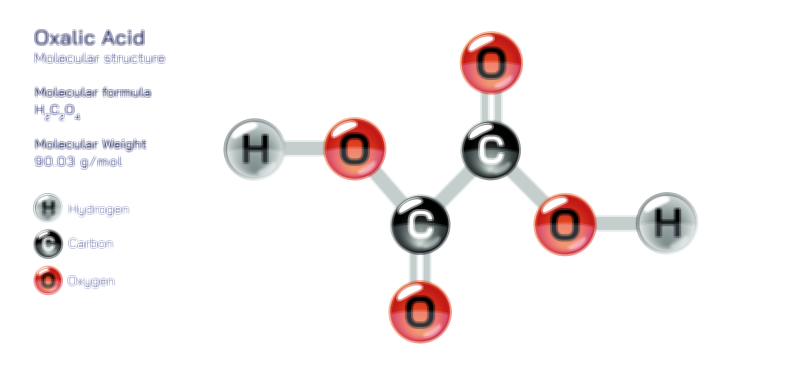
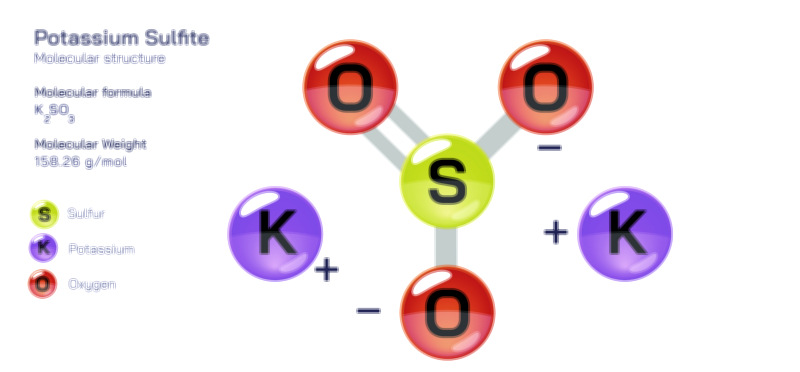
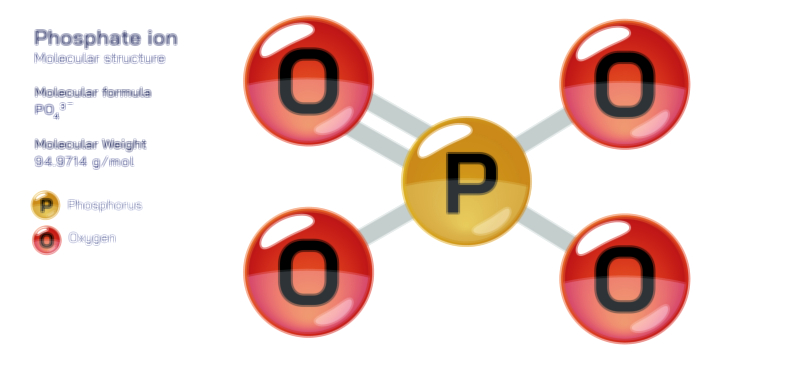
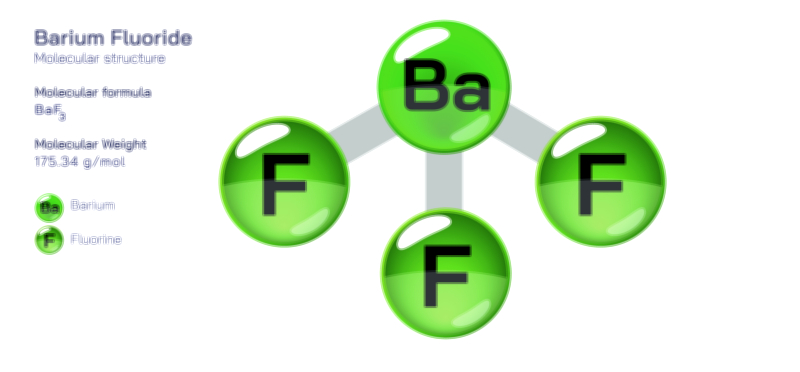
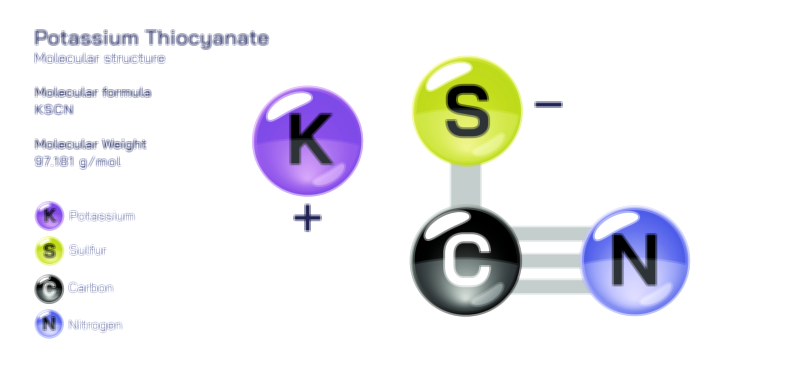
