Phosphorous Acid – Structure, Formula, Properties, Functional Behavior, and Applications Across Agriculture, Industry, and Chemical Synthesis
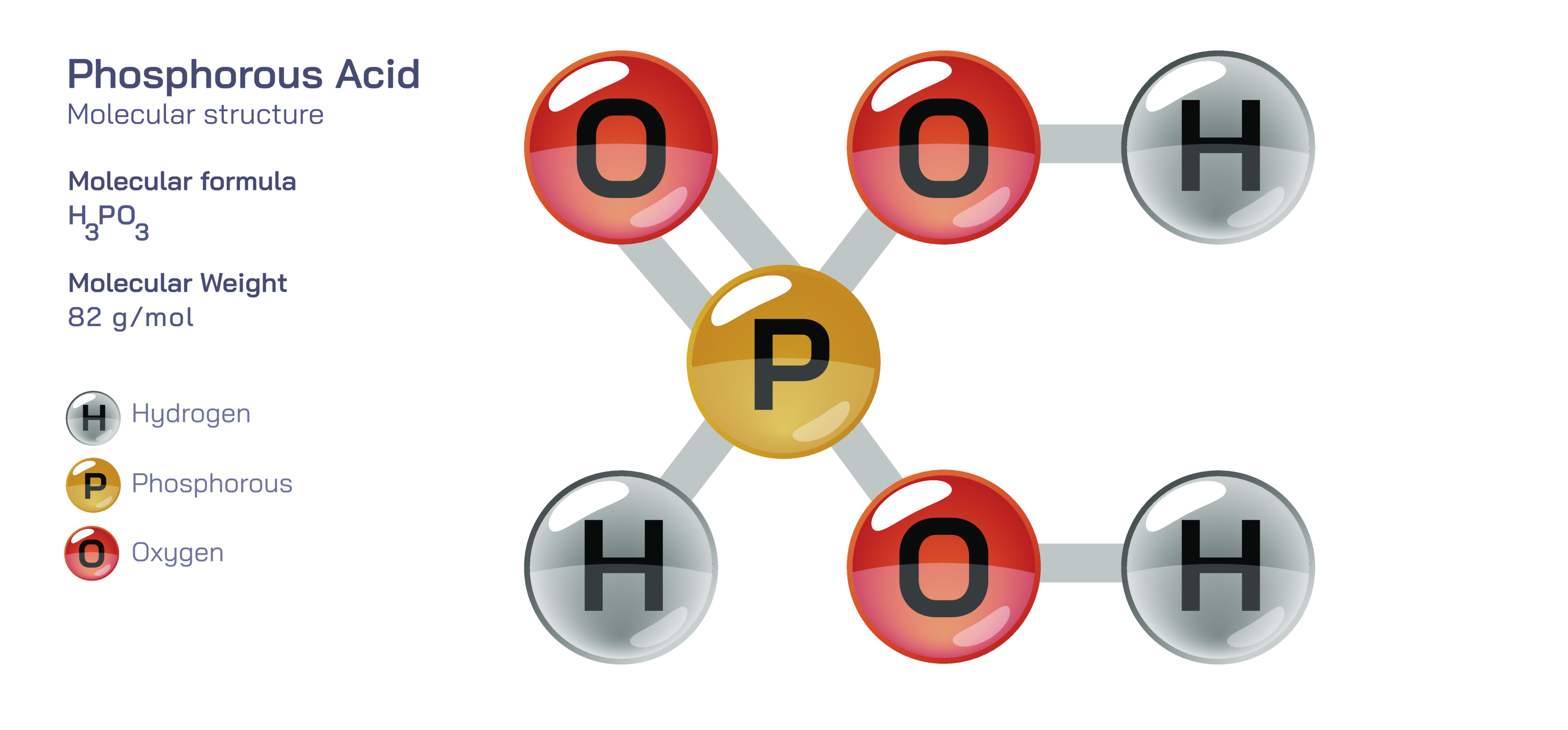
Phosphorous acid is an important inorganic oxyacid of phosphorus whose behavior, structure, and applications distinguish it significantly from the more commonly encountered phosphoric acid. Its chemical formula is H₃PO₃, yet this representation can be slightly misleading because the molecular structure does not contain three acidic hydrogen atoms capable of dissociation. At the structural level, phosphorous acid consists of a central phosphorus atom in a pyramidal geometry bonded to three oxygen atoms and one hydrogen atom, but the key feature lies in the exact bonding arrangement. Two of the hydrogen atoms are directly bonded to oxygen atoms in hydroxyl groups (P–OH), while the third hydrogen is bonded directly to the phosphorus atom (P–H). This means that only the two hydrogens attached to oxygen are ionizable, making phosphorous acid a diprotic acid rather than triprotic, despite the “H₃” formula. The presence of the P–H bond is the most defining characteristic of the molecule and places phosphorous acid in the category of phosphonic acids, which behave differently from the phosphoric acid family. In aqueous solutions, these hydroxyl-linked hydrogens dissociate stepwise to produce hydrogen phosphite (H₂PO₃⁻) and phosphite (HPO₃²⁻) ions, while the P–H hydrogen remains covalently bonded and does not ionize. This bonding pattern reveals how the structure directly determines both acidity and reactivity, emphasizing that the formula alone cannot capture the molecule’s precise chemistry; the spatial arrangement of atoms explains why phosphorous acid behaves as it does in biological, agricultural, and synthetic environments.
