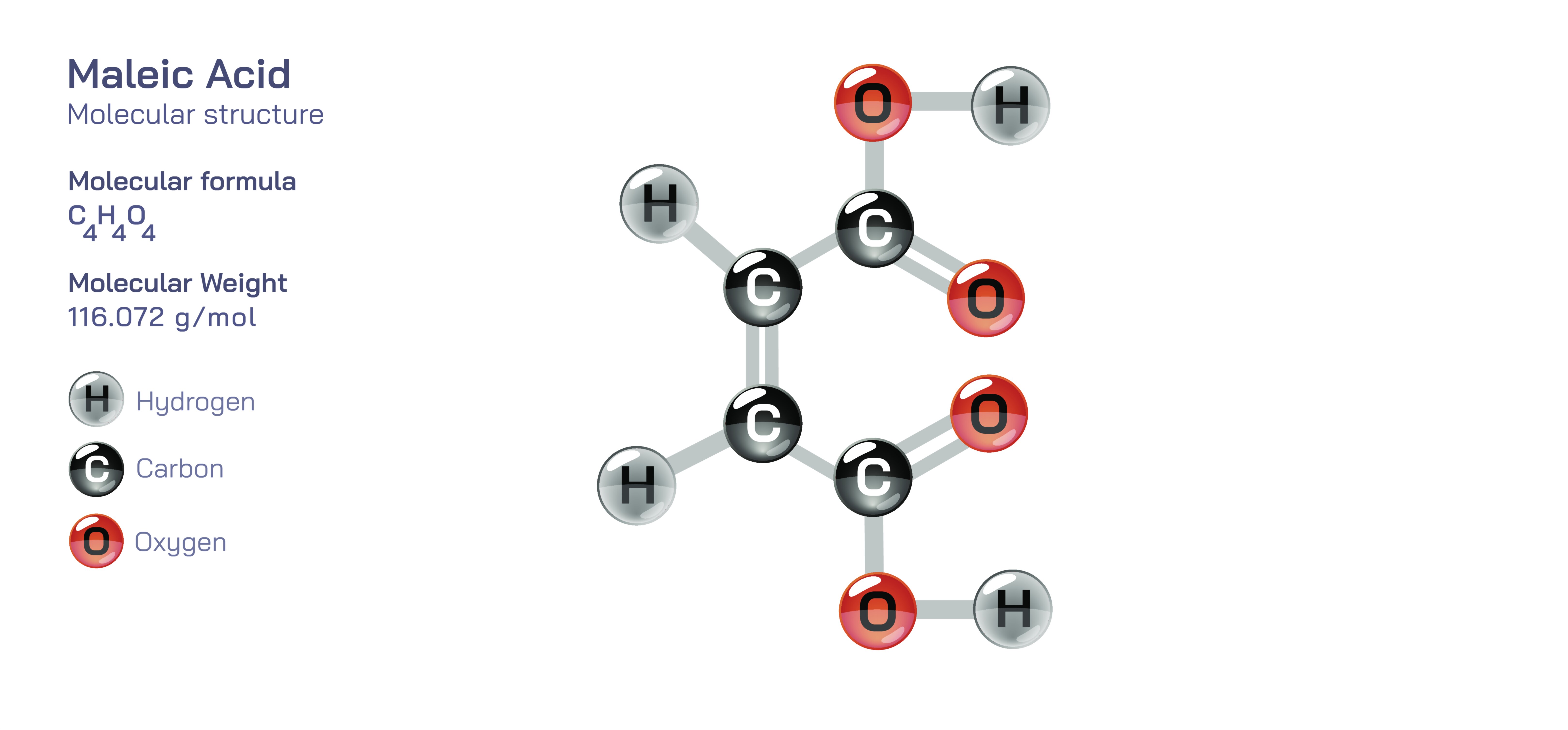
Maleic Acid — Molecular Structure and Composition
Maleic acid is an organic compound of major significance in chemistry and industry, recognized for its unique molecular arrangement, its reactivity arising from the specific geometry of its carbon–carbon double bond, and its capacity to interact with other molecules in ways that continue to shape fields such as polymer science, pharmaceuticals, agriculture, resins, coatings, and water treatment. At the molecular level, maleic acid belongs to the dicarboxylic acid family, consisting of a carbon backbone with two carboxyl groups positioned in a cis configuration across a carbon–carbon double bond. This geometric detail is the most defining structural feature of maleic acid; both carboxyl groups sit on the same side of the double bond, creating a close spatial relationship that influences electron distribution, internal hydrogen bonding, acidity, and reactivity. While maleic acid shares the same atomic composition as its geometric isomer fumaric acid, the change in the arrangement of atoms leads to dramatic differences in solubility, melting behavior, reactivity with nucleophiles, and participation in addition reactions, with maleic acid exhibiting a much higher degree of internal strain and electron density localization that make it far more chemically active. This reactivity explains why maleic acid frequently appears as an industrial feedstock and laboratory reagent: its double bond and its two carboxyl groups offer multiple sites for further transformation and polymerization, making it a highly adaptable platform for generating new molecular structures.
