Phosphate Ion – Structural Arrangement, Bonding Nature, Acid–Base Behavior, Charge Distribution, Biological Importance, Environmental Role, and Chemical Reactivity of One of the Most Fundamental Polyatomic Ions in Chemistry
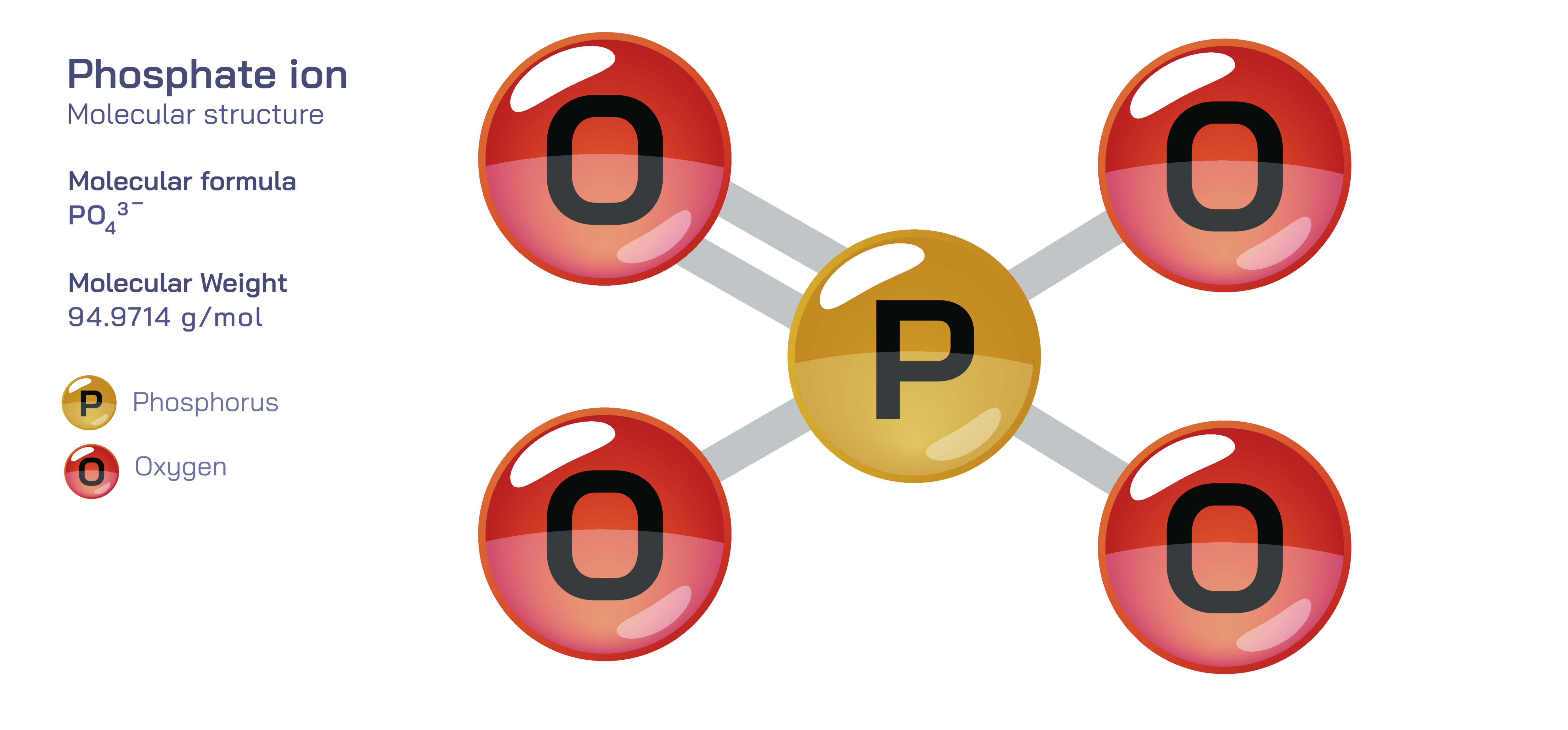
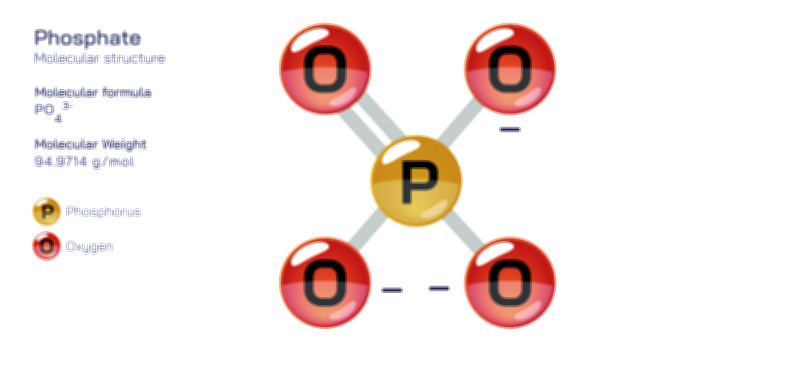
The phosphate ion is one of the most significant and versatile polyatomic ions in chemistry, central to the functioning of biological life, geochemical processes, and industrial materials. Represented by the formula PO₄³⁻, the phosphate ion consists of one phosphorus atom surrounded tetrahedrally by four oxygen atoms. The geometry is governed by the principles of molecular bonding, where the phosphorus atom forms four P–O single bonds in an arrangement that minimizes electron pair repulsion and creates a perfectly symmetrical tetrahedral shape. The ion carries an overall charge of −3 because three of the oxygen atoms each bear a formal negative charge after bonding, while one oxygen typically forms a double bond or partial double-bond character through resonance with phosphorus. The resonance stabilization across the four P–O bonds distributes electron density evenly, giving the phosphate ion a remarkably stable and low-energy electronic structure. This stability is one of the reasons phosphate is abundant in minerals, biological molecules, and aqueous chemistry. The tetrahedral architecture and balanced charge distribution also contribute to strong ionic interactions, hydrogen bonding tendencies, and complexation with metal ions in natural and industrial contexts.
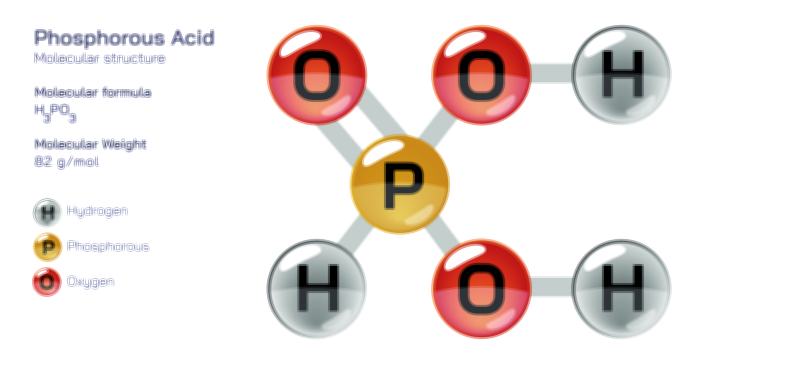
Chemically, the phosphate ion is classified as the conjugate base form of phosphoric acid (H₃PO₄), and its charge state reflects the acid–base equilibria of this parent molecule. Phosphoric acid is triprotic, meaning it can lose three hydrogen ions in successive steps, and the phosphate ion represents the fully deprotonated form produced in strongly basic