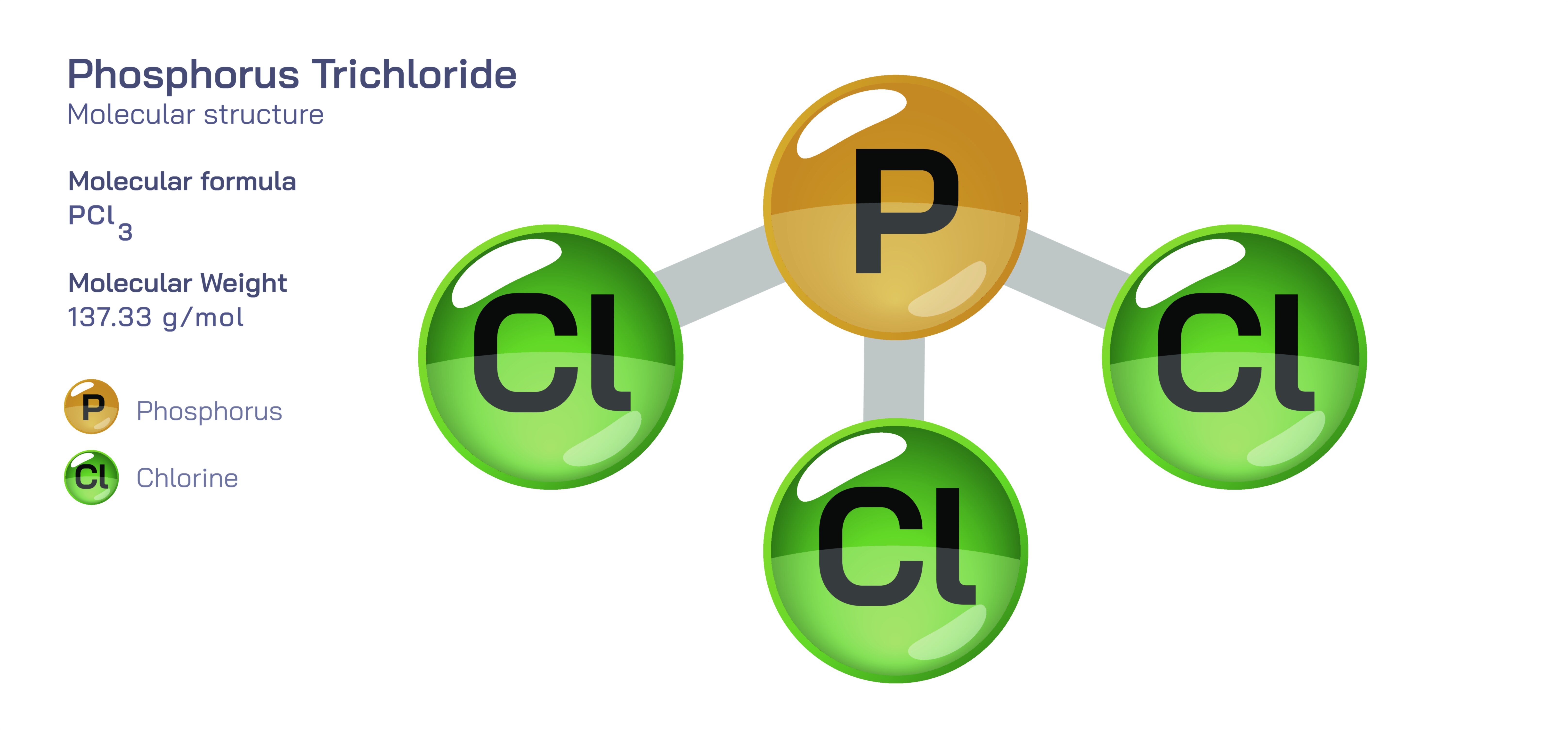
Phosphorus Trichloride – Structure, Formula, Bonding Characteristics, Chemical Behavior, Industrial Uses, and Its Central Role as a Foundation Chemical in Manufacturing and Synthesis
Phosphorus trichloride is one of the most strategically important inorganic compounds in the global chemical industry because its structure allows it to act as a reactive precursor for countless phosphorus-containing materials, fertilizers, pharmaceuticals, plastics, pesticides, flame retardants, additives, and specialty chemicals. The molecule’s formula is PCl₃, meaning it contains one phosphorus atom covalently bonded to three chlorine atoms. At the structural level, phosphorus trichloride has a trigonal pyramidal geometry: three chlorine atoms occupy three corners of a triangular base, while phosphorus sits at the apex with a lone electron pair that pushes the P–Cl bonds downward. This lone pair contributes strongly to the molecule’s reactivity by making the phosphorus electrophilic and susceptible to nucleophilic attack, while the chlorine atoms act as leaving groups during substitution or hydrolysis reactions. The result is a molecule that behaves as a controlled chlorinating agent, a ligand for metal coordination, and a gateway to derivative compounds such as phosphites, phosphonates, and phosphates. Even though the structure appears simple, the electronic properties arising from P–Cl polarity and phosphorus’s empty d-orbitals give phosphorus trichloride an immense chemical influence — a fact that explains why it is considered a “building block chemical” in industrial production.
