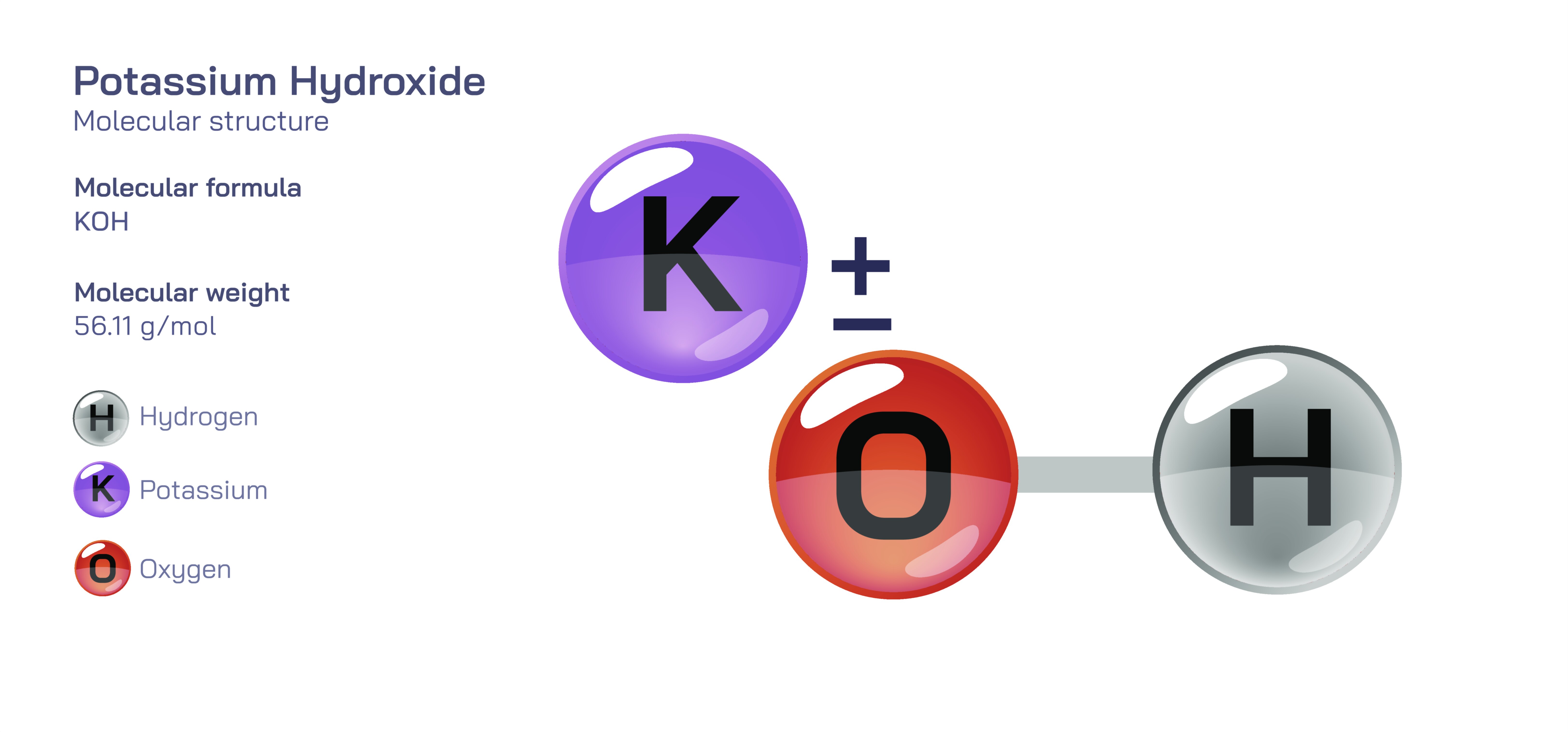
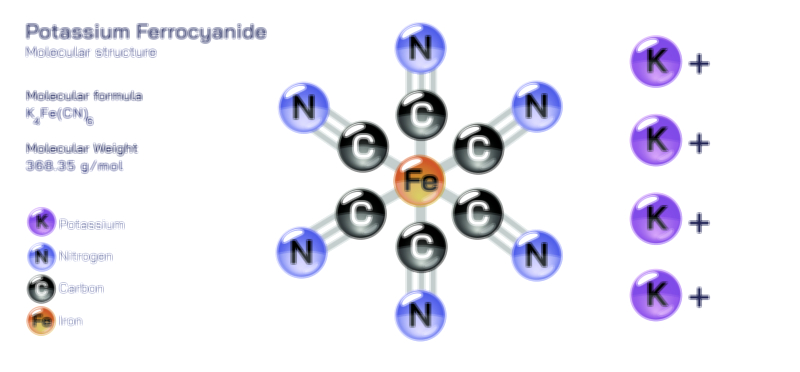
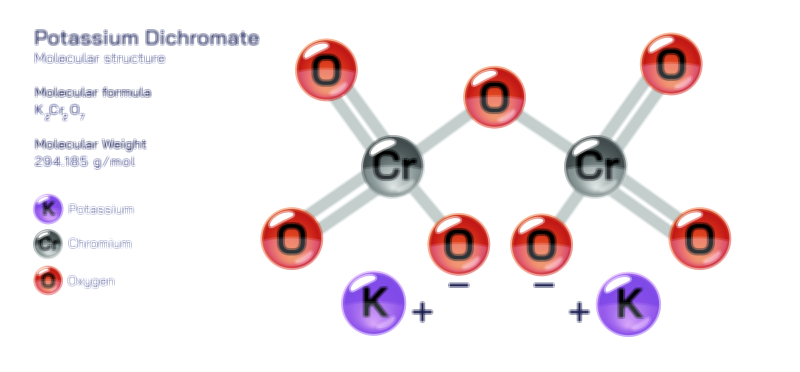
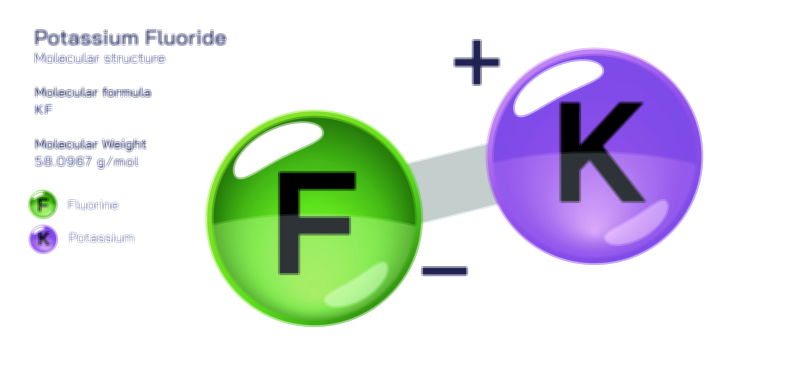
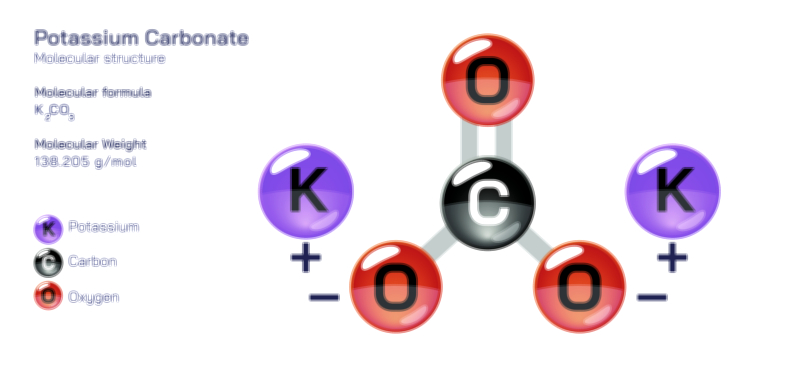
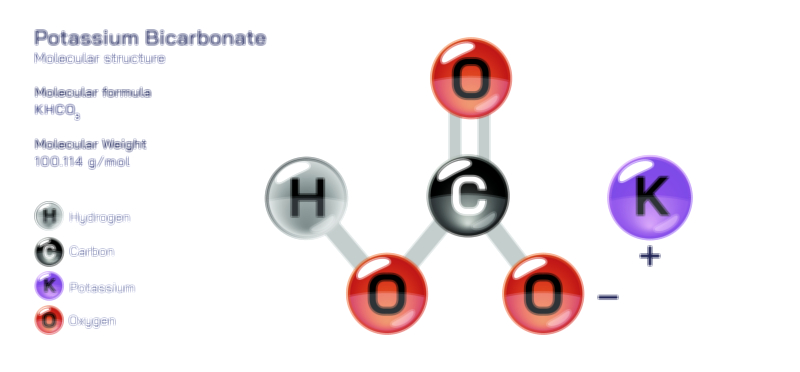
𝐏𝐨𝐭𝐚𝐬𝐬𝐢𝐮𝐦 𝐇𝐲𝐝𝐫𝐨𝐱𝐢𝐝𝐞 𝐌𝐨𝐥𝐞𝐜𝐮𝐥𝐚𝐫 𝐒𝐭𝐫𝐮𝐜𝐭𝐮𝐫𝐞
Potassium hydroxide is a highly recognizable inorganic base whose molecular structure reflects both simplicity and strong chemical reactivity. At the core of the compound is the ionic attraction between a positively charged potassium ion and a negatively charged hydroxide ion. The potassium ion originates from a neutral potassium atom that has lost one electron, while the hydroxide ion consists of one oxygen atom and one hydrogen atom joined by a covalent bond and carrying an overall negative charge. The ionic nature of the bond between potassium and hydroxide reveals that potassium hydroxide is not a molecule in the traditional covalently bonded sense, but rather an ionic compound in which independent ions are held together by electrostatic forces. In solid form, the potassium and hydroxide ions assemble into a crystal lattice, with each potassium ion surrounded by hydroxide ions in a repeating geometric pattern that extends throughout the solid. This lattice framework is responsible for the solid’s brittleness, high melting point, and strong attraction to water, demonstrating how structural arrangement at the atomic level dictates macroscopic material properties.
