Pyridine – Structure, Formula, Aromaticity, Bonding Behavior, Chemical Properties, Reactivity, and the Industrial and Biological Significance of This Foundational Heterocyclic Compound
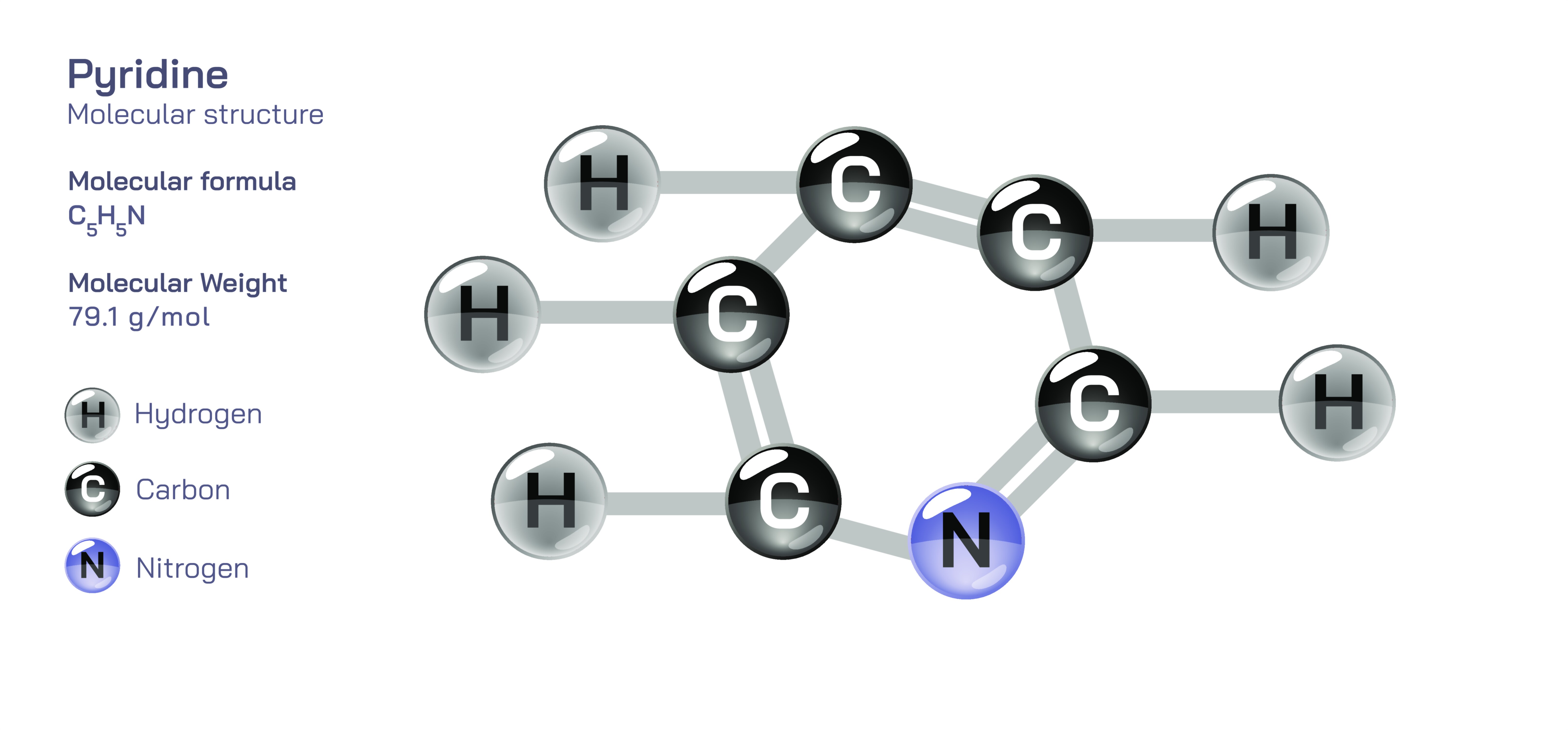
Pyridine is one of the most influential molecules in heterocyclic and organic chemistry because its structure combines the familiar aromaticity of benzene with the chemical versatility of nitrogen. Its molecular formula is C₅H₅N, and its structure consists of a six-membered ring containing five carbon atoms and one nitrogen atom. Although the ring resembles benzene in overall shape and electron delocalization, substituting a nitrogen atom fundamentally alters the molecule’s physical and chemical behavior. In pyridine, the nitrogen atom replaces one CH group of benzene and contributes a lone pair of electrons that is not part of the aromatic π-electron system. This lone pair, positioned in an sp² orbital instead of participating in resonance, gives pyridine its mildly basic nature and makes it capable of donating electrons to acids and metal centers. At the same time, the aromatic ring maintains a six-π-electron conjugated system, preserving the stability, planarity, and ring currents associated with aromatic compounds. Thus, pyridine combines the stability of benzene with the reactivity of nitrogenous bases, unlocking a wide spectrum of chemical interactions and industrial applications.
In its physical appearance, pyridine is a colorless to pale yellow liquid with a strong, distinctive smell often described as sharp and unpleasant, reminiscent of stale fish or decayed tobacco. It mixes freely with water due to the polarity created by the electronegative nitrogen atom, making pyridine much more water-soluble than benzene. The boiling point of pyridine (about 115°C) is relatively high for a six-membered ring system, reflecting the influence of dipole–dipole interactions and hydrogen bonding between pyridine and water molecules. The molecule is planar, maintaining a trigonal geometry around the nitrogen atom, and the lone pair remains orthogonal to the aromatic π cloud. This structure clarifies why pyridine behaves as a Lewis base and proton acceptor without disturbing its aromaticity: the nitrogen lone pair is available for bonding, while the π-electrons maintain cyclic conjugation. This duality—basicity without sacrificing aromatic stability—forms the key to many of its chemical properties.