conditions. Between phosphoric acid (H₃PO₄) and phosphate (PO₄³⁻) exist intermediate species — dihydrogen phosphate (H₂PO₄⁻) and hydrogen phosphate (HPO₄²⁻) — that dominate at different pH ranges. This acid–base versatility allows phosphate to function as an exceptionally effective buffer system in biological fluids, laboratory solutions, and industrial processes. In living organisms, the phosphate system in blood and intracellular fluids helps resist sudden shifts in pH by absorbing or releasing protons depending on environmental acidity. The ability of phosphate species to interconvert smoothly according to pH makes them indispensable to cellular chemistry, where metabolic reactions and biochemical stability depend on remarkably precise control of hydrogen ion concentration.
The bonding characteristics of the phosphate ion explain many of its physical and reactive properties. Phosphorus in the center of the tetrahedral structure forms strong covalent bonds with oxygen, but because the oxygen atoms bear high electronegativity, the P–O bonds exhibit partial ionic character. This combination of covalent and ionic character creates a robust structural integrity that resists hydrolysis and prevents phosphate from spontaneously gaining or losing oxygen atoms under normal conditions. Additionally, the negative charges distributed over the oxygen atoms make phosphate highly attracted to positively charged ions, especially multivalent metal cations such as calcium, magnesium, iron, and aluminum. This strong affinity governs phosphate precipitation reactions in soils, lakes, oceans, and wastewater systems. In the geological cycle, phosphate readily binds with calcium to form insoluble minerals such as apatite, which form essential components of rocks and skeletal tissues in animals. At the same time, overly strong binding in soil can limit phosphate bioavailability for plants, linking the chemistry of phosphate to agricultural productivity and fertilizer science.
