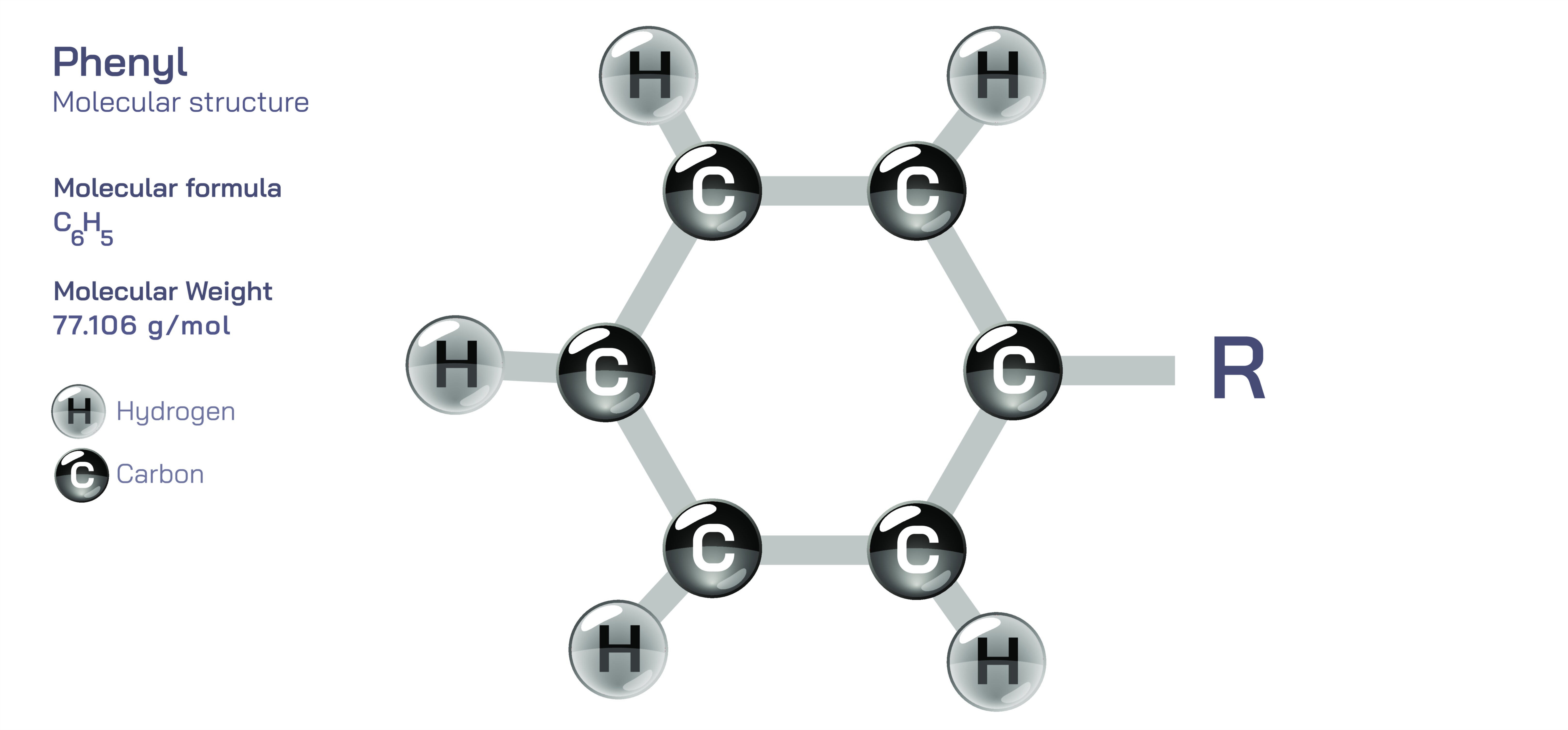
Phenyl Group – Structure, Bonding Behavior, Resonance Effects, Substitution Patterns, Reactivity Characteristics, and Its Wide-Ranging Uses in Organic Chemistry and Industrial Applications
The phenyl group is one of the most fundamental building blocks in organic chemistry, and its presence dramatically influences the behavior of molecules in which it appears. Represented structurally as –C₆H₅, the phenyl group consists of a benzene ring that has lost one hydrogen atom, allowing it to attach to another atom or molecular fragment through a single covalent bond. Because benzene itself is stabilized by aromaticity—a special electronic arrangement in which π-electrons are delocalized across six carbon atoms—the phenyl group carries this stability into any compound it becomes part of. In visual structural form, the phenyl group appears as a six-membered ring with alternating double and single bonds, but in reality, the π-electrons are evenly distributed through resonance, giving all carbon–carbon bonds the same length. This delocalized electron cloud defines the chemical personality of the phenyl group, determining how it interacts with electrophiles, nucleophiles, radicals, and substituent groups. When a phenyl group is attached to a molecule, its aromatic system influences reactivity, acidity, polarity, and even physical properties such as melting point, solubility, volatility, and odor. The phenyl fragment is therefore not simply a decorative structural element—it is an active functional group with significant impact on how molecules behave in chemical, biological, and industrial environments.