One of pyridine’s most defining chemical characteristics is its basicity. Compared to aliphatic amines, pyridine is a weaker base because its nitrogen lone pair is held in an sp² hybrid orbital close to the nucleus, reducing its availability for proton acceptance. Nonetheless, pyridine readily forms salts with strong acids, generating pyridinium ions in which the nitrogen becomes positively charged. The presence of nitrogen also significantly influences the electrophilic and nucleophilic reactivity of the ring. Electrophilic aromatic substitution occurs much more slowly than in benzene because nitrogen withdraws electron density from the ring, reducing its susceptibility to attack by electrophiles. When such reactions do occur, they preferentially take place at the meta-position relative to nitrogen, because the ortho and para positions are destabilized by the ring’s electron deficiency. In contrast, nucleophilic aromatic substitution reactions proceed more easily in pyridine than in benzene, typically favoring the ortho and para positions. These contrasting patterns highlight how nitrogen reshapes the electron distribution of the ring and makes pyridine a tunable and selective substrate in synthetic chemistry.
Pyridine also acts as a ligand, coordinating to metal ions through the lone pair on nitrogen. In coordination chemistry, pyridine binds to transition metals to form stable complexes that influence metal reactivity, electron transfer, catalysis, and ligand substitution kinetics. The aromatic ring stabilizes the coordination bond through electron delocalization, helping pyridine participate in catalyst design and organometallic frameworks. Pyridine’s involvement as a coordinating ligand is especially important in homogeneous catalysis, nanoparticle stabilization, metal–organic frameworks, and in bioinorganic models that mimic metalloprotein binding sites. This property reinforces that pyridine is not only a reagent but a structural component that shapes the architecture and function of catalytic systems.
In laboratory synthesis, pyridine occupies a central practical role. It is commonly used as a mild base, solvent, and acid scavenger—particularly in reactions involving acyl chlorides and sulfonyl chlorides, where pyridine binds to the released HCl and prevents unwanted protonation of reactants. It also helps activate alcohols in esterification processes, forming stabilizing intermediates that drive reactions forward. Pyridine derivatives act as catalysts in reactions such as the Vilsmeier–Haack formylation, Hantzsch synthesis, and chlorination of alcohols, illustrating how pyridine can function both as a base and as a precursor to aromatic reagents. Because pyridine is heat stable and dissolves both organic and polar compounds, it serves effectively as a solvent in specialized reactions where water and alcohols are unsuitable. This versatility has made pyridine an indispensable laboratory tool throughout the history of organic synthesis.
On an industrial scale, pyridine is valuable both as a raw material and as an intermediate. It is used in the synthesis of agricultural chemicals, pharmaceuticals, antioxidants, dyes, adhesives, rubber accelerators, and corrosion inhibitors. Pyridine derivatives form the backbone of important compounds such as nicotinamide (vitamin B₃), antihistamines, antibacterial agents, tuberculosis medications, and anti-inflammatory drugs. Many herbicides and insecticides—including those used in crop protection—contain pyridine rings because the nitrogen functionality allows biological targeting and selective enzyme interaction. In polymer and material science, pyridine derivatives contribute to stabilizers, plasticizers, and cross-linking agents that enhance flexibility, thermal resistance, and UV durability. Flame retardants and battery electrolyte additives also rely on pyridine-based molecules due to their ability to modulate charge transfer and ion transport.
Pyridine’s biological relevance extends beyond synthetic drugs. It is structurally related to nicotinamide adenine dinucleotide (NAD⁺/NADP⁺)—crucial cofactors that mediate metabolic redox reactions in living organisms. The energy flow inside cells depends heavily on electron transfer involving these cofactors, both of which contain a pyridine ring at their core. This profound connection between a laboratory molecule and cellular metabolism demonstrates how nitrogen-containing aromatic systems support life’s biochemical energy network. It also explains why pyridine rings often appear in medicinal compounds: biological receptors evolved to interact with nitrogenous heterocycles long before chemists synthesized them.
Despite its usefulness, pyridine requires careful handling. It is toxic when inhaled or ingested and can cause severe irritation to skin and membranes. Its strong odor makes leaks detectable, but laboratory ventilation and industrial exposure controls are essential. Pyridine’s environmental persistence requires regulated disposal, and greener synthetic routes continue to be developed to reduce ecological impact. Yet, with responsible management, pyridine remains an invaluable component of chemistry and industry.
Ultimately, pyridine provides a vivid demonstration of how a small structural change—replacing one carbon atom of benzene with nitrogen—can transform the properties and reactivity of an entire molecule. By maintaining aromatic stability while introducing basicity, polarity, and electron-withdrawing behavior, pyridine becomes far more than an aromatic ring; it becomes a multifunctional building block that drives synthesis, catalysis, biology, and industrial technology. Whether enabling laboratory reactions, forming the framework of pharmaceutical structures, controlling electronic and catalytic behavior, or participating in metabolic chemistry, pyridine stands as one of the most important heterocyclic compounds in science. Understanding its structure, formula, and chemical properties reveals not only how heterocycles work, but how carefully tuned molecular design empowers the advancement of chemistry, medicine, and materials across the modern world.