Phosphorus Pentachloride – Structure, Formula, Bonding Behavior, Reaction Patterns, Chemical Transformations, and Its Influence on Industrial and Laboratory Synthesis
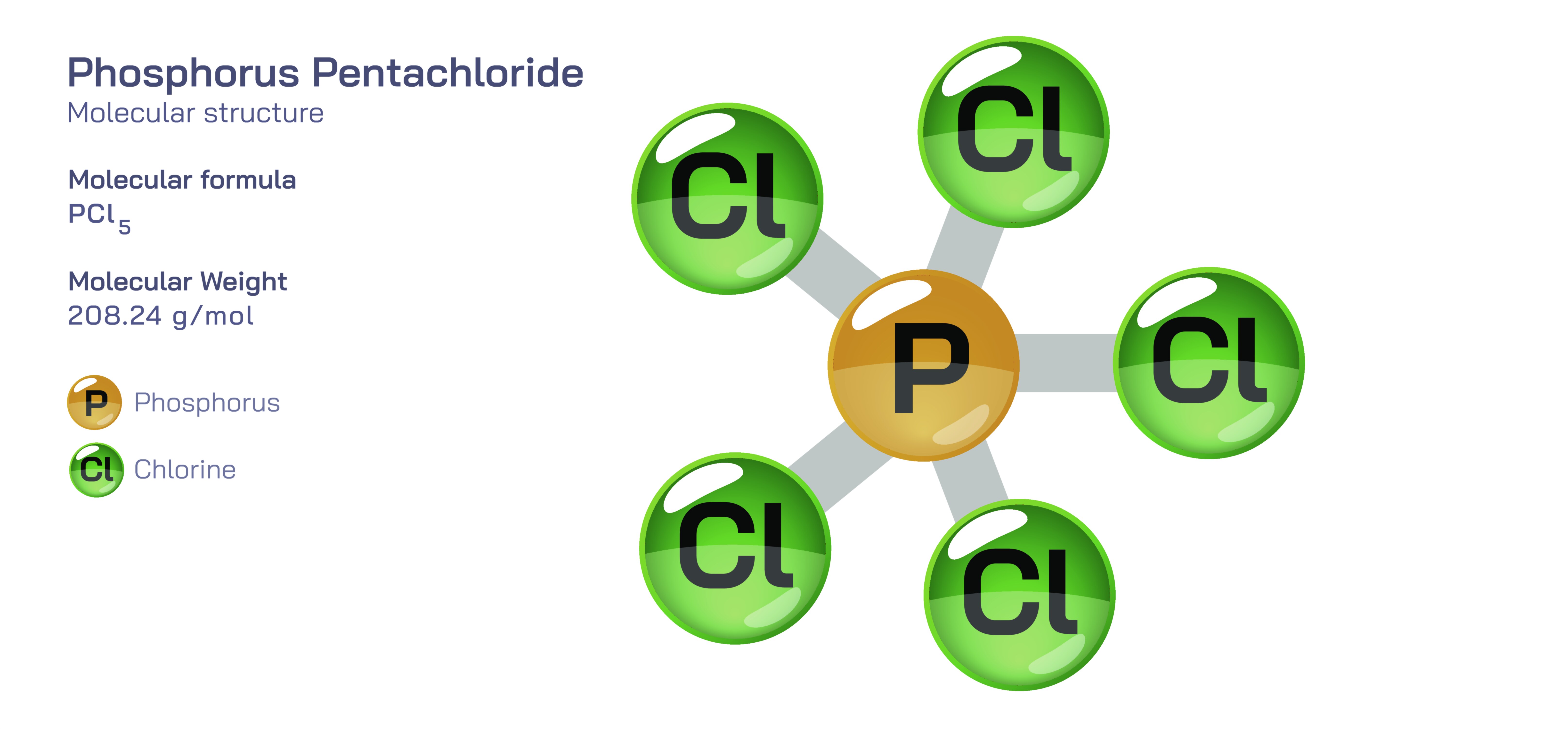
Phosphorus pentachloride is one of the most fascinating and strategically important reagents in inorganic and industrial chemistry because its molecular structure gives it the versatility to act as a chlorinating agent, a dehydration facilitator, a catalyst, and a precursor to several other phosphorus-based compounds. With the chemical formula PCl₅, this compound represents a maximum chlorination state of phosphorus, where a single phosphorus atom is bonded to five chlorine atoms. In the solid state, phosphorus pentachloride exists as an ionic lattice made of [PCl₄]⁺ cations and [PCl₆]⁻ anions, a structural adaptation that stabilizes the strong polarization of its P–Cl bonds. However, in the gas phase and many nonpolar solutions, PCl₅ adopts a trigonal bipyramidal geometry, with three chlorine atoms arranged in an equatorial plane around the phosphorus atom and two axial chlorine atoms positioned vertically above and below. This dual structural identity — ionic in the solid state and trigonal bipyramidal in the molecular state — contributes to PCl₅’s characteristic reactivity and expands its usefulness across a wide span of chemical transformations. Because phosphorus is in the +5 oxidation state, PCl₅ is an electron-deficient compound that behaves as a powerful electrophile, enabling it to substitute hydroxyl groups with chlorine and participate in reactions that shape organic, inorganic, and industrial chemistry.
