ØÉüØÉÜØɽØÉóØÉ«ØÉ” ØÉŹØÉóØÉŁØɽØÉÜØÉŁØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉÜØɦØÉØ ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×.
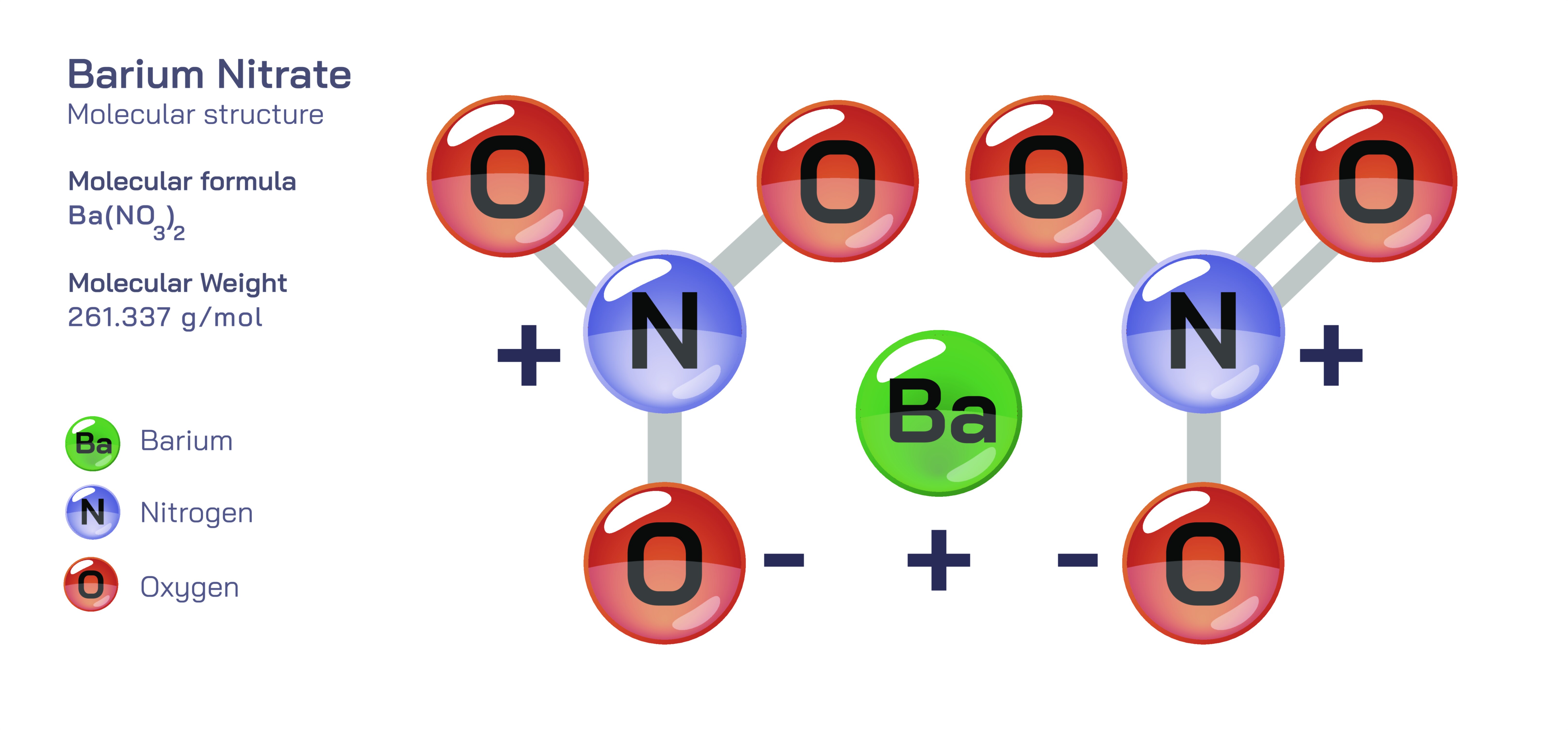
Barium nitrate is an inorganic compound widely recognised for its crystalline stability, high solubility, strong oxidizing capability, and significant industrial value, especially in pyrotechnics and laboratory chemistry. Known chemically as Ba(NOŌéā)Ōéé, it is composed of a divalent barium cation (Ba┬▓Ōü║) surrounded by two nitrate anions (NOŌéāŌü╗). At first glance, its formula suggests a simple ionic composition, yet the internal structural organization reveals a complex lattice shaped by electrostatic attraction and resonance-stabilized nitrate groups. The behaviour of barium nitrateŌĆöits combustion properties, reactivity in solutions, thermal decomposition, and toxicityŌĆöcan all be traced back to the structural characteristics that define how the ions are arranged and how they interact chemically with other substances.
