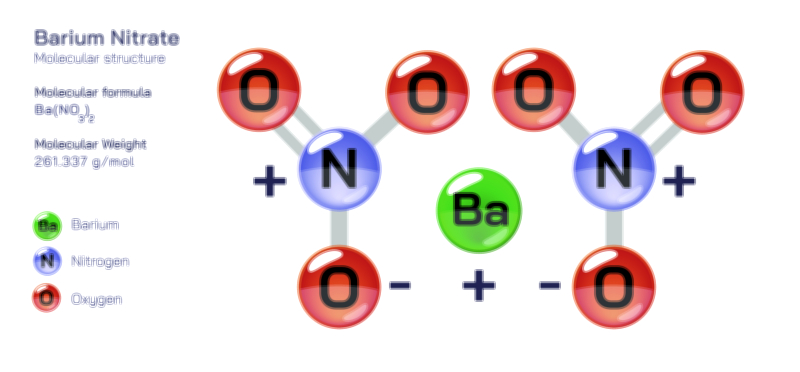
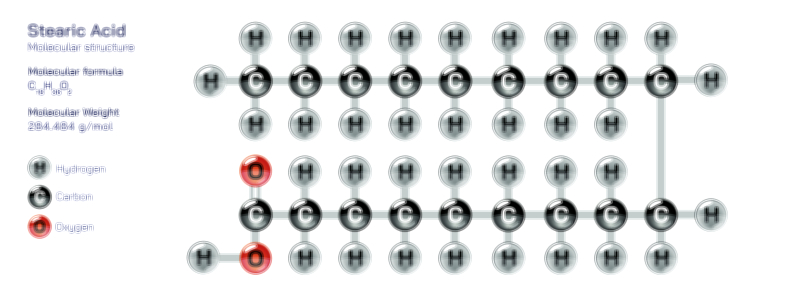
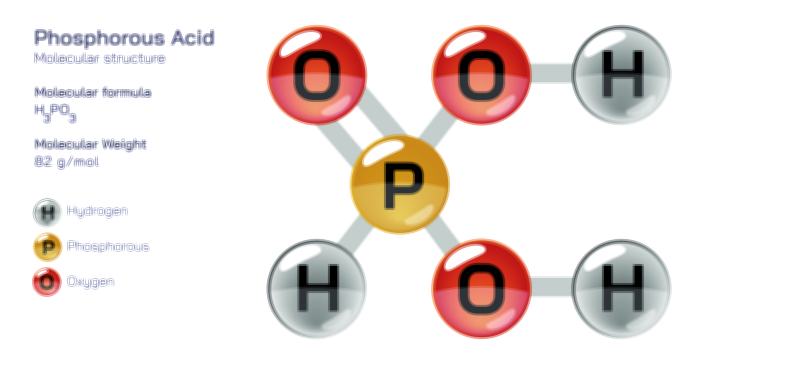
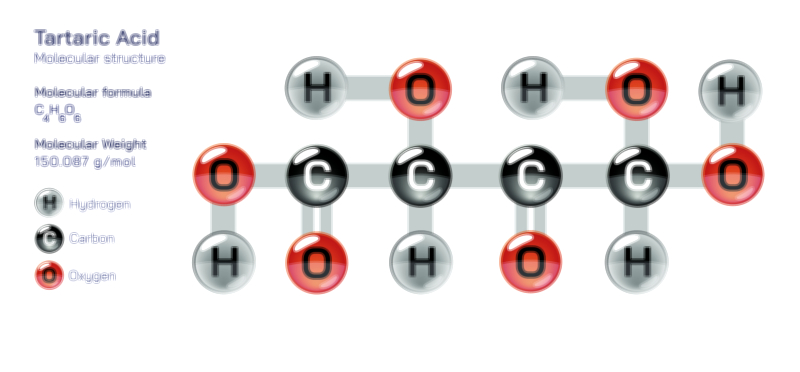
ØÉÄØÉ▒ØÉÜØÉźØÉóØÉ£ ØÉĆØÉ£ØÉóØÉØ ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉÜØɦØÉØ ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ× ØÉäØÉ▒ØÉ®ØÉźØÉÜØɦØÉÜØÉŁØÉ©ØɽØÉ▓ ØÉÆØÉ«ØÉ”ØÉ”ØÉÜØɽØÉ▓.
