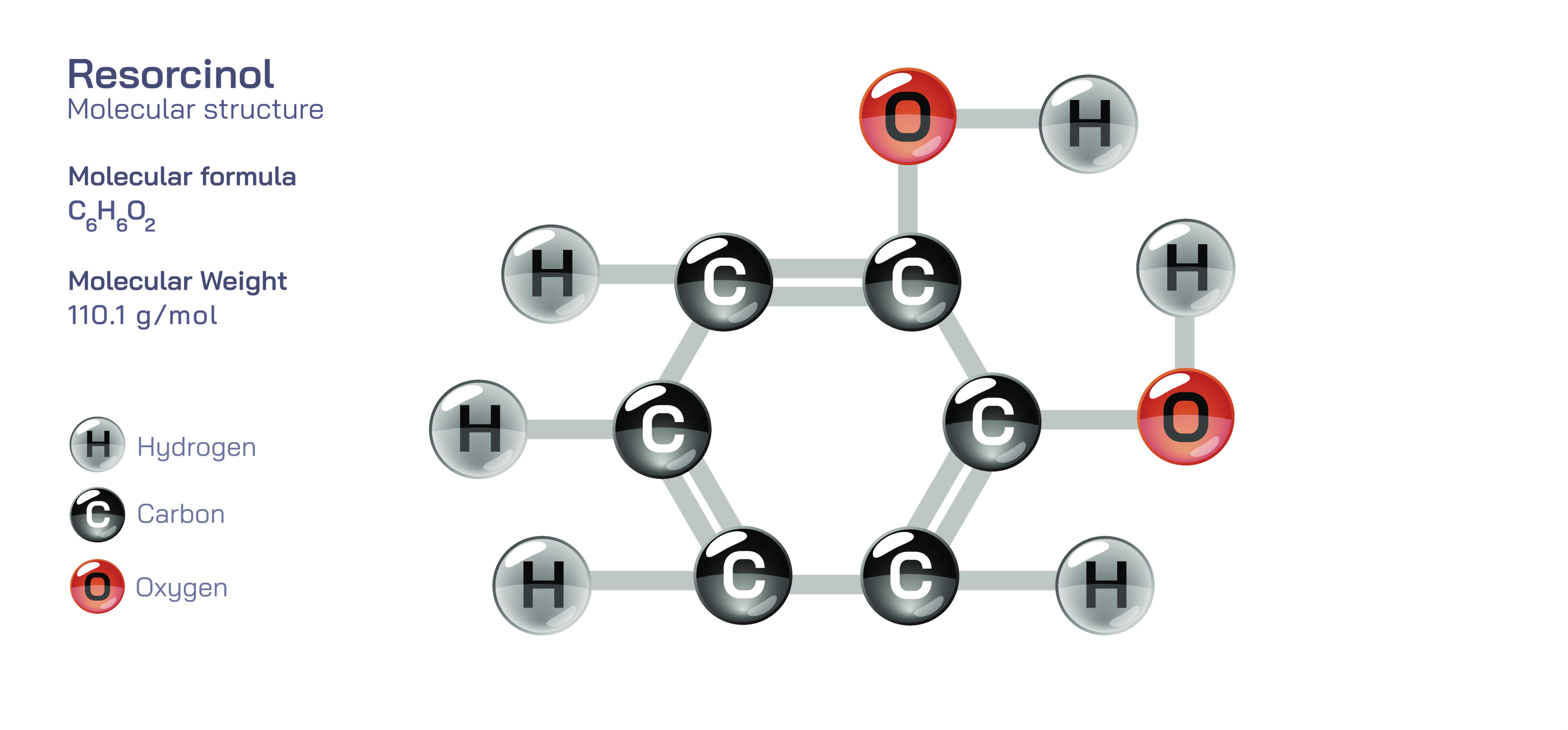
Resorcinol – Structure, Formula, Molecular Features, Reactivity, and Industrial, Medical, and Material Science Applications of This Versatile Aromatic Compound
Resorcinol is an important organic compound whose molecular architecture and reactivity have made it valuable in pharmaceutical formulations, polymer chemistry, adhesive manufacturing, resin technology, disinfectants, and specialty chemical synthesis. Its chemical formula is C₆H₄(OH)₂, and it belongs to the family of dihydroxybenzenes, meaning it contains a benzene ring substituted with two hydroxyl (–OH) groups. What gives resorcinol its distinct character is the meta-orientation of these two hydroxyl groups (specifically at positions 1 and 3 on the benzene ring), which differentiates it from other isomers such as catechol (1,2-dihydroxybenzene) and hydroquinone (1,4-dihydroxybenzene). This structural positioning affects electron distribution across the ring, influences hydrogen bonding behavior, and shapes the compound’s enzymatic interaction and reactivity. Resorcinol appears as white to pale pink crystalline solid under normal conditions and dissolves readily in water, alcohol, and ether due to the strong hydrogen bonding capability of the hydroxyl groups. The combination of aromatic stability and dual hydroxyl reactivity explains why resorcinol plays a central role in a wide range of chemical and technological fields.
On a molecular level, resorcinol displays both phenolic acidity and nucleophilic aromatic behavior, anchored in its two hydroxyl substituents. Each hydroxyl group donates electron density into the aromatic ring but also participates in hydrogen bonding—internally within the crystal lattice and externally with solvents. This ability to engage in both intra- and intermolecular hydrogen bonding accounts for its moderate melting point, high solubility, and characteristic solid-state stability. Because the hydroxyl groups activate the ring at the 2-, 4-, and 6- positions, resorcinol undergoes electrophilic substitution more readily than benzene, enabling the synthesis of dyes, pharmaceuticals, UV absorbers, and protective coatings. Unlike other dihydroxybenzenes, resorcinol tends to form stable condensation products with aldehydes, especially formaldehyde, leading to adhesive and resin systems with high mechanical strength and thermal durability. These reactivity trends originate directly from its meta-substitution pattern and electron distribution, showing how small structural differences drive large variations in chemical behavior.
