Aluminum Fluoride — Chemical Structure and Industrial Compound Illustration
Fundamental Identity and Structural Basis of Aluminum Fluoride
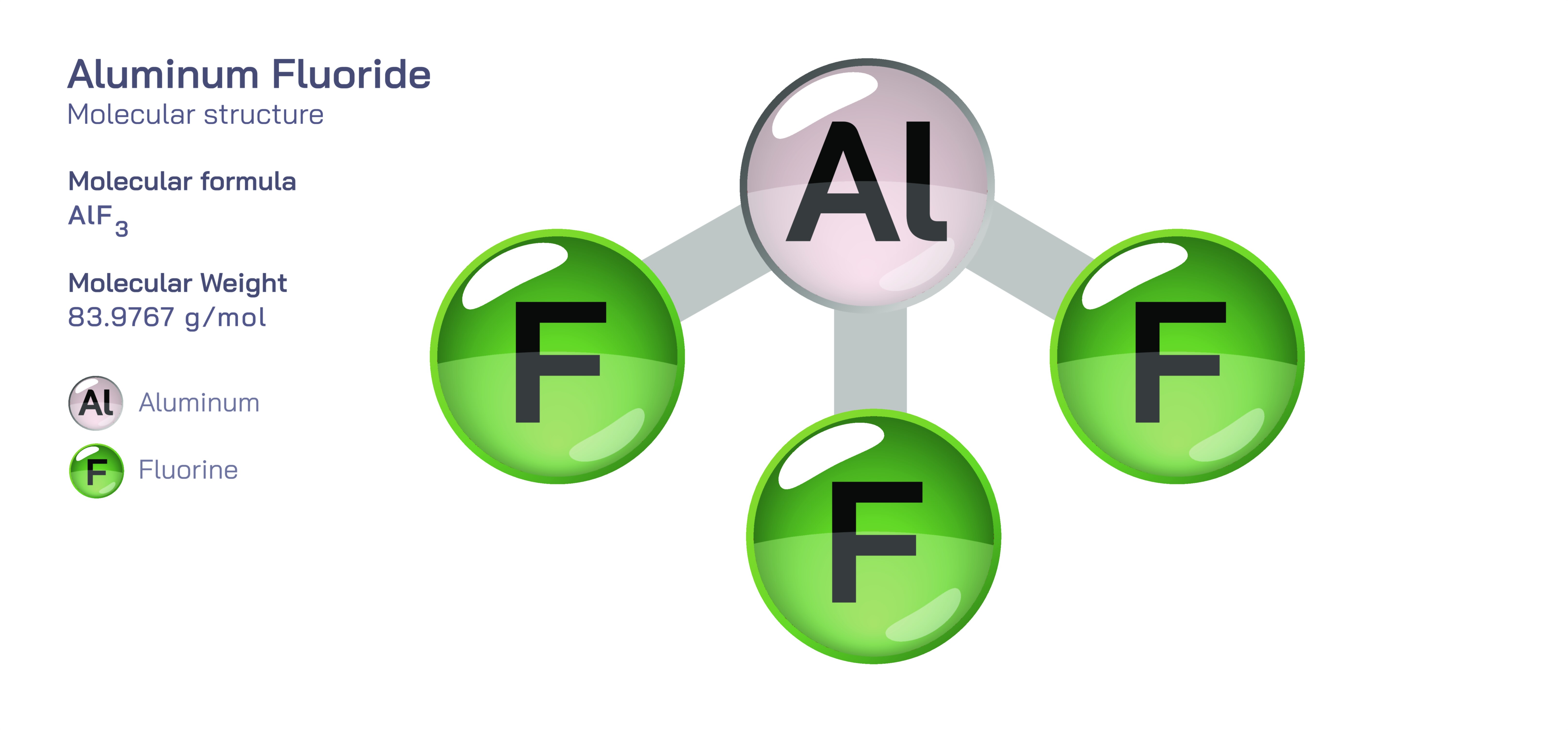
Aluminum fluoride, commonly represented by the formula AlF₃, is an essential inorganic compound deeply embedded in the chemical and metallurgical industries. Its structure, bonding characteristics, and physical resilience arise from the combination of highly electronegative fluorine with the trivalent aluminum ion. What appears at first glance to be a simple binary compound expands into a complex crystalline network, displaying strong ionic interactions and high thermal stability. Aluminum fluoride naturally occurs in several mineral forms, including rosenbergite, and is also produced synthetically for use in industrial processes that rely on the stability and reactivity of aluminum halides. Its primary role is found in the aluminum smelting industry, where it functions as a flux, lowering the melting point of alumina and enabling efficient electrolysis during the Hall–Héroult process. The overarching identity of AlF₃ is anchored in its ability to withstand extreme conditions while maintaining predictable behavior, making it indispensable in environments that demand chemical toughness, ionic conductivity, and structural rigidity. Understanding its fundamental structure reveals why aluminum fluoride remains a key compound wherever high-performance inorganic materials are needed.