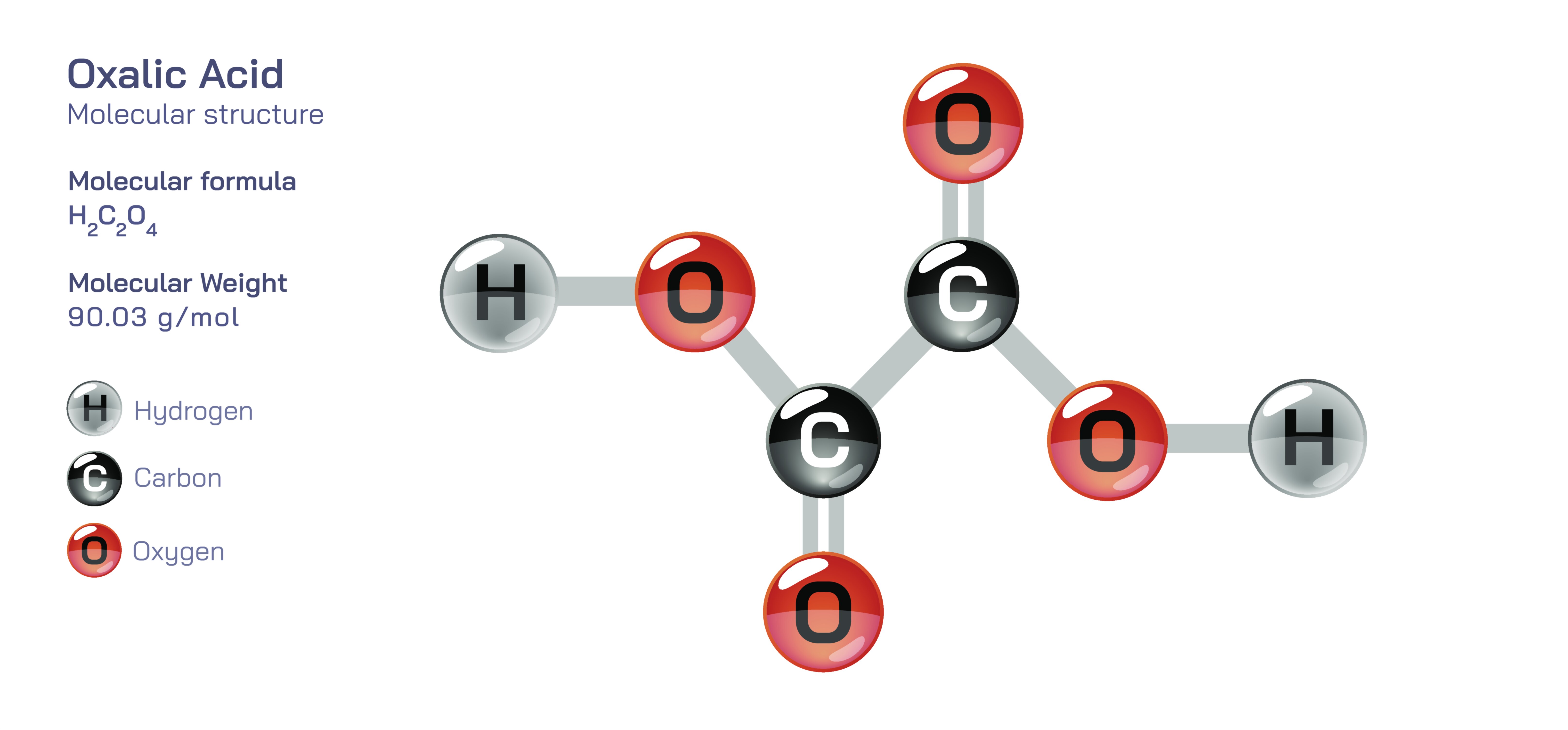
Oxalic acid is an organic chemical compound known for its distinct strongly acidic nature and its unique molecular structure that makes it one of the simplest representatives of the dicarboxylic acids. It occurs naturally in many plants, such as spinach, beet leaves, rhubarb, almonds, cocoa and tea, and it also forms in metabolic and biochemical pathways inside living organisms. Despite its natural origin, oxalic acid has intense reactivity and biological importance, and its structure explains both its usefulness and its risks. With the molecular formula CŌééHŌééOŌéä in its pure form and (COOH)Ōéé in its common notation, oxalic acid consists of two carboxyl groups bonded directly to one another. This arrangement makes the molecule compact, highly polar and extremely capable of donating protons, giving it strong acidic behavior. The pair of carboxyl groups also allow it to bind metallic ions, making it an effective chelating agent. Therefore, oxalic acid serves simultaneously as a natural metabolic product, an industrial chemical, a laboratory reagent andŌĆöwhen uncontrolledŌĆöa source of biological toxicity.
The molecular structure of oxalic acid lies at the center of its chemical behavior. At the structural level, the compound consists of two carbon atoms bonded to each other, and each carbon atom is also bonded to two oxygen atomsŌĆöone through a double bond and one through a single bond that forms a hydroxyl group. Thus, each carbon participates in a carboxyl group, and the molecule exists chemically as HOOCŌĆōCOOH. These two neighboring carboxyl groups enable the release of two acidic hydrogen ions, classifying oxalic acid as a diprotic acid. The electron-withdrawing effect of one carboxyl group strengthens the acidity of the other, making oxalic acid far stronger than many other organic acids such as acetic acid. This powerful proton-donating ability explains why oxalic acid dissolves minerals, reacts vigorously with metal carbonates and forms crystalline salts easily. Because of its polar nature, oxalic acid molecules also develop hydrogen bonding networks, giving the solid form its crystalline character and high solubility in hot water.