At the molecular level, barium nitrate is not a covalent molecule in the traditional sense but an ionic compound composed of independent Ba┬▓Ōü║ and NOŌéāŌü╗ ions. The barium ion forms the positive core, and each nitrate ion remains structurally intact, bound through ionic attraction rather than shared electrons. The nitrate ion itself consists of one nitrogen atom bonded to three oxygen atoms in a trigonal planar arrangement. The true structure of the nitrate ion is not a single fixed configuration but a resonance-stabilized arrangement in which the double bond between nitrogen and oxygen is delocalized, meaning all three nitrogenŌĆōoxygen bonds are equivalent and share partial double-bond character. This delocalization gives the nitrate ion high stability and makes it an efficient carrier of oxidizing oxygen in chemical reactions. When combined with the Ba┬▓Ōü║ ion, the two NOŌéāŌü╗ ions position themselves symmetrically in the lattice around the cation, minimizing total lattice energy through optimized ion packing. This arrangement gives barium nitrate a well-defined cubic crystalline form that appears colourless or white under ambient conditions.
The structural identity of barium nitrate strongly influences its solubility characteristics. Unlike many other salts of barium that are poorly soluble or practically insoluble in waterŌĆösuch as barium sulfate and barium carbonateŌĆöbarium nitrate dissolves completely in water because the hydration energy released by interaction with water molecules is high enough to break the internal ionic lattice. When Ba(NOŌéā)Ōéé dissolves, the compound dissociates into hydrated Ba┬▓Ōü║ cations and hydrated NOŌéāŌü╗ anions that behave independently in solution. The separated ions contribute to strong electrical conductivity because the charged particles move freely throughout the solvent. This dissociation is important in analytical and inorganic chemistry because it allows barium nitrate to act as a source of barium ions for precipitation reactions and as a provider of nitrate ions for redox chemistry. The barium ions released from the solution can react with sulfate or carbonate ions to form characteristic insoluble precipitates useful for ion identification in chemical tests. Meanwhile, the nitrate ions can serve as oxidizing agents or participate in a variety of coordination and acidŌĆōbase reactions.
One of the most notable consequences of the nitrate ionŌĆÖs structural characteristics is the oxidizing ability of barium nitrate. Because the nitrate group stores oxygen in a chemically available form, it can release oxygen during high-temperature or combustion reactions. This property explains the prominent role of barium nitrate in pyrotechnics. When mixed with combustible materials such as powdered aluminum, magnesium, sulfur, or charcoal, barium nitrate supplies oxygen that intensifies the burning reaction. During ignition, the nitrate breaks down, producing gases containing nitrogen oxides and releasing oxygen, which supports the rapid combustion of fuel. Meanwhile, the barium component emits a vivid green flame when heated because barium ions emit visible light at characteristic wavelengths. This unique flame colour makes barium nitrate a critical ingredient in green fireworks, signal flares, tracer ammunition, and pyrotechnic displays. The green illumination is not a pigment but an optical effect produced by electrons in barium atoms transitioning between energy levels under heat energy. Thus, both structural components of Ba(NOŌéā)ŌééŌĆöthe oxidizing nitrate and the colour-emitting bariumŌĆöwork together to create the characteristic green pyrotechnic flame.