In pure form, phosphorus trichloride is a colorless to pale yellow fuming liquid with a sharp, penetrating odor reminiscent of hydrochloric acid and chlorine. It reacts vigorously with water, and even atmospheric moisture, releasing hydrogen chloride gas and forming a mixture of phosphorous acid and other oxychlorinated species. This hydrolysis behavior demonstrates the instability of P–Cl bonds in aqueous environments and explains why phosphorus trichloride must always be handled in dry, moisture-free conditions. Its high reactivity arises from the electron-poor character of the phosphorus atom, which can expand its coordination number and form additional bonds — a capability that transforms PCl₃ into a versatile reagent in synthesis. In non-aqueous systems, phosphorus trichloride reacts with alcohols, phenols, carboxylic acids, amines, and polyhydroxy compounds to replace hydroxyl groups with chlorine or generate phosphorus-containing esters and amides. The molecule also acts as a Lewis base through its lone pair and as a Lewis acid through its vacant orbitals, contributing to its reactivity in catalysis, chlorination chemistry, and organophosphorus compound formation.
The most dominant industrial role of phosphorus trichloride is as a precursor for phosphorus-based pesticides and herbicides. A very large percentage of the world’s production of PCl₃ is consumed in the manufacturing of organophosphorus agrochemicals — including glyphosate, phosphonate fungicides, and related crop-protection agents. In these applications, the P–Cl bonds act as reactive sites enabling substitution by organic groups, giving rise to phosphonate and phosphinate structures that function biologically in plant disease suppression, weed control, or pest regulation. Without phosphorus trichloride, a majority of modern agricultural pesticides would not exist, and global crop yields would be far lower. However, the industrial value is accompanied by responsibility, since agrochemical runoff can impact ecosystems. The connection between phosphorus trichloride and fertilizer-adjacent chemical industries illustrates how this single compound sits at the foundation of agricultural technology.
Beyond agriculture, phosphorus trichloride plays a defining role in the development of flame retardant chemicals and plastic additives. Under controlled substitution reactions, PCl₃ gives rise to phosphites and phosphonates — compounds incorporated into polymer systems to provide oxidative stability, thermal endurance, and fire resistance. In flame retardants, phosphorus derivatives promote char formation and inhibit flammable gas release when heated, making them crucial components in electronics, building materials, upholstery, automotive interiors, and aerospace composites. The central idea is that phosphorus trichloride is not used directly in consumer products; rather, it enables the creation of compounds that control ignition, slow burning, or reduce smoke generation. Its structural ability to form phosphorus–carbon and phosphorus–oxygen bonds makes it ideal for customizing polymer performance without compromising material strength.
In the pharmaceutical and medical chemistry sector, phosphorus trichloride serves as a reagent and intermediate in the synthesis of organophosphate-based medicines, antiviral drugs, and nerve pain treatments. The introduction of phosphorus into biologically active molecules often requires selective substitution chemistry, which PCl₃ facilitates by enabling the formation of phosphoramidates, phosphonates, and related structures. It also plays a role in creating intermediates used for vitamin synthesis, antibiotic production, and formation of specialty reagents that target cell signaling or enzymatic processes. Because biological pathways depend heavily on phosphorylation reactions, the ability to build P–O and P–N bonds from phosphorus trichloride supports the development of molecules that interact with enzymes, membranes, and metabolic systems. This impact occurs indirectly: PCl₃ creates key intermediates that medicinal chemists transform into drugs with precise therapeutic function.