At room temperature, phosphorus pentachloride appears as a pale yellow crystalline solid with a sharp, pungent odor and a high affinity for moisture. Upon exposure to atmospheric humidity, it hydrolyzes vigorously and releases hydrogen chloride vapors, which give the compound its characteristic fuming appearance. This sensitivity to water illustrates one of PCl₅’s most important properties: it reacts readily with hydroxyl-containing substances and converts –OH groups into chlorides. When phosphorous pentachloride is dissolved in polar solvents such as nitrobenzene or chlorinated hydrocarbons, it largely retains its trigonal bipyramidal character. When molten or dissolved in strongly ionic environments, it may exist in equilibrium between molecular PCl₅ and ionic fragments [PCl₄]⁺ and [PCl₆]⁻, a balance that explains why its structural identity shifts depending on the phase and medium. The presence of multiple stable configurations highlights the compound’s electronic flexibility — an attribute that not only influences how it behaves but also how it modifies other molecules during transformations.
One of the most important chemical reactions associated with phosphorus pentachloride is its interaction with water, which produces phosphoryl chloride (POCl₃) and hydrogen chloride gas in a stepwise hydrolysis process. When water is present in excess, further hydrolysis converts POCl₃ into phosphoric acid, releasing additional HCl. These reactions are intensely exothermic and illustrate why PCl₅ must be handled strictly under anhydrous conditions. The strong preference of the molecule for reacting with –OH groups is what makes it such a dominant reagent for chlorination of alcohols, conversion of carboxylic acids into acyl chlorides, and transformation of inorganic oxyacids into their corresponding chlorides. In organic chemistry, phosphorus pentachloride replaces hydroxyl groups with chlorine, often using a mechanism that proceeds through transient formation of chlorophosphonium intermediates. For alcohols, PCl₅ converts primary and secondary alcohols into alkyl chlorides and tertiary alcohols into tertiary chlorides through a mechanism that usually goes to completion with high yield. For carboxylic acids, PCl₅ forms acid chlorides, which are key intermediates in the synthesis of esters, amides, anhydrides, pharmaceuticals, polymers, and specialty organic materials. This capability fundamentally underpins many industrial and fine-chemical processes because acid chlorides serve as activated derivatives that streamline further reaction pathways.
Phosphorus pentachloride also reacts with inorganic oxy compounds, such as phosphorus oxychloride, sulfuric acid, nitric acid, and borates, to convert terminal oxygen atoms into chlorine or to generate mixed oxychlorides. Its ability to act as a dehydrating agent further extends its role beyond chlorination: PCl₅ is used in the synthesis of nitriles from amides, alkenes from alcohols, and heterocyclic compounds from diols or amino alcohols, especially when water removal is required during ring closure. In polymer chemistry, PCl₅ participates in the functionalization of polyesters, polycarbonates, and fire-retardant additives. In research settings, its controlled reactivity with organometallic compounds allows chemists to tailor metal coordination spheres, influencing catalyst behavior and ligand design. The compound’s ability to switch between ionic and molecular configurations helps support electrophilic and nucleophilic pathways depending on the reagents present.