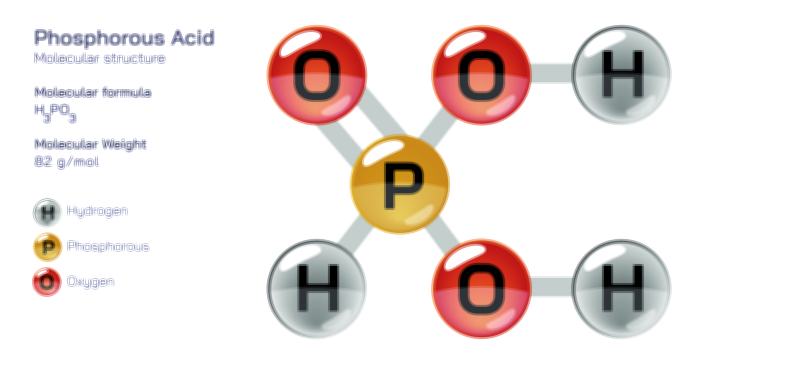
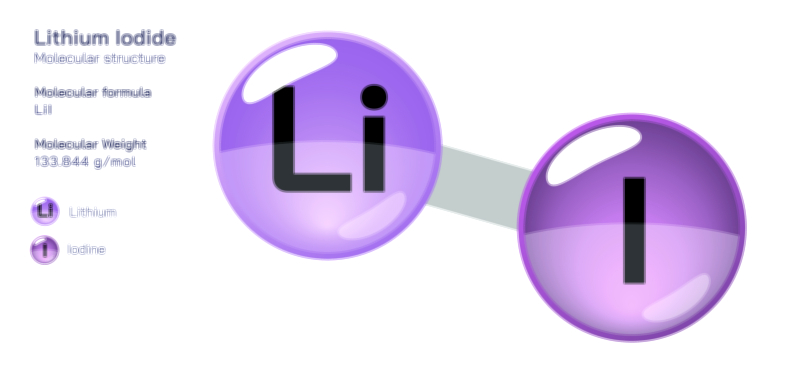
ØÉüØÉÜØɽØÉóØÉ«ØÉ” ØÉÆØÉ«ØÉźØɤØÉÜØÉŁØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉÜØɦØÉØ ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×.