Biologically, the phosphate ion holds extraordinary importance because it is integrated into the molecular foundation of life. DNA and RNA contain phosphate groups as part of their sugar–phosphate backbone, which provides structural support and directionality to genetic material. The negatively charged phosphate groups stabilize the helical structure and allow the strands to interact with proteins and metal ions in highly regulated ways. In cellular energy transfer, phosphate is the central functional component of adenosine triphosphate (ATP). When ATP is hydrolyzed into ADP (adenosine diphosphate) and inorganic phosphate, energy is released to drive biochemical reactions. The ability to form and break high-energy phosphate bonds is the fundamental principle underlying muscle contraction, nerve transmission, biosynthesis, and metabolic regulation. In membranes surrounding cells, phosphate contributes to the polar head groups of phospholipids, creating the amphiphilic character that allows lipid bilayers to self-assemble and form selective barriers essential for maintaining life.
Phosphate maintains key roles beyond genetic material and energy metabolism. In bones and teeth of vertebrates, calcium phosphate appears in the form of hydroxyapatite, a crystalline mineral that gives the skeletal system rigidity and stability. The interaction between calcium ions and phosphate ions in biological mineralization highlights how the inorganic chemistry of phosphate intersects with structural biology. In plant biochemistry, phosphate enables the activation of enzymes through phosphorylation, regulates the photosynthetic cycle, and supports the synthesis of essential biomolecules. Because plants require phosphate for growth and crops extract it continuously from soil, phosphate fertilizers are crucial to agriculture and global food supply. However, the environmental implications of phosphate use are significant. Excess phosphate from agricultural runoff and detergents can accumulate in lakes and rivers, causing eutrophication — a rapid increase in algae that depletes oxygen and destroys aquatic ecosystems. Thus, phosphate chemistry links food production, environmental sustainability, and ecological balance.
From an industrial perspective, phosphate ions and their derivatives are employed in manufacturing detergents, corrosion inhibitors, flame retardants, water treatment additives, fertilizers, and metal surface treatment chemicals. Phosphate buffers are essential in pharmaceutical formulations and biotechnology research because they maintain stable pH without interfering with biochemical activity. Phosphate esters function as intermediates in organic synthesis, while condensed phosphates such as polyphosphates act as sequestrants and water softeners to prevent mineral buildup in household and commercial systems. In metallurgy, phosphate coatings protect steel from rust and improve adhesion of paint. Even in electronics, phosphate-based materials contribute to lithium-iron-phosphate (LiFePO₄) batteries, valued for safety and stability in rechargeable energy storage systems. These widespread applications demonstrate that phosphate is not merely a laboratory species — it is a cornerstone of modern industry and technology.
The chemical properties of phosphate also illustrate fundamental principles of molecular behavior. The stability of the tetrahedral structure highlights how geometry and electron distribution influence the energy of a molecule. The resonance stabilization of the phosphate anion reveals how electrons delocalize to produce unusually strong acid–base behaviors. The connectivity between phosphoric acid and its three successive conjugate bases showcases the relationship between pH and molecular speciation. These ideas help connect general chemistry to biochemistry, inorganic chemistry, environmental chemistry, and industrial process design. Students who explore the phosphate ion learn not only about a particular molecule, but about the deeper logic that organizes the periodic table, supports life’s biochemical cycles, and governs the sustainability of global ecosystems.
In summary, the phosphate ion is far more than a simple PO₄³⁻ formula on a page — it represents the intersection of molecular stability, acid–base equilibria, biological energy transfer, ecological balance, and industrial utility. Its tetrahedral structure, distributed negative charge, and resonance stabilization give rise to unique chemical behavior that influences living organisms, mineral formations, agricultural systems, and engineered materials. Whether embedded in DNA, cycling through soils and oceans, powering cellular ATP reactions, forming bones and teeth, or supporting global fertilizer and manufacturing industries, phosphate remains indispensable. Understanding its structure and chemical properties provides insight into how chemistry shapes life, technology, and the environment — from the smallest molecular scale to the planetary scale.