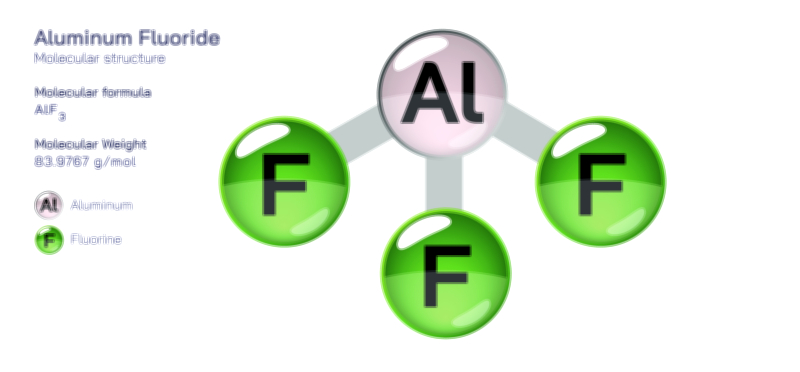
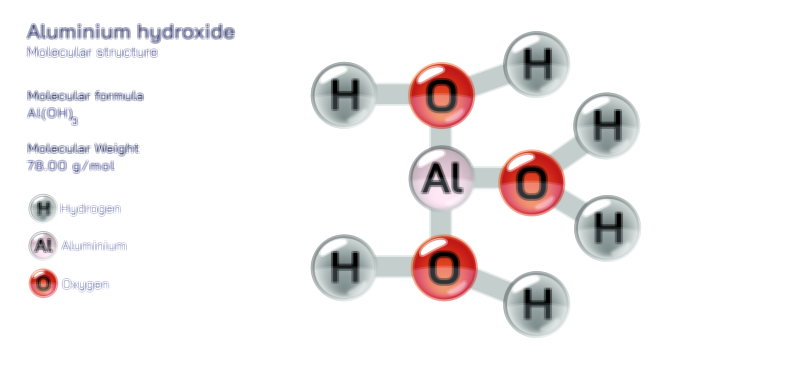
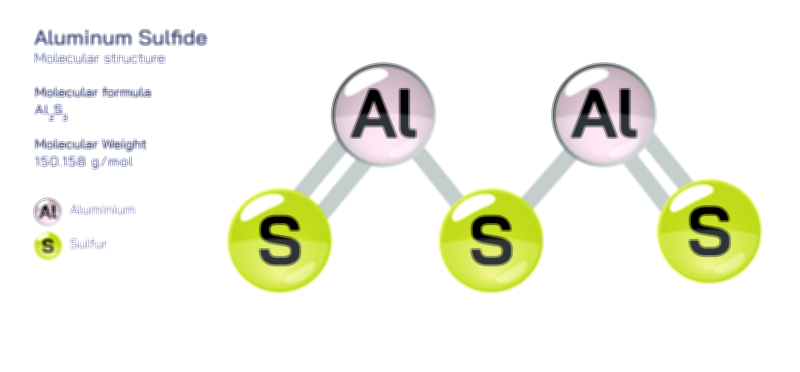
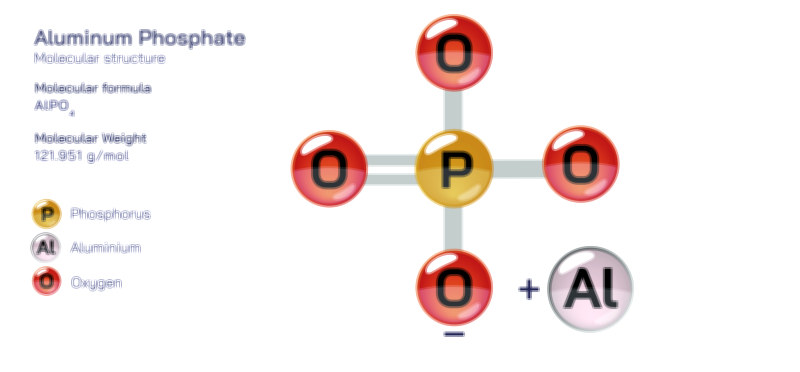
Aluminium Bromide — Chemical Structure and Inorganic Compound Illustration
Fundamental Identity and Molecular Nature of Aluminium Bromide
Aluminium bromide, commonly represented by the formula AlBr₃, is an important inorganic compound that plays a significant role in chemical synthesis, catalysis, and various industrial reactions. Although simple in composition—consisting of aluminium atoms bonded to bromine atoms—its structural behavior is far richer than its formula suggests. Like other aluminium halides, aluminium bromide exhibits notable differences between its solid-state and gaseous forms. In the vapor phase, the molecule exists primarily as AlBr₃ monomers, but in the solid and liquid state it prefers dimerization, forming the structure Al₂Br₆. This occurs because aluminium, in its +3 oxidation state, has a strong tendency to achieve a more stable electron configuration through coordination with electron-rich species. The structure of aluminium bromide therefore reflects the nuanced balance between Lewis acidity, halide bonding, and the geometric constraints that define how this compound behaves under varying environmental conditions. Its strong reactivity, particularly as a Lewis acid, makes it highly valuable for electrophilic aromatic substitution reactions, Friedel–Crafts processes, and a variety of halogenation and alkylation pathways that lie at the heart of synthetic organic chemistry.