The structural composition of oxalic acid also determines its role as an excellent chelating agent. The negatively charged oxygens of the deprotonated carboxyl groups have strong affinity for metal ions. When oxalic acid loses its acidic hydrogens, the resulting oxalate ion (CŌééOŌéä┬▓Ōü╗) forms stable complexes with metals such as calcium, iron, magnesium, copper and manganese. This chelation capacity leads to both beneficial industrial uses and important biochemical effects. In cleaning and metal treatment processes, oxalic acid removes rust and metal stains by binding to iron and other metal ions and converting them into soluble oxalate salts. This ability is valuable in textile cleaning, marble polishing, and stain removal for wood and tiles. In chemistry laboratories, oxalic acid appears as a reducing agent in redox reactions, particularly those involving iron, manganese and chromium compounds, because its structural composition allows electron donation in well-defined reaction pathways. The moleculeŌĆÖs simplicity makes these reactions predictable and precise.
However, the same structural ability to tightly hold metal ions generates risks when oxalic acid interacts with biological systems. In humans and other animals, oxalic acid binds strongly to calcium ions, forming calcium oxalate, an insoluble compound that can crystallize inside tissues. In the kidneys, accumulation of calcium oxalate leads to kidney stones, making oxalic acid and oxalate metabolism medically significant. Some individuals produce oxalate internally through endogenous processes, and many foods contain oxalic acid naturally, meaning that both diet and metabolism contribute to oxalate buildup. While the presence of oxalate in the body is normal at low levels, high concentrations create conditions for crystal formation. The sharp microscopic structures of calcium oxalate crystals irritate tissue and cause pain during kidney stone passage. The structural concept of strong metal chelation, therefore, explains why oxalic acid is both a natural biochemical compound and a contributor to pathological mineral deposits.
The molecular structure also supports the formation of hydrates. In its solid state, oxalic acid often exists as oxalic acid dihydrate (HŌééCŌééOŌéä┬Ę2HŌééO), where each molecule becomes linked to water through hydrogen bonding. This crystalline form is thermally stable at room temperature and widely used in industrial and laboratory applications. Heating the dihydrate releases water and produces anhydrous oxalic acid, which then decomposes at higher temperatures to carbon monoxide, carbon dioxide and water. These decomposition pathways show how the electron-rich oxygen environment around the carboxyl groups facilitates breakdown under heat with the release of gaseous byproducts, linking the compound to combustion chemistry and industrial processing.
Oxalic acid plays a significant role in nature as part of the metabolic cycles of plants, fungi and bacteria. Many plants synthesize oxalic acid as a defense system because the compound can immobilize harmful metal ions in the soil and protect roots from toxicity. Inside the plant body, oxalic acid helps regulate calcium levels and maintain ionic balance. In fungi and lichen ecosystems, oxalic acid participates in mineral weathering by dissolving rock surfaces through chelating mechanisms. These ecological functions reveal that oxalic acid is more than a harmful or reactive compoundŌĆöit is a strategic molecule that influences environmental chemistry and nutrient cycles.
In chemical industries, oxalic acid is indispensable in textile purification, removing rust residues, cleansing mineral deposits, bleaching wood, processing leather and producing high-purity metals. It is involved in the preparation of rare-earth compounds because the formation of oxalate complexes can selectively precipitate certain ions while leaving others in solution. Pharmaceutical manufacturing and analytical chemistry also utilize oxalic acid when precise reduction activity is required. In household products, oxalic acid appears in some stain removers and rust cleaners because of its metal-binding structure, though safe handling is important due to its corrosive nature.
Toxicity arises not simply from the acidic character of oxalic acid, but from its structural affinity for calcium and other essential minerals in the human body. Ingesting large amounts of oxalic acid or concentrated oxalate salts can lead to hypocalcemia, kidney problems, cellular irritation, or metabolic imbalances. However, dietary oxalic acid contained naturally in foods such as leafy greens is not inherently dangerous when consumed in moderation, especially when paired with sufficient calcium intake. The risk increases when very high-oxalate foods are overconsumed while mineral intake is low. Therefore, biochemical context matters just as much as structural chemistry when discussing safety.
The story of oxalic acid demonstrates that the identity of a chemical compound is a direct consequence of its molecular structure. The two tightly linked carboxyl groups create strong acidity, enable metal chelation, support crystal formation, affect solubility behavior and dictate biological and industrial effects. It is a compound that bridges organic chemistry, biochemistry, industrial science and medicine. Its usefulness in cleaning, manufacturing and laboratory reactions arises from the same structural forces that contribute to kidney stones and biological irritation under certain circumstances. Oxalic acid illustrates the broader principle that chemical structure determines chemical behavior: the same atoms arranged differently would produce a completely different compound, but the precise arrangement in oxalic acid unlocks both wide utility and biological caution.
Understanding oxalic acid is therefore not merely a matter of knowing its formula; it requires acknowledging how its structure enables its acidity, its reactivity, its chelation power, its natural roles