In everyday industry, resorcinol is best known for its influence on rubber and tire manufacturing, where it is used to strengthen adhesion between rubber compounds and reinforcing fabrics such as nylon, polyester, rayon, or steel cord. It achieves this through resorcinol–formaldehyde resins, which create molecular bridges between polymer chains and textile surfaces, dramatically improving durability and fatigue resistance under repeated flexing stresses. Without this chemistry, modern vehicle tires, conveyor belts, and high-performance rubber components would fail prematurely. Resin systems incorporating resorcinol also appear in wood adhesives, engineered timber bonding, and industrial glues that require water resistance and long-term stability under thermal cycling. In coatings and plastics, resorcinol-based additives act as stabilizers and UV absorbers because their aromatic hydroxyl structure can neutralize reactive species created by ultraviolet radiation, preventing the degradation, yellowing, or brittleness of polymers exposed to sunlight. These applications illustrate how resorcinol’s structure supports resilience against both mechanical and environmental stress.
Resorcinol’s role extends strongly into pharmaceutical and medical applications, where it appears in topical formulations due to its antiseptic, keratolytic, and exfoliating properties. It is used in acne creams, antifungal solutions, wart-removal preparations, and skin descaling treatments for hyperkeratosis because it can break down surface proteins while suppressing microbial activity. In low concentrations, resorcinol assists skin regeneration by accelerating the removal of dead cells; in higher concentrations, it induces deeper peeling for therapeutic dermatological procedures. The compound’s disinfectant behavior arises from its ability to interfere with cellular enzymes and membrane structures in microbes. Its phenolic backbone makes it structurally similar to other topical antiseptics, though it must be used with controlled dosage due to possible irritation or systemic absorption. Regulatory frameworks guide its use, ensuring benefits are provided safely and without toxicity. The pharmaceutical value of resorcinol demonstrates that molecular functionality—especially hydroxyl positioning and acidity—translates directly to biological impact.
In dye and colorant manufacturing, resorcinol serves as a precursor for azo dyes, triarylmethane dyes, and fluorescent pigments. Its activated aromatic ring enables coupling reactions that form vivid and stable chromophore structures. This dye chemistry supports textiles, hair-care formulations, printing inks, leather finishing, and artistic pigment production. In hair-dye formulations specifically, resorcinol acts as a color-developing intermediate that participates in oxidative coupling to generate durable shades. Its involvement in permanent hair dye systems is carefully regulated in cosmetic chemistry due to its biological activity, demonstrating once again how structural reactivity must be balanced with safety considerations.
Beyond coloring agents, resorcinol is central in the synthesis of benzoxazines—a class of advanced thermoset materials valued for high thermal stability, low shrinkage, flame resistance, and dimensional precision. These polymers are now widely used in aerospace composites, printed circuit boards, adhesives for electronic assemblies, and high-performance coatings. The formation of benzoxazine monomers heavily depends on resorcinol’s dual hydroxyl arrangement, enabling ring-closing reactions not easily achievable with other phenolic compounds. In the field of photoresists and UV-curable coatings, resorcinol derivatives help control crosslinking reactions in response to ultraviolet radiation, supporting microelectronics fabrication and precision lithography.
Environmental and biological chemistry has also provided unique roles for resorcinol and its derivatives. Some marine organisms naturally produce resorcinol-based compounds as biochemical defense agents, highlighting the ecological relevance of meta-dihydroxy aromatic structures. In laboratory environments, resorcinol acts as a reagent for analytical tests, including assays that quantify sugars by producing characteristic color changes upon reaction with specific carbohydrate structures. In agriculture, certain resorcinol derivatives contribute to herbicides, plant-growth regulators, and treatments that influence hormonal responses in crops. These diverse applications reinforce that resorcinol’s value stems not from a single function but from a wide versatility driven by its intrinsic molecular behavior.
Despite its usefulness, resorcinol requires careful handling, storage, and dosing to avoid irritation or toxicity. In industrial settings, protective equipment and ventilation are standard to reduce inhalation or skin exposure. Waste-processing technologies ensure safe disposal to prevent ecological damage. At the consumer level, dermatological and cosmetic formulations adhere strictly to regulated concentration limits to deliver therapeutic benefits without adverse effects. The ongoing development of greener synthesis methods—reducing harsh reagents and waste byproducts—reflects the balance modern industry seeks between chemical utility and environmental responsibility.
In summary, resorcinol occupies a powerful position in both organic chemistry and industrial technology because its structural framework delivers an ideal blend of aromatic stability, electron activation, and hydrogen-bonding capacity. Its formula C₆H₄(OH)₂ may appear simple, but the meta-orientation of hydroxyl groups provides unique chemical behavior unlike its isomeric counterparts. This structure allows it to form high-strength adhesives, durable polymers, UV-stable coatings, vivid dyes, dermatological medications, and multi-functional specialty chemicals. Resorcinol demonstrates that the placement of atoms within a molecule defines not only theoretical reactivity but also the practical possibilities of technology, medicine, and materials science. Understanding its structure, formula, and applications offers insight into how organic chemistry transforms small aromatic frameworks into solutions that shape manufacturing, health, design, and innovation across the modern world.