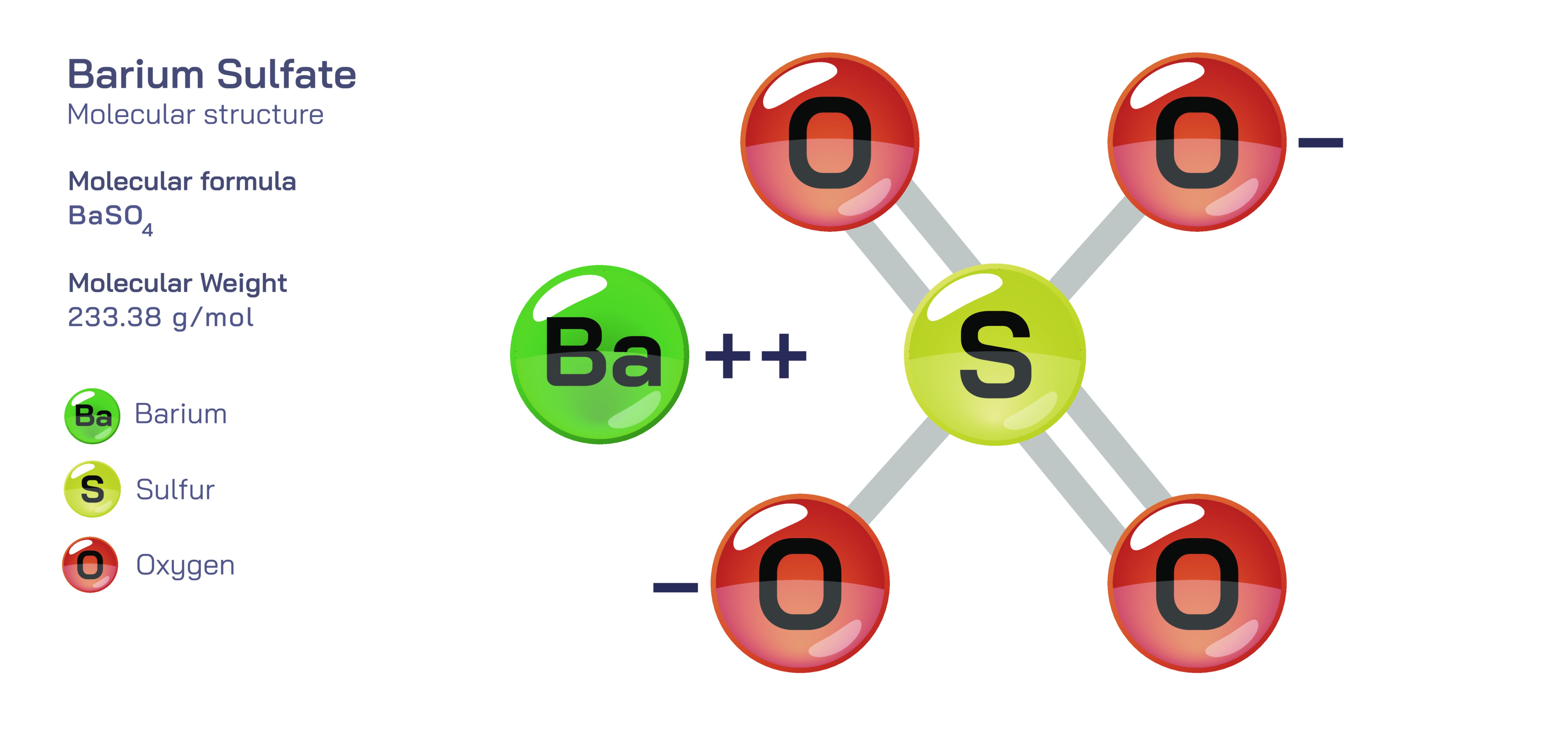
Barium sulfate is an inorganic compound represented chemically by the formula BaSOŌéä, consisting of a barium cation (Ba┬▓Ōü║) and a sulfate anion (SOŌéä┬▓Ōü╗). Although the formula appears simple, the compoundŌĆÖs structure reveals a complex ionic network where ions are held together by strong electrostatic forces. Barium sulfate is particularly well known for its extremely low solubility in water and its chemical inertness, traits that arise directly from the internal arrangement of the Ba┬▓Ōü║ and SOŌéä┬▓Ōü╗ ions. This structural stability gives barium sulfate properties that sharply distinguish it from other barium compounds. While many soluble barium salts release Ba┬▓Ōü║ ions and therefore pose significant toxicity risks, barium sulfateŌĆÖs low solubility means that its ions remain locked within the solid lattice and do not dissociate in the digestive system or bloodstream. As a result, BaSOŌéä is safe for medical use and industrial applications, despite the fact that free barium ions are highly toxic. The stability of BaSOŌéä is therefore a structural phenomenon, showing that safety or danger is dictated not only by the chemical elements present but by how they are bonded and arranged.