At the molecular level, attaching a phenyl group to another organic structure generally increases rigidity and decreases molecular flexibility because the aromatic ring is planar and highly structured. This rigid backbone restricts rotational freedom, making phenyl-containing compounds mechanically stronger and less conformationally variable than aliphatic chains of similar size. The phenyl ring is also hydrophobic and non-polar in nature, which decreases solubility of molecules in water but increases solubility in organic or lipid environments. This fundamental property explains why many phenyl-containing drugs readily pass through cell membranes composed of lipid bilayers. Another structural consequence of phenyl attachment is the effect on electron distribution. Through resonance and inductive effects, the phenyl group can donate or withdraw electron density depending on the attached substituents. For example, when electron-donating groups such as alkyls or hydroxyls are attached to phenyl, the ring becomes more reactive toward electrophilic substitution. In contrast, electron-withdrawing groups such as nitro or carbonyl moieties reduce electron density, making the ring less reactive. This delicate balance between resonance donation and inductive withdrawal makes phenyl groups central to controlling organic reactivity.
One of the most recognizable roles of the phenyl group appears in the formation of substituted aromatic compounds. Because of the delocalization of electrons, the phenyl ring directs electrophilic aromatic substitution toward the ortho and para positions when bonded to activating groups, and toward the meta position when bonded to strongly deactivating groups. These predictable orientation patterns are essential in designing dyes, fragrances, pharmaceutical molecules, antibiotics, and polymer precursors. When chemists need to synthesize a compound with a specific substitution pattern on an aromatic ring, the phenyl group acts as both a platform and an influence mechanism. The aromatic ring not only determines reaction pathways but also governs how additional groups affect electron flow, reactivity, and steric arrangement. The phenyl group therefore acts as a control element that channels chemical reactions toward targeted molecular architectures.
Across organic chemistry, the phenyl group plays multiple roles beyond structural reinforcement and reactivity modulation. Because aromatic rings absorb ultraviolet light in predictable ways, phenyl groups are incorporated into compounds used as UV stabilizers, sunscreens, optical brighteners, and fluorescent dyes. In medicinal chemistry, the phenyl fragment contributes to molecular binding in active sites of enzymes and receptors due to π–stacking, hydrophobic interactions, and van der Waals attraction between aromatic rings. These subtle interactions are not physical bonds but rather orientation-dependent attractions that help drugs fit into biological targets with high precision. As a result, countless pharmaceutical molecules—including analgesics, antihistamines, antidepressants, anti-inflammatories, and anesthetics—contain phenyl groups that increase biological activity, solubility in lipid membranes, metabolic stability, and receptor affinity. In polymer science, phenyl-containing monomers such as styrene form long-chain plastics like polystyrene, ABS, and epoxy resins. The phenyl rings embedded in polymer backbones provide thermal stability, tensile strength, hardness, and resistance to ultraviolet degradation, making these materials standard in packaging, electronics, automotive parts, construction materials, and household goods.
Industrial chemistry depends on phenyl-based compounds for large-scale manufacturing. Phenyl is present in synthetic aroma molecules used in perfumes and flavors, where the ring stabilizes volatile molecules without sacrificing sensory intensity. Phenyl derivatives act as intermediates in agricultural chemicals, petrochemical additives, antioxidants, dyes, pigments, and rubber processing agents. Even high-performance materials such as Kevlar and polycarbonate plastics trace their strength to phenyl-containing molecular frameworks. In analytical chemistry, phenyl groups appear in specialized solvents, chromatography stationary phases, and chemical probes that separate or detect other organic molecules through their affinity for aromatic systems. These applications illustrate how the phenyl fragment supports industries ranging from medicine and transportation to packaging, agriculture, and environmental monitoring.
Biologically, phenyl-containing structures also appear naturally in amino acids, hormones, and plant metabolites. A well-known example is phenylalanine, an essential amino acid that incorporates a phenyl group and serves as a precursor to neurotransmitters such as dopamine, epinephrine, and norepinephrine. In plant systems, phenolic compounds—formed from phenyl derivatives—assist in defense mechanisms, pigmentation, antioxidant functions, and aroma production. Although synthetic phenyl-based chemicals can sometimes pose toxicity risks when misused, the phenyl ring itself is a normal structural component in the biochemistry of life. The dual identity of phenyl—both naturally occurring and industrially produced—shows how deeply intertwined aromatic chemistry is with metabolism, ecology, and technology.
Beyond its direct applications, the phenyl group plays a huge conceptual role in teaching and understanding organic chemistry. It introduces students to aromaticity, resonance, substitution patterns, directing effects, π-electron systems, and structure–property relationships. When chemists observe significant shifts in melting point, solubility, stability, or biological activity following phenyl substitution, they learn how powerful molecular fragments can be. The phenyl group becomes a tool for rational design, enabling chemists to fine-tune molecules with predictable outcomes. In this sense, the phenyl fragment symbolizes the intellectual strategy behind organic synthesis: rather than randomly assembling carbon chains, chemists select structural modules that produce desirable performance in a final product.
Ultimately, the phenyl group is not just one of many functional groups—it is one of the most transformative motifs in organic chemistry. It shapes the geometry, reactivity, biological behavior, industrial usefulness, and sensory properties of the molecules that contain it. Whether incorporated into drugs, polymers, dyes, enzymes, solvents, or fragrances, the phenyl group functions as a molecular architect, imposing aromatic stability while guiding electron movement and intermolecular interactions. The moment a phenyl ring is introduced into a chemical structure, the molecule gains not only stability but new physical and functional capabilities. This is why the phenyl group remains one of the most iconic and indispensable structural features in modern organic chemistry, linking the microscopic beauty of aromatic electron resonance to the macroscopic world of materials, medicines, and everyday products.