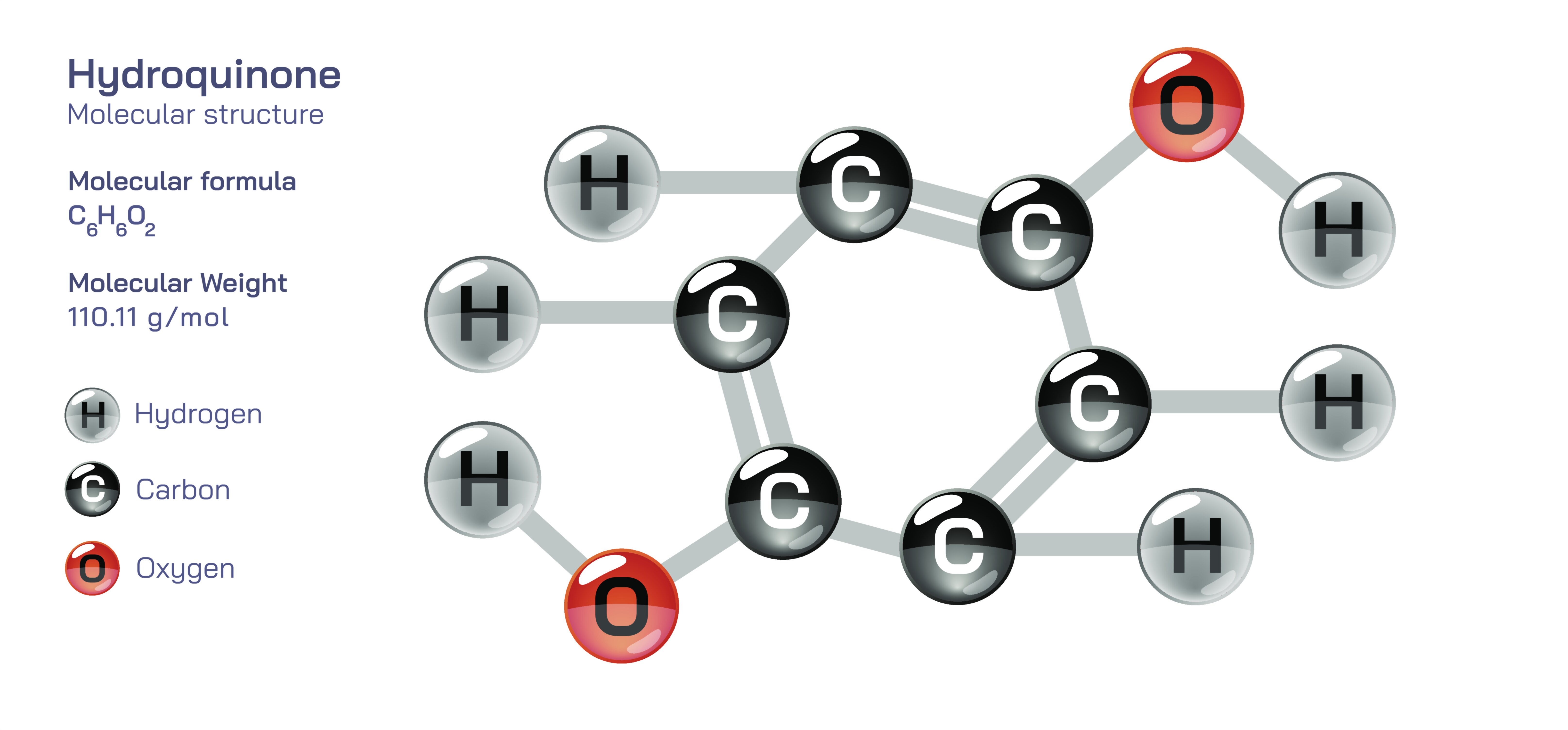
Hydroquinone Chemical Structure — C₆H₆O₂ Molecule Diagram and Molecular Behavior Explained
Hydroquinone is a small but highly influential organic compound whose structure, reactivity, and biological effects make it one of the most important molecules in both chemistry and dermatology. Although its chemical formula, C₆H₆O₂, appears simple on the surface, the deeper meaning of this structure becomes clear only when the arrangement of those atoms inside the benzene ring is examined. Hydroquinone is a dihydroxybenzene, meaning that it consists of a benzene ring — an aromatic ring of six carbon atoms connected in a conjugated cyclic structure — bearing two hydroxyl (–OH) groups positioned directly opposite one another. This orientation, known as the para position, distinguishes hydroquinone from other dihydroxybenzene isomers such as catechol (ortho) and resorcinol (meta). The para arrangement of the hydroxyl groups dictates not only the electron distribution and hydrogen bonding potential of the molecule but also the way it participates in redox chemistry, how it interacts with biological enzymes, and why it possesses both therapeutic and hazardous effects depending on context and concentration. The molecule’s aromatic core provides exceptional stability, but its hydroxyl groups create reactivity that makes hydroquinone capable of influencing pigmentation pathways in skin and functioning as a highly effective reducing agent in industrial and laboratory settings.