5. This capacity for oxidation makes phosphorous acid important in industrial processes that require controlled reduction, where it can convert metal ions or reactive compounds into more stable forms. The interplay between acidity and reducing behavior gives phosphorous acid a dual personality: it can behave as an acid in proton transfer reactions and as a reducing agent in electron transfer reactions. These two modes of reactivity expand its versatility in laboratory synthesis, corrosion control, metallurgical treatments, polymer manufacture, and agricultural formulations.
Although phosphorous acid is a relatively small inorganic molecule, it plays a surprisingly large role in agriculture, not because it delivers phosphorus as a plant nutrient in the same way as phosphate fertilizers do, but because phosphite ions formed from the dissociation of phosphorous acid have unique biological effects on plants. Phosphites are readily absorbed and translocated throughout plant tissue, where they display antifungal and antimicrobial properties by inhibiting pathogenic oomycetes such as Phytophthora, Pythium, and Downy mildew. This discovery transformed phosphorous acid derivatives into a new category of agricultural inputs known as phosphonate fungicides. Unlike conventional phosphate-based fertilizers, phosphorous acid does not directly provide the form of phosphorus used in metabolic energy transfer or structural development. Instead, its value lies in suppressing fungal infections, stimulating plant defense responses, and supporting healthier root systems. For specialty crops such as citrus, grapes, potatoes, and ornamentals, phosphite-based products have become essential tools in disease management. These formulations are often applied as foliar sprays, soil drenches, or trunk injections, demonstrating how a simple inorganic acid can contribute to sustainable crop protection with lower environmental toxicity than many traditional pesticides.
Phosphorous acid also plays a role in industrial chemistry and materials science, where its reducing properties allow it to manipulate the oxidation states of certain compounds during synthesis. It participates in the manufacturing of plastics and polymers, particularly as an intermediate or catalyst in producing phosphonates, compounds used as plastic stabilizers and flame retardants. In water treatment processes, phosphorous acid and its derivatives help prevent scale formation by sequestering metal ions that would otherwise precipitate into mineral deposits. In metal surface processing, phosphorous acid participates in controlled reduction reactions that prevent corrosion and improve surface preparation. It helps convert metal oxides into more stable forms, enhancing coating adhesion in electroplating and industrial painting. These applications rely on the subtle chemical feature of the P–H bond, which enables carefully controlled redox interactions that do not result in excessive corrosion or material degradation. The versatility of phosphorous acid in dealing with metals and industrial surfaces emerges directly from its structural properties, turning a molecular detail into practical utility.
Laboratory and synthetic chemistry represent another domain where phosphorous acid plays a crucial role. It is used to produce phosphite salts and esters, which act as reducing agents, stabilizers, and intermediates in fine chemical synthesis. In organic chemistry, phosphorous acid contributes to the Michaelis–Arbuzov reaction, which produces phosphonate esters—key compounds in agrochemicals, pharmaceuticals, and polymer additives. These reactions demonstrate the importance of phosphorous acid not simply as an acid but as a multifunctional reagent capable of participating in proton transfer, nucleophilic attack, and redox transformations. In analytical chemistry, phosphorous acid can be used to prevent oxidation of certain sensitive solutions or to maintain reducing environments during experiments. Chemical researchers value it as a reagent that offers reliable reduction without the unpredictability or toxicity associated with stronger reducing agents.
In the pharmaceutical world, phosphorous acid derivatives show increasing value. Phosphonates synthesized from phosphorous acid have become active ingredients in certain antiviral and antibiotic medications. Their structural similarity to phosphate groups allows them to mimic biological molecules and interfere with metabolic pathways in targeted microorganisms. Because phosphite- and phosphonate-based compounds are metabolized differently from phosphate-based biomolecules, they can selectively disrupt disease-causing cells without harming human biochemistry at therapeutic doses. This illustrates how the structure of phosphorous acid contributes indirectly to medical innovation by enabling the creation of biologically active compounds that interact with life’s biochemical scaffolding.
While phosphorous acid has important uses, it also requires responsible handling. In concentrated form, it is corrosive to skin, eyes, and mucous membranes and should be managed with proper safety precautions. In agricultural contexts, overuse can lead to soil imbalance if misapplied, because phosphite does not substitute for phosphate nutrition and cannot correct phosphorus deficiencies in plants. However, when used in appropriate disease-control programs alongside balanced fertilization, phosphite-based products reduce environmental pressure compared with more toxic fungicides. In industrial discharge, phosphite must be monitored to prevent unwanted environmental accumulation, though it is generally less damaging to waterways than excessive phosphate runoff. These considerations highlight the dual nature of chemical technology — every useful molecule must be applied with knowledge and regulation to deliver benefits without ecological cost.
Phosphorous acid stands as a clear demonstration that chemistry is not defined only by formulas but by the deeper truth that molecular structure determines function. A single P–H bond distinguishes phosphorous acid from phosphoric acid and creates an entirely different set of properties — converting a nutrient-oriented acid into a reducing agent, converting a simple oxyacid into a precursor for industrial polymers, and converting a fertilizer-like compound into a plant-protective fungicide. Across agriculture, industry, synthesis, pharmaceuticals, and environmental management, phosphorous acid remains a molecule whose structure fuels its utility. Understanding its composition, formula, and applications offers insight into the subtle ways in which chemical bonding shapes technology, supports biological systems, and supplies solutions to challenges in modern science and sustainable development.