When potassium hydroxide dissolves in water, the crystal lattice disintegrates effortlessly because of the strong affinity between the ions and water molecules. The dissolution process separates the potassium ions and hydroxide ions completely into solution, and once released, the hydroxide ion begins to dominate the chemical behaviour of the compound. The hydroxide ion’s structure is defined by the oxygen atom bearing extra electron density and a hydrogen atom bonded to it, forming a bent arrangement in terms of electron distribution even though the unit is treated chemically as a single functional ion. This configuration allows hydroxide to act as one of the strongest bases in aqueous chemistry, readily attracting protons from water or other substances and increasing the alkalinity of the solution. The potassium ion, in contrast, remains comparatively inert, stabilizing the system electrically while leaving all reactive behaviour to the hydroxide ion. The separation of roles between ions is a direct reflection of the compound’s structural arrangement, showing that even when dissociated, the identity of potassium hydroxide persists through the behaviour of its constituent ions.
The hydroxide ion inside potassium hydroxide not only governs reactivity but also influences the compound’s physical characteristics. Because hydroxide has a strong hydrogen–oxygen dipole and an intense attraction toward other polar molecules, it causes potassium hydroxide to act as a powerful desiccant when solid. The solid strongly absorbs moisture from the air, dissolving itself in the water it captures and forming concentrated alkaline solutions. This behaviour arises not from any complex molecular network but from the simple fact that the hydroxide ion aggressively seeks stabilizing hydrogen bonds, while potassium remains content to follow as counter-ion. These tendencies also explain why potassium hydroxide is caustic and corrosive to skin and organic matter; the hydroxide ion disrupts molecular structures by removing protons and breaking down biological compounds at the molecular level. The structural basis of the hydroxide ion makes the compound chemically strong enough to saponify fats, degrade proteins, and dissolve cellulose, leading to uses in soap manufacturing, cleaning agents, and industrial chemical processing.
In molten form, potassium hydroxide is composed entirely of mobile potassium and hydroxide ions without a surrounding lattice, and this mobility allows it to conduct electricity strongly. The free movement of ions enables the compound to take part in electrolytic reactions, notably in the production of hydrogen gas. The conversion between the solid lattice, the aqueous dissolved state, and the molten ionic medium demonstrates that the identity of potassium hydroxide is tied not to a fixed molecular shape but to the interaction of ions that reorganize structurally depending on their environment. The hydroxide ion always maintains its internal structure while potassium adjusts to whatever arrangement is needed to maintain charge balance. This consistency across conditions gives potassium hydroxide the dependability required in applications ranging from laboratory chemistry and battery electrolyte preparation to industrial neutralization and biodiesel synthesis.
The structural nature of potassium hydroxide thus provides a clear picture of why it behaves with such characteristic strength and versatility. The ionic bond between potassium and hydroxide explains the compound’s crystalline solid state, solubility in water, and electrical conductivity. The bent electron distribution and proton-attracting tendency of the hydroxide ion explain its extreme alkalinity, reactivity, and corrosive power. The inert behaviour of the potassium ion explains why potassium hydroxide solutions act primarily through hydroxide rather than through metal-driven reactions. Although the formula of potassium hydroxide appears deceptively simple, the arrangement of ions and the chemistry of the hydroxide unit give the compound a profound impact across chemical reactions, industrial processing, biological interactions, and material transformation. In every context, its structure is the origin of its identity, showing how even a minimal combination of atoms can develop into a material with vast chemical power and multidimensional usefulness.