At the structural level, hydroquinone derives its properties from the interaction between its electron-rich benzene ring and its two hydroxyl substituents. Each hydroxyl group donates electron density into the aromatic system, making the ring highly activated toward redox reactions. These hydroxyl groups also make the molecule capable of forming intermolecular and intramolecular hydrogen bonds, affecting solubility, melting point, and reactivity. One of the defining chemical features of hydroquinone is its ability to reversibly oxidize to benzoquinone by losing two hydrogen atoms and forming two carbonyl groups on the ring. This redox pair — hydroquinone and benzoquinone — constitutes a fundamental reversible system in organic chemistry. Because the transformation can occur readily under mild conditions, hydroquinone is an excellent electron donor and participates in reduction processes in photography, polymerization inhibition, metal finishing, and dye chemistry. Its redox cycling ability is central to its biological activity as well: hydroquinone can interfere with melanin synthesis in skin by competing with the amino acid tyrosine, disrupting the enzymatic process that creates pigment. This interference is what allows hydroquinone to function as a pigment-lightening agent in dermatology, but it also creates the foundation for its potential toxicity when misused, because melanin inhibition and oxidative stress pathways are closely intertwined.
Within dermatology, hydroquinone became widely known for its ability to treat hyperpigmentation conditions such as melasma, post-inflammatory discoloration, age spots, and sun-related darkening. When applied topically under medical supervision, hydroquinone penetrates the epidermis and interacts directly with melanocytes — the pigment-producing cells of the skin. It inhibits the enzyme tyrosinase, which catalyzes the conversion of tyrosine into melanin, and it also affects the transport and distribution of pigment inside skin cells. Over time, melanin production decreases and existing pigmentation becomes less apparent. The scientific principle behind this effect is directly related to the molecule’s structural similarity to the natural melanin precursor, allowing hydroquinone to act as a molecular “decoy.” However, because hydroquinone participates in redox cycles, prolonged or excessive exposure may lead to oxidative stress in skin cells, and without medical guidance, this can result in irritation, inflammation, paradoxical darkening (exogenous ochronosis), or hypersensitivity. For this reason, although hydroquinone is extremely effective in pigmentation management, its therapeutic potential cannot be separated from the need for responsible use, appropriate concentration, limited duration, and professional oversight.
The industrial value of hydroquinone arises from the same redox behavior that governs its biological activity. In chemical manufacturing, hydroquinone is employed as a polymerization inhibitor — preventing monomers such as styrene, acrylics, and vinyl compounds from reacting prematurely during storage and transport. It accomplishes this by trapping reactive radical species that would otherwise initiate uncontrolled polymer growth. Hydroquinone’s ability to donate electrons makes it essential in photographic developing solutions, where it reduces silver ions to metallic silver on film surfaces. It is also used in the production of antioxidants, dyes, rubber stabilizers, and fuel additives. Across all of these applications, the para-dihydroxy arrangement of the molecule determines its chemical reactivity: neither catechol nor resorcinol provides the same balance of electron distribution needed for such predictable redox cycling, highlighting how a seemingly small change in structure can dramatically change the function of an aromatic compound.
The safety profile of hydroquinone must be considered with equal attention to its structure. Because hydroquinone is an aromatic reducing agent, its interaction with biological tissues is dose-dependent — safe in controlled dermatological quantities under supervision, but potentially harmful when overused or taken internally. High concentrations can cause skin irritation, allergic reactions, cytotoxicity, and chronic pigmentation disorders. Systemic absorption in large quantities is associated with oxidative damage to red blood cells and cellular biomolecules. Regulatory agencies around the world therefore set concentration limits for hydroquinone in cosmetic products, and many regions permit only prescription-based formulations to ensure that its use is monitored appropriately. These safety restrictions are not a reflection of hydroquinone’s lack of usefulness but an acknowledgment that a powerful biochemical tool must be applied with precision rather than casual experimentation.
When viewed from a chemical perspective, hydroquinone demonstrates how a small structural motif can produce immense functional diversity. The benzene ring grants stability, the para-positioned hydroxyl groups generate selective reactivity, and the redox cycling capacity defines both industrial and biological roles. The molecule can brighten skin, stabilize polymers, develop photographic images, and inhibit free-radical reactions — all because of the unique coexistence of aromatic stability and electron-donating hydroxyl groups. Hydroquinone is therefore not a compound whose identity can be reduced to a single application or hazard. It is a molecular framework whose chemical behavior is inseparable from the positions of its atoms and the movement of electrons across its aromatic system.
Understanding hydroquinone requires looking beyond its formula to the structural story that determines its real-world behavior. Its C₆H₆O₂ composition becomes meaningful only through the arrangement of atoms on the ring, the location of the hydroxyl groups, and the ease with which it can move between reduced and oxidized states. It is in this story — where geometry shapes reactivity and reactivity shapes application — that hydroquinone reveals its true nature as one of the most versatile, powerful, and scientifically significant aromatic molecules in modern chemistry and medicine.