To understand the behaviour and properties of barium sulfate, one must look at the sulfate anion. The SOŌéä┬▓Ōü╗ ion contains a central sulfur atom surrounded by four oxygen atoms arranged in a tetrahedral geometry. The SŌĆōO bonds exhibit resonance, meaning electron density is distributed evenly across the oxygen atoms, making the molecule highly symmetrical and energy-stable. When sulfate pairs with Ba┬▓Ōü║, the resulting electrostatic attraction between the doubly positive metal ion and the doubly negative sulfate anion creates a strongly bound three-dimensional lattice. This lattice is dense, tightly packed and energetically unfavourable to separate. Because breaking the lattice requires more energy than can be supplied from hydration interactions in water, barium sulfate does not dissolve. Water molecules cannot surround or separate the ions effectively, so the compound remains in its crystalline form even under high temperature and pressure. The insolubility property is not an accidental feature; it is a direct consequence of the balanced ionic radii and charging symmetry shared between Ba┬▓Ōü║ and SOŌéä┬▓Ōü╗.
The physical crystal structure of barium sulfate is orthorhombic, producing well-formed, often opaque or white crystals that occur naturally as the mineral barite (or baryte). The density of barite is notably high and is one of the reasons this mineral is used in drilling operations and heavy industrial processes. The structural packing of barium and sulfate ions accounts for the mineralŌĆÖs weight, reflecting how atomic arrangement determines macroscopic properties. Finely ground BaSOŌéä is used as a weighting agent in drilling fluids in the oil and gas sector because its mass counteracts upward pressure from underground gases and fluids. The crystals themselves do not react readily with acids or bases under normal conditions and resist dissolution even in strong acids, except under highly specialized reactive conditions involving hot concentrated sulfuric acid or molten alkalis. This stability again illustrates how the lattice shields the ions inside from disruption.
Despite being chemically inert, barium sulfate has tremendous importance in medicine because its solubility characteristics eliminate toxicity concerns. Since BaSOŌéä does not release free Ba┬▓Ōü║ ions in the gastrointestinal tract, it is widely used as a radiocontrast agent for imaging studies of the digestive system. When suspended in water to form a drinkable or injectable contrast mixture, it coats the internal walls of the stomach and intestines. Because barium atoms have a high atomic number, they strongly absorb X-rays, allowing physicians to view structural abnormalities on radiographs or CT scans with exceptional clarity. This remarkable medical usage would be impossible with any soluble barium compound. Structural insolubility is key: the crystalline form passes safely through the body without dissolving, reacting or entering biological tissues. This shows how dramatically chemical safety can reverse based solely on ionic arrangementŌĆöelements that are toxic in one compound become harmless in another.
Barium sulfate also plays an important role in material science and industrial chemistry. Owing to its whiteness, opacity, and fine particle characteristics, BaSOŌéä is used as a pigment extender and filler in paints, plastics, coatings, ceramics and rubber compounds. Its high refractive index contributes brightness and smoothness to surfaces without interfering chemically with other pigments. In plastics manufacturing, BaSOŌéä increases density, stiffness and heat resistance. In automotive coatings, it contributes to scratch resistance and gloss retention. In paper production, BaSOŌéä enhances printing quality and opacity. In each of these applications, the compound is not functioning chemically but physically, based on its structural hardness, density, particle size and light-reflecting properties. Thus, even when BaSOŌéä is inert in chemical reactions, it still exerts strong influence in industrial formulations thanks to its inherent physical traits.