In manufacturing of plastic stabilizers, lubricants, and antioxidant additives, phosphorus trichloride is indispensable. When converted into phosphite esters, PCl₃ derivatives protect polymers such as polyethylene, polypropylene, PVC, and engineering plastics from heat-induced degradation during extrusion, molding, and long-term use. Phosphite stabilizers capture free radicals and suppress oxidation, preventing brittleness and discoloration of plastics. In lubricants and hydraulic fluids, phosphorus-containing additives improve anti-wear properties, reducing metal-to-metal friction in engines and machinery. These applications show how phosphorus trichloride contributes quietly to durability, energy efficiency, and sustainability in industrial equipment, even though it never appears directly in the final product.
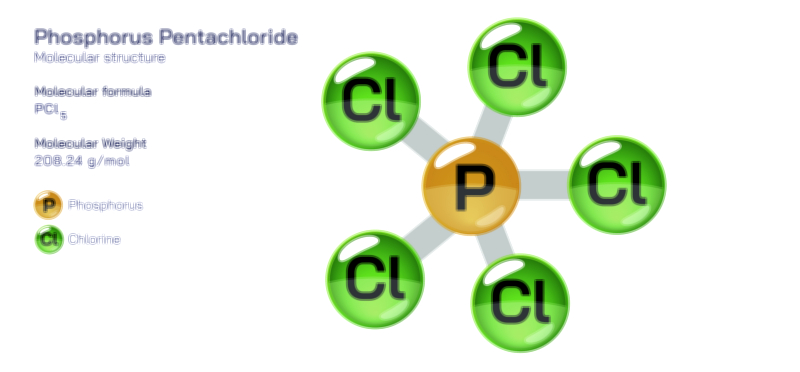
Another major application of phosphorus trichloride is its role in chemical synthesis and laboratory research. PCl₃ acts as a reagent in chlorination reactions, transformation of alcohols into alkyl chlorides, formation of acid chlorides from carboxylic acids (via the intermediate POCl₃), and production of numerous organophosphorus compounds. It is also used in the preparation of phosphorus pentachloride (PCl₅) and phosphorus oxychloride (POCl₃), two reagents widely employed in peptide synthesis, dye manufacturing, pharmaceutical development, semiconductor processing, and flame-retardant chemistry. In analytical science, phosphorus trichloride is used to produce reagents that bind trace metals, enabling detection and quantification in environmental or biomedical samples. Its utility in synthesis comes from reliability: when moisture is excluded and reaction partners are chosen correctly, PCl₃ reacts in predictable and selective pathways that offer chemists precise control over molecular transformation.
From a safety and environmental perspective, phosphorus trichloride requires careful handling due to its corrosive nature and sensitivity to moisture. Contact with water releases hydrogen chloride gas, which can cause respiratory irritation and severe tissue damage. PCl₃ should therefore be stored in airtight containers made of corrosion-resistant materials and handled only with appropriate ventilation, respiratory protection, and moisture control. In industrial environments, closed-system manufacturing minimizes exposure risks and prevents unintentional release into the environment. Although phosphorus trichloride itself is rarely a direct pollutant because it reacts quickly with moisture, the downstream products it helps create — pesticides, plastic additives, and flame retardants — must be managed responsibly to prevent cumulative environmental and health impacts. Regulatory frameworks in agriculture, manufacturing, and chemical safety play a vital role in ensuring that the benefits of PCl₃-based products are delivered without unnecessary harm.
The long-term significance of phosphorus trichloride stems from its special position as a gateway molecule: a structural platform used to turn raw phosphorus into a vast family of industrial and biologically active compounds. It sits at the crossroads of agricultural chemistry, polymer science, pharmaceuticals, surface engineering, lubricant technology, and laboratory synthesis. The molecule’s pyramidal structure, electron-deficient phosphorus center, and highly reactive P–Cl bonds give it a unique capability to transform into an enormous spectrum of phosphorus-based materials. If phosphoric acid symbolizes the nutrient side of phosphorus chemistry, then phosphorus trichloride symbolizes the synthetic and industrial side — the branch of chemistry that converts molecular potential into practical technologies.
In essence, phosphorus trichloride demonstrates that a molecule does not need to be complex to shape entire industries. A single phosphorus atom surrounded by three chlorine atoms becomes the cornerstone of agricultural productivity, polymer durability, fire safety, medication synthesis, lubrication efficiency, and laboratory innovation. Understanding its structure, formula, and industrial uses offers a window into how chemistry supports modern civilization — by turning simple molecular frameworks into technologies that feed, build, protect, and power the world.