Chemical Structure, Coordination, and Bonding Characteristics
The structure of aluminum fluoride is predominantly ionic, driven by the powerful attraction between the positively charged Al³⁺ cation and the negatively charged F⁻ anion. In its most stable form, AlF₃ adopts a rhombohedral or trigonal crystal lattice analogous to that of β-quartz, where each aluminum ion is octahedrally surrounded by six fluorine atoms. This octahedral coordination creates a three-dimensional network of corner-sharing AlF₆ octahedra, producing a lattice that is both dense and robust. The fluorine atoms bridge between aluminum centers, generating an extended structure rather than discrete molecular units, reflecting the compound’s purely inorganic character. Unlike aluminum chloride or aluminum bromide, which form dimeric molecules in the solid state, AlF₃ maintains a rigid, extended ionic lattice that resists cleavage or molecular rearrangement. Because fluorine is the most electronegative element, the Al–F bond exhibits strong polarization, reinforcing the ionic nature of the compound. This intense polarity contributes to aluminum fluoride’s resistance to melting, its non-volatility under normal conditions, and its ability to function effectively as a thermally stable flux. Its crystalline nature also influences how it interacts with other ions, reactants, or molten electrolytes, making its structural identity a critical factor in industrial chemistry.
Physical Properties and Their Functional Implications
Aluminum fluoride is a white or colorless crystalline solid that exhibits a high melting point, low solubility in water, and significant chemical inertness. These properties reflect the strength of the ionic lattice and the low polarizability of fluoride ions. Its melting point is exceptionally high, making it capable of withstanding temperatures involved in smelting operations and high-temperature ceramic processes. The compound’s low density and high hardness contribute to its stability, while its minimal vapor pressure ensures it remains unchanged under conditions where other halides might volatilize. Aluminum fluoride is only sparingly soluble in water, a characteristic resulting from the strong lattice energies binding the ions within the solid state. However, it becomes more reactive in molten states or in the presence of strong acids, where it can form complex fluoroaluminate species. These behaviors are instrumental in industrial electrolytic systems, where the molten environment allows controlled ion exchange and conductivity. Physically, AlF₃’s crystalline structure and thermal tolerance make it suitable for specialized ceramics, optical materials, and refractory applications. Its characteristic stability under harsh conditions reinforces its role in some of the world’s most energy-intensive industrial systems.
Reactivity and Transformation Pathways Across Chemical Systems
Despite its robust structure, aluminum fluoride does participate in specific chemical transformation pathways characteristic of metal halides. It can react with strong bases to form aluminate and fluoride-containing species, reflecting the amphoteric behavior of aluminum-centered compounds, though this amphoterism is less pronounced in fluorides than in hydroxides or oxides. When subjected to molten salt environments or strongly acidic conditions, AlF₃ participates in complex formation, generating fluoroaluminate ions such as AlF₆³⁻. These fluoroaluminate species play a central role in the Hall–Héroult process, where alumina dissolves in molten cryolite enhanced with aluminum fluoride. The presence of AlF₃ lowers the melting temperature and modifies the viscosity and electrical conductivity of the electrolyte, enabling efficient separation of metallic aluminum from oxygen. Additionally, aluminum fluoride reacts with certain metal oxides or other halides to produce mixed-metal fluorides or to facilitate halogen exchange reactions. Although relatively inert at room temperature, its reactivity under high temperatures or in molten systems highlights its versatility in modern industrial chemistry.
Industrial Significance and Technological Applications
The industrial importance of aluminum fluoride cannot be overstated. Its primary use is in aluminum production, where it serves as a critical component of the electrolytic bath that reduces alumina to pure aluminum metal. By lowering the melting point of alumina and improving the conductivity of the molten mixture, AlF₃ helps reduce the energy required for electrolysis, making large-scale aluminum production both feasible and economically viable. Beyond metallurgy, aluminum fluoride serves important functions in the manufacturing of fluorinated organic compounds, where it acts as a catalyst or fluorinating agent. Its stability and ionic characteristics make it useful in creating specialty glasses, ceramics, and optical materials where transparency, durability, and resistance to chemical corrosion are required. In certain catalytic applications, aluminum fluoride participates in reactions involving halogenation or isomerization, owing to its Lewis acidity and surface reactivity. Environmentally, its resistance to dissolution ensures that it remains relatively stable in soil and waste materials, though proper handling during industrial use prevents fluoride release. Across multiple technological fields—from aluminum smelting to high-performance ceramics—aluminum fluoride stands as a compound whose structural resilience translates into unmatched functional value.
Illustrating the Structural and Functional Characteristics of Aluminum Fluoride
Scientific illustrations of aluminum fluoride typically emphasize the octahedral coordination environment surrounding the aluminium ion, showcasing the arrangement of fluorine atoms and the extended network of AlF₆ units that define the crystalline lattice. These diagrams often depict the similarity between AlF₃’s structure and that of quartz-like mineral frameworks, highlighting how the compound’s geometry influences its hardness and thermal stability. Industrial illustrations may also show the compound’s role in the Hall–Héroult cell, visualizing how AlF₃ integrates into the molten cryolite bath, modifies electrolyte properties, and enables efficient aluminum extraction. Conceptual images also depict fluoroaluminate ions and their formation, helping explain how aluminum fluoride interacts within molten systems and participates in conductivity enhancement. Through such structural and functional illustrations, one gains deeper insight into how aluminum fluoride’s molecular organization governs its industrial power, making it a cornerstone of metallurgical and chemical technology.