A deeper understanding of maleic acid begins with recognizing that its carboxyl groups, positioned close together, experience electronic repulsion and steric strain, which elevate their ability to dissociate in solution and release hydrogen ions more readily than many other dicarboxylic acids. As a result, maleic acid behaves as a stronger acid than fumaric acid or succinic acid, and its aqueous solutions show distinctive buffering and complexing behavior. The internal hydrogen bonding that occurs between the two carboxyl groups further modulates its acidity and leads to a predictable pattern of dissociation that chemists take advantage of when designing reactions that require controlled proton release. The double bond in the center of the molecule imposes rigidity and prevents rotational motion, freezing the molecule into a fixed orientation and enhancing directional selectivity during chemical reactions. When maleic acid is converted to its anhydride form through dehydration, its reactivity becomes even more pronounced, allowing it to serve as a gateway for synthesizing a wide range of esters, copolymers, cross-linked resins, and specialty additives used in modern manufacturing.
Maleic acid was introduced into industrial chemistry not only because of its structure but because of how that structure interacts with other materials. When incorporated into polymer chains through copolymerization, it adds sites for binding, adhesion, and cross-linking, dramatically altering the performance of plastics, coatings, and composite materials. Maleic acid–containing polymers contribute to improved durability, flexibility, water resistance, and adhesion to metal or wood surfaces, making them valuable in automotive finishes, construction coatings, marine paints, adhesives, fiber-reinforced materials, and water-soluble dispersants. The chemical architecture of maleic acid allows it to act as a compatibilizer in polymer blends, improving the interfacial bonding between different polymers that would not otherwise mix effectively. This role is crucial in modern recycled plastics strategies, where blending polymers that normally phase-separate can be stabilized by maleic structures that provide molecular bridges between components. These benefits are not simply incidental; they arise directly from the ability of maleic acid to introduce polar regions, reactive carboxyl groups, and structural rigidity into polymer networks.
In biomedical and pharmaceutical research, maleic acid has a very different but equally meaningful identity. Its two carboxyl groups allow it to form stable salts and complexes with active pharmaceutical ingredients, improving solubility, bioavailability, chemical stability, or controlled release properties. The maleate form of many drugs has been used to ensure predictable absorption and delivery within the body, highlighting the role of maleic acid not as a therapeutic molecule itself, but as a structural partner that supports drug transport and formulation. In biochemical studies, the molecule is sometimes employed to modulate pH, chelate metal ions, or stabilize enzymes in controlled conditions where small shifts in acidity or ionic balance can significantly influence biological activity. The inherent reactivity of maleic acid also allows researchers to modify proteins, peptides, or polysaccharides in laboratory settings, creating conjugates or altered biomolecular surfaces that help investigate cellular systems or design biomedical materials.
Maleic acid also plays a sophisticated role in environmental technology and water treatment. Its strong tendency to complex with metal ions makes it useful in preventing unwanted deposits and scaling in water circulation systems, cooling towers, boilers, and irrigation pipelines. In formulations used for soil remediation, industrial wastewater purification, and municipal water treatment, maleic-based copolymers and dispersants bind or suspend inorganic particles so they remain soluble rather than precipitating out and accumulating. This same principle contributes to detergent and cleaning formulations, where maleic acid derivatives prevent mineral buildup on fabrics or surfaces by sequestering metal ions and preventing insoluble deposits. Because the molecule and its optimized derivatives can function effectively at a range of pH levels, they have become an enabling technology in sustainable water management systems designed to reduce chemical consumption and improve system efficiency over long operational lifespans.
In agriculture, maleic acid is used not as a nutrient but as a regulator of plant physiology and as an intermediate for compounds that influence plant development. Certain maleic acid derivatives have been evaluated for their ability to modify germination rates, slow excessive sprouting in stored potatoes or onions by temporarily interrupting biochemical signaling, and alter responses to environmental stimuli. These effects do not arise from general toxicity but from the subtle influence of maleic-based compounds on metabolic pathways that involve energy transfer, organic acid balance, and cellular regulation. The chemical profile of maleic acid also lends itself to the production of environmentally friendly pesticide carriers and slow-release agrochemicals, where its ability to form biodegradable matrices helps ensure that active compounds remain available for an extended period without rapid washout or environmental accumulation.
Even though maleic acid has a wide spectrum of uses, its handling must be thoughtful and informed. Its strong acidity means that in concentrated form it can be corrosive to tissues, and in industrial environments inhalation of mist or dust should be prevented. When used properly, however, maleic acid is a highly predictable and controllable compound, and its breakdown products occur naturally in biochemical and environmental cycles. The molecule’s reactivity is intertwined with its value: the same structural features that make it corrosive at high concentrations are also what allow it to polymerize, chelate, disperse, bind, and modify materials in beneficial ways when placed in controlled systems.
When viewed from a broader perspective, the story of maleic acid illustrates how a single change at the molecular scale can ripple outward and influence entire fields of science and technology. By having both carboxyl groups in a cis arrangement, maleic acid becomes a molecule of heightened chemical potential and selectivity. That seemingly small structural detail influences acidity, solubility, reactivity, polymer performance, biological formulation, and environmental compatibility. It demonstrates the principle that composition alone is not the sole determinant of function; rather, the spatial arrangement of atoms, the distribution of electrons, and the relationship between neighboring groups define the true chemical identity of a molecule. In chemistry laboratories, in industrial plants, in pharmaceutical development teams, and in the design of new sustainable materials, maleic acid continues to act as a cornerstone molecule whose geometric and electronic properties make it indispensable for innovation. Through its structural uniqueness and wide-ranging functional behavior, maleic acid stands as a compelling example of how molecular architecture shapes modern science, industry, and environmental progress.