Lithium Chloride — Chemical Structure, Properties, Hygroscopic Behaviour, and Industrial Applications
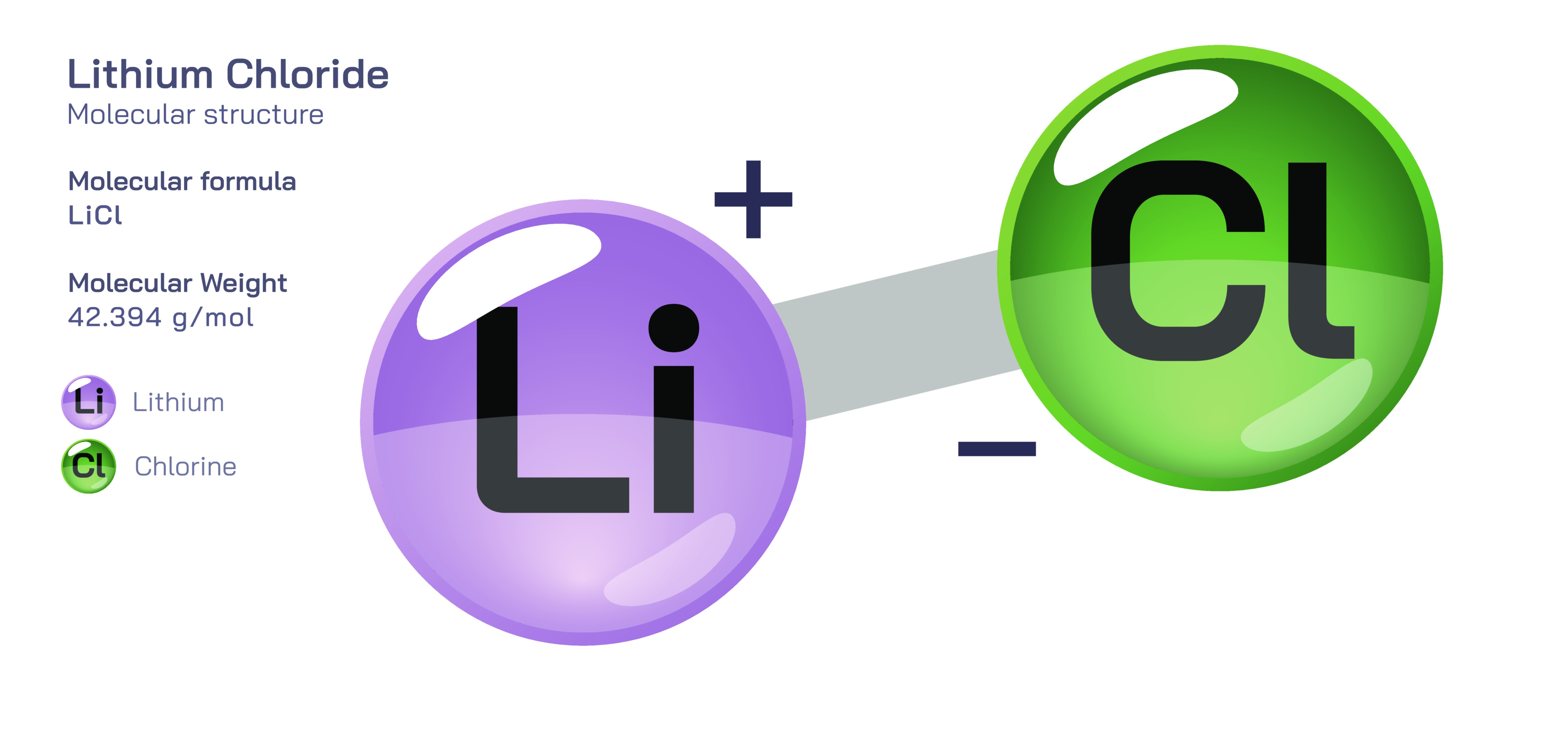
Lithium chloride is an inorganic chemical compound with the formula LiCl, composed of a lithium cation (Li⁺) and a chloride anion (Cl⁻). At first glance, it appears to be a simple salt similar to sodium chloride, yet its properties differ dramatically because of the small ionic radius and high charge density of the lithium ion. These structural and ionic characteristics give lithium chloride a strong attraction to water, extremely high solubility, and a wide range of uses in chemical processing, air-conditioning systems, metallurgy, desiccant applications, and electrochemical technology. Lithium chloride exemplifies how slight variations in ionic size and chemical bonding can produce behaviours that diverge significantly from those of related alkali metal chlorides, especially regarding thermal response, moisture absorption, melting behaviour and solvation dynamics.
In its solid form, lithium chloride crystallizes in a cubic lattice, where each chloride ion is surrounded by multiple lithium ions and vice versa. The small radius of Li⁺ produces a compact lattice structure with strong electrostatic bonding, resulting in a salt that melts at a lower temperature than expected relative to heavier alkali metal chlorides. The crystalline form is typically white and highly deliquescent, meaning that when exposed to air it readily absorbs moisture until it dissolves into a concentrated solution. This hygroscopic property distinguishes lithium chloride from many other common salts and is central to its technological applications in humidity control and air-drying systems. Unlike sodium chloride, which remains solid and relatively dry in open air, lithium chloride becomes wet rapidly because it has a much lower vapour pressure over its solutions, causing water from the atmosphere to be pulled inward until equilibrium is reached.