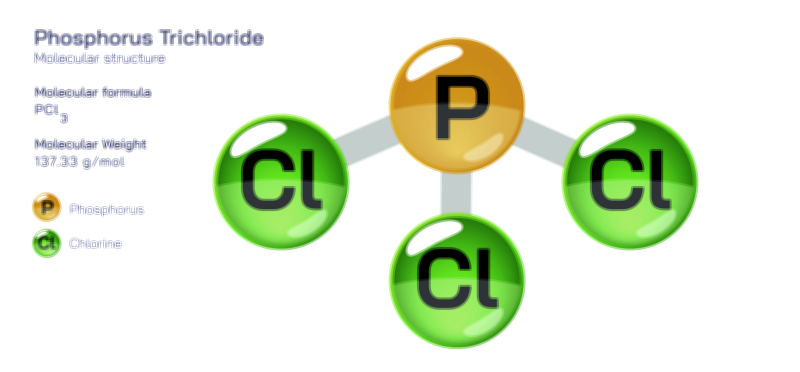
Another important consequence of phosphorus pentachloride chemistry is its ability to act as a precursor to other phosphorus chlorides, such as PCl₃ (phosphorus trichloride) and POCl₃ (phosphoryl chloride). At elevated temperatures or in the presence of chlorine acceptors, PCl₅ decomposes to PCl₃ and Cl₂, establishing an equilibrium that chemists and industry exploit for chlorine transfer reactions. Meanwhile, its controlled reaction with partially oxidized phosphorus compounds yields phosphoryl chloride, one of the most critical industrial reagents for the manufacture of dyes, pharmaceuticals, plasticizers, fire retardants, and semiconductors. This network of transformations explains why phosphorus pentachloride is considered a central member in the phosphorus halide family — because it connects reduced and oxidized phosphorus species through reactions that redistribute chlorine.
Industrially, phosphorus pentachloride serves as a critical reagent for manufacturing pharmaceutical intermediates, pesticides, flame retardants, surfactants, synthetic fibers, dyes, and specialty polymers. The conversion of carboxylic acids to acyl chlorides using PCl₅ forms the backbone of manufacturing urethane materials, polyamides, polyesters, and many drug molecules, since acyl chlorides provide high reactivity toward nucleophiles. In the pesticide sector, PCl₅ allows the formation of organophosphorus herbicides, insecticides, and fungicides, especially through synthesis routes that require substitution at the phosphorus atom or rearrangement into phosphonate structures. For flame-retardant chemistry, it enables the manufacture of phosphates and phosphonates that reduce flammability in electronics, building materials, textiles, and automotive interiors. In semiconductor and electronics fabrication, phosphoryl chloride derived from PCl₅ plays an irreplaceable role in doping silicon wafers, supporting microchip development and digital device performance. These applications show that phosphorus pentachloride may not appear in everyday consumer products, but it is foundational to the production processes that support technology, agriculture, and medicine.
Due to its vigorous reactivity, phosphorus pentachloride must be handled responsibly, especially in the presence of moisture, bases, or organic functional groups that may react explosively or release toxic HCl. Industrial processes therefore use closed systems and anhydrous solvents to manage exposure and prevent accidental hydrolysis. Proper ventilation, dry nitrogen atmospheres, corrosion-resistant storage materials, and safety monitoring protocols are standard requirements for handling this chemical. Although PCl₅ itself seldom persists in the environment because it hydrolyzes quickly, the downstream products it helps make — especially pesticides and flame retardants — must be regulated to protect ecosystems and public health. This dual nature, practical and cautionary, emphasizes that industrial chemistry yields great value only when guided by strict safety and sustainability principles.
When phosphorus pentachloride is examined from a broader scientific perspective, it becomes a compelling case study of how structure dictates function. A trigonal bipyramidal arrangement, a lone pair-accepting phosphorus center, electron-withdrawing chlorine ligands, and the ability to shift into ionic configurations all combine to make the molecule a highly selective chlorination and dehydration reagent. Because the structure can reorganize to suit the surrounding conditions, phosphorus pentachloride has an unusual ability to influence reaction pathways and generate useful molecular precursors from relatively inert starting materials. Whether transforming alcohols, activating carboxylic acids, modifying inorganic frameworks, generating industrial phosphorus derivatives, or supplying essential materials for agriculture and electronics, phosphorus pentachloride stands as one of the cornerstone reagents in synthetic chemistry.
In summary, phosphorus pentachloride is far more than a five-chlorine inorganic compound. It is a structural gateway molecule whose hybrid geometries and reactivity patterns support the transformation of simple raw materials into the complex chemical technologies that drive modern society. From chlorination and dehydration reactions in laboratories to large-scale production of pharmaceuticals, pesticides, polymer intermediates, electronics materials, and flame-retardant additives, PCl₅ continues to shape the technical capabilities and scientific innovations of the world. Understanding its structure, formula, and reactions offers a powerful demonstration of how inorganic chemistry underpins the evolution of industrial progress, technological development, and advanced synthesis in the modern age.