Chemical Structure, Bonding, and Molecular Geometry
The structure of aluminium bromide illustrates classical principles of electron deficiency and compensating molecular interactions. In the solid-state dimer Al₂Br₆, two aluminium atoms are bridged by two bromine atoms, forming a configuration where each aluminium achieves coordination with four bromine atoms: two terminal bromides and two bridging bromides. This arrangement emerges due to the strong Lewis acidity of the aluminium center, which lacks sufficient electrons to maintain stability in a simple trigonal planar monomeric form. The bridging bromine atoms donate electron density to the aluminium atoms, helping fill this electronic gap. As a result, the dimer adopts a configuration reminiscent of Al₂Cl₆ but with bond lengths and degrees of electron sharing influenced by bromine’s larger atomic radius and lower electronegativity compared to chlorine. In gaseous form, where intermolecular forces weaken, the compound shifts back to trigonal planar AlBr₃ molecules with sp² hybridization. These molecules maintain a strong dipole moment due to the polar nature of the Al–Br bond, and this polarity plays a significant role in the compound's interaction with solvents, reactants, and atmospheric humidity.
Physical Properties and Their Significance in Chemical Processes
Aluminium bromide appears as a white to yellowish solid that is highly hygroscopic, meaning it readily absorbs moisture from the air. This tendency arises from the strong Lewis acidity of the aluminium center, which attracts water molecules and reacts vigorously to form hydrated products or hydrolyzed species such as aluminium hydroxide and hydrogen bromide. When exposed to humid air, aluminium bromide can emit fumes as hydrolysis proceeds, making careful handling necessary. The compound melts to a liquid that remains predominantly dimeric in moderate temperatures, transitioning to monomeric vapor only at higher temperatures. Its solubility behavior reflects its ionic-covalent hybrid nature: it dissolves well in nonpolar and polar organic solvents but reacts violently with water due to rapid hydrolysis. In solvents such as nitrobenzene, carbon disulfide, or dichloromethane, aluminium bromide functions as a powerful catalytic agent because the solvent environment supports the maintenance of its Lewis-acidic character. Its boiling and melting points reflect the strength of the dimeric bonds in the solid state, with the compound becoming increasingly reactive as temperature rises.
Reactivity, Lewis Acidity, and Transformation Pathways
Aluminium bromide’s reactivity arises largely from its electron-deficient aluminium center, which makes it a potent Lewis acid capable of accepting electron pairs from a broad range of donors. This property forms the foundation of its role in Friedel–Crafts reactions, where it activates halides, alkyl groups, or acyl groups for electrophilic aromatic substitution. In these reactions, aluminium bromide coordinates with the halogen or acylating agent, polarizing the bond and increasing the formation of the positively charged electrophile. This allows aromatic rings to undergo substitution more readily, forming carbon–carbon bonds that are central to industrial synthesis of dyes, fragrances, pharmaceuticals, and polymer precursors. Aluminium bromide also participates in halogen exchange, dehydration, and condensation reactions, enabling the construction or modification of various organic molecules. When reacting with water, it undergoes hydrolysis to form aluminium hydroxide and hydrogen bromide, demonstrating its high affinity for moisture. Combined with its strong coordination ability, this reactivity allows aluminium bromide to form adducts with ethers, amines, phosphines, and other electron-rich molecules. These adducts often play important roles in specialized catalytic cycles in both laboratory and industrial chemistry.
Industrial, Environmental, and Practical Applications
Industrially, aluminium bromide is indispensable in processes that require strong Lewis acids. Its catalytic influence supports the synthesis of complex aromatic compounds, which serve as intermediates in pharmaceuticals, agrochemicals, polymers, and fragrances. Its ability to catalyze isomerization and halogenation reactions provides manufacturers with tools to manipulate molecular structures with precision. In some speciality processes, aluminium bromide assists in the formation of organoaluminium compounds, which are valuable in polymer chemistry and metallorganic synthesis. Environmental considerations focus largely on its reactivity with water and moisture, as improper handling can lead to the release of corrosive hydrogen bromide. Proper containment, dry storage, and controlled reactor environments ensure safe and effective use. In research settings, aluminium bromide serves as a reference compound for studying halide influence on aluminium coordination chemistry, helping chemists understand how atomic size and electronegativity shape structural and catalytic properties across the series of aluminium halides.
Illustrating the Molecular Structure and Compound Behavior of Aluminium Bromide
Illustrations of aluminium bromide typically highlight the dimeric Al₂Br₆ structure, emphasizing the two bridging bromine atoms that connect the aluminium centers. These depictions show the angular geometry around each aluminium, the bond lengths between terminal and bridging bromides, and the electron-deficient nature of the aluminium site. Visual diagrams often contrast the trigonal planar form of gaseous AlBr₃ with the bridged dimer of the solid state, helping clarify how intermolecular forces influence structural transitions. Reaction pathway illustrations may highlight its interaction with aromatic compounds during catalytic activation, showing how aluminium bromide polarizes bonds and facilitates electrophile formation. Through these structural and mechanistic representations, one can appreciate how the chemical architecture of aluminium bromide determines its widespread utility, high reactivity, and essential role in both fundamental inorganic chemistry and applied industrial synthesis.