Lithium chloride dissolves extremely easily in water, and the dissolution process releases significant heat due to strong hydration of the lithium ion. While many salts dissolve endothermically, LiCl becomes warmer when dissolving—a property that reflects how strongly water molecules coordinate around Li⁺. In solution, lithium ions become tightly surrounded by hydration shells, forming stable complexes that influence both boiling and freezing behaviour. Because of this intense solvation, lithium chloride depresses the freezing point of water effectively and is used in laboratory brines for calibrating low-temperature equipment. At elevated temperatures, molten lithium chloride becomes an efficient electrolyte due to the mobility of ions within the melt; this characteristic contributes to its role in industrial extraction and alloy production.
The hygroscopic nature of lithium chloride places it at the centre of industrial dehumidification systems and liquid desiccant technologies. Solutions of LiCl absorb moisture so aggressively that they can remove humidity from circulating air streams without cooling the air to the dewpoint. In specialized HVAC systems, air is passed through contact surfaces coated or sprayed with lithium chloride solution, enabling dehumidification with lower energy consumption compared to mechanical drying systems. This approach is preferred in factories, pharmaceutical production halls, semiconductor facilities, museums, cold-storage units and other environments where moisture management is critical. Lithium chloride’s thermodynamic behaviour also allows it to regenerate easily: applying heat expels captured moisture, restoring the solution to a concentrated state and enabling reuse. This regenerative property gives LiCl systems an advantage in industrial humidity control where sustainability and energy efficiency matter.
Beyond its behaviour with water vapour, lithium chloride has key industrial applications due to its ionic properties. It is widely used in metallurgy, especially in flux mixtures for aluminium, magnesium, and other reactive metals. When mixed with salts such as potassium chloride, LiCl lowers melting points and improves molten metal fluidity, reducing oxidation and improving casting quality. In electrolysis processes, molten lithium chloride acts as an electrolyte for extracting metallic lithium from lithium-bearing minerals. The wide liquid temperature range and electrical conductivity of molten LiCl enable separation of elemental lithium in controlled high-temperature cells. This role in lithium metal production connects lithium chloride directly to the manufacturing of lithium batteries, aerospace alloys, and advanced rechargeable energy storage materials.
Lithium chloride also plays a role in organic and inorganic synthesis. Because lithium ions act as strong Lewis acids in polar solvents, LiCl modifies reaction environments and influences reaction selectivity and yields. It is sometimes used as a catalyst or co-catalyst for dehydration, condensation, and polymerization reactions, and its presence can alter the solubility of organometallic intermediates. In peptide synthesis and fine chemical manufacturing, LiCl affects solvent coordination and reduces aggregation of reactive species, improving reaction efficiency. In polymer chemistry, lithium chloride is used to dissolve cellulose in amide-based solvents, producing clear solutions suitable for fibre formation, coatings and research on biopolymer processing.
Additional applications extend into electronics, sensors and optics. In the ceramics and glass industry, lithium chloride contributes to formulations that lower melting temperatures, enhance thermal stability and improve clarity. In specialized photographic developing solutions and infrared optics, LiCl has historically been used for its controlled hygroscopic and refractive properties. In laboratory equipment, saturated lithium chloride solutions serve as humidity reference standards because the compound maintains a predictable equilibrium moisture content under fixed temperature conditions, allowing precise calibration of hygrometers and climate-measuring devices.
Lithium chloride requires proper handling because concentrated solutions are corrosive to metal surfaces and can irritate skin and eyes. Ingestion of large quantities is hazardous due to the reactivity of lithium ions with biological systems. Industrial facilities that use LiCl in absorption or chemical processes employ corrosion inhibitors, coating materials, compatible piping and controlled circulation to prevent damage to equipment. Unlike lithium bromide, which is primarily used in absorption chillers for cooling, lithium chloride is most dominant in applications involving moisture control, humidity conditioning, metallurgical fluxes and molten-salt electrochemistry.
What makes lithium chloride scientifically compelling is the way its microscopic ionic structure defines its macroscopic behaviour across multiple industries. The small size of the lithium ion explains its high solubility and strong hydration, which in turn explains its hygroscopicity. Hygroscopicity leads to its major function as a desiccant and humidity controller, while molten ionic behaviour leads to its role in metal extraction. Every application links back to the fundamental chemistry of lithium and chlorine, illustrating how atomic-level properties scale into large-scale industrial capability.
In summary, lithium chloride—though chemically simple—is a versatile and strategically important industrial compound. Its structure determines its solubility; its solubility determines its water-binding power; its water-binding power determines its usefulness in humidity control; and its thermal and ionic characteristics enable metallurgy, lithium production and chemical synthesis. Through these linked behaviours, LiCl demonstrates how the inherent properties of a salt can ripple across chemistry, environmental control and advanced manufacturing, making lithium chloride a significant compound across multiple scientific and engineering fields.