The structural stability of barium sulfate also informs geochemical processes. In certain natural environments, BaSOŌéä forms scale deposits in oil and gas pipelines and geothermal systems. Because the compound is so insoluble, once precipitation begins under the right ionic conditionsŌĆöincluding high sulfate concentration and presence of dissolved bariumŌĆöit forms hard deposits that are extremely resistant to removal. Industrial cleaning often requires mechanical removal or dissolution by chelating agents rather than conventional acids. The slow solubility kinetics of BaSOŌéä reveal that although the ions are tightly bound once crystallized, formation conditions in natural systems still play a vital role. This highlights the dual nature of insoluble compounds: they may be stable under most conditions, yet their formation can be triggered suddenly in highly concentrated ionic environments.
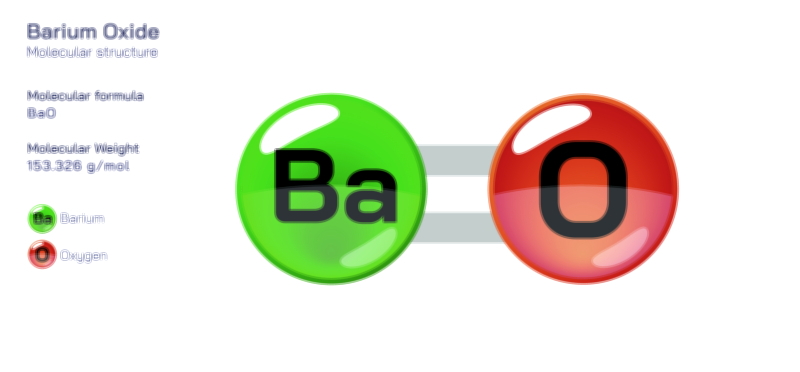
Thermal behaviour also aligns with the structure of BaSOŌéä. When heated, it does not melt easily but instead decomposes only under extreme temperatures above 1,580 ┬░C, yielding barium oxide (BaO) and sulfur trioxide (SOŌéā). This breakdown demonstrates that BaSOŌéä does not simply soften and flow; it resists thermal disturbance until high-energy input forces the ionic lattice apart. Its decomposition products reflect the elemental nature of the compoundŌĆöBaO is strongly basic and reactive, while SOŌéā is acidic and volatile. These fragments further reinforce that the stability of BaSOŌéä lies in the ionic bond between Ba┬▓Ōü║ and SOŌéä┬▓Ōü╗; once separated by intense heat, their inherent individual reactivities become exposed.
Toxicological discussions of barium sulfate cannot ignore the context of solubility. Many barium compounds are health hazards because soluble Ba┬▓Ōü║ ions disrupt potassium signalling in nerves and muscles. Barium sulfate, however, is one of the very few forms of barium that is considered biologically inert because its ions remain locked within the crystalline matrix. It does not dissolve in stomach acid or react with biological molecules. The compound can be ingested and excreted without systemic absorption, explaining its medical approval when other barium compounds would be dangerous. The example of BaSOŌéä teaches an important concept in chemistry and toxicology: toxicity is not solely determined by composition but by bioavailability. Structural insolubility protects the body from accessing the free ions.
Ultimately, barium sulfate shows how the molecular structure of an ionic solid governs every characteristic of the compoundŌĆöits safety, reactivity, solubility, industrial use, medical value, and physical appearance. The pairing of the large Ba┬▓Ōü║ cation and the tetrahedral SOŌéä┬▓Ōü╗ anion yields a lattice that is extremely resistant to disruption, enabling chemical inertness in most environments. At the same time, the high atomic weight of barium and the optical scattering of the crystalline material give BaSOŌéä applications across medicine, imaging, manufacturing and materials engineering. This compound demonstrates a fundamental lesson of chemistry: the behaviour of a substance cannot be predicted from elemental identity alone. Instead, it emerges from the structural arrangement of atoms, the strength of their interactions and the symmetry of the resulting crystalline architecture. Barium sulfate stands as an enduring illustration of how structure defines function and how the microscopic arrangement of ions determines the macroscopic impact of a compound in science, technology and medicine.