The thermal decomposition of barium nitrate illustrates another dimension of its structural behaviour. When heated to sufficiently high temperatures, the compound breaks down into barium oxide, oxygen, and nitrogen dioxide gases. This decomposition releases oxygen into the environment, a hallmark of oxidizing salts. The formation of barium oxide from barium nitrate during decomposition reflects the inherent tendency of the barium cation to form stable oxide compounds when nitric oxygen bonds break down. Many industrial and military initiator compositions rely on this decomposition behaviour to generate heat and gas rapidly. The strong oxidizing ability also requires careful handling, since the compound can intensify combustion if it comes into contact with organic matter or flammable chemicals.
Because barium nitrate contains Ba┬▓Ōü║, a biologically active and potentially toxic ion, the compound must be managed with caution. Soluble barium salts release their cations into the bloodstream and interfere with potassium-channel function in muscle and nerve tissues. This disruption can cause muscle spasms, cardiac irregularities, gastrointestinal distress, and other symptoms of barium poisoning. However, the toxicity originates not from the nitrate ionŌĆönitrate salts of alkali metals are commonly harmless in moderate dosesŌĆöbut from the free barium ion liberated from the compound in aqueous environments. The same feature that makes barium nitrate useful in chemical testing and pyrotechnicsŌĆöits ability to dissolve and fully ionizeŌĆömakes it hazardous if ingested or improperly handled. Therefore, unlike insoluble barium sulfate, which passes harmlessly through the digestive system and is used medically in imaging, barium nitrate must never be consumed and must be stored to prevent contamination of food or water.
In laboratory and manufacturing contexts, barium nitrate is valued for its predictable chemical reactivity and high purity potential. It functions as a raw material in the synthesis of other barium-based compounds including barium oxide, barium carbonate, and barium chromate. In ceramics and specialty glass fabrication, barium nitrate contributes to high-density materials with enhanced thermal and electrical characteristics. In vacuum tube technology and electron device engineering, barium compounds derived from barium nitrate play a role in cathode coatings and emissive surfaces. These applications depend on the consistent release of Ba┬▓Ōü║ ions from solution or through thermal decomposition, reinforcing that industrial roles are inseparable from molecular structure.
Environmental and safety considerations linked to barium nitrate reflect once again the direct influence of chemical structure on real-world effects. Because the compound dissolves so readily, accidental release into soil or water can introduce mobile Ba┬▓Ōü║ ions into ecosystems, where they can enter food chains. Regulatory handling protocols for barium nitrate therefore stress containment, controlled disposal, and proper personal protection in settings where airborne dust or water contamination is possible. The nitrate component itself, though not the primary toxic agent here, can contribute to nutrient imbalance in ecosystems if present in large quantities, emphasizing the need for responsible use.
Every behavioural trait of barium nitrateŌĆöfrom its crystal shape to its flame colour, from its solubility to its reactivityŌĆöcan be traced to one unifying principle: the structure and arrangement of its ions. The Ba┬▓Ōü║ cation and NOŌéāŌü╗ anions do not simply coexist; they organize into a stable yet dissociable lattice whose balance between electrostatic strength and hydration accessibility gives the compound chemical versatility. The resonance-stabilized nitrate groups enable oxygen release, while the barium ions generate characteristic emission spectra and reactive precipitates. These characteristics make barium nitrate an indispensable material in science and industry, but they also demonstrate that utility and hazard are often rooted in the same structural features.
Understanding barium nitrate is therefore about more than memorizing a formula. It requires appreciating how ionic geometry, resonance stabilization, thermal decomposition pathways, solubility behaviour, and biological interactions arise from atomic arrangement. Barium nitrate is a clear example of how structure determines behaviour, how behaviour determines applications, and how those applications shape the value and risk of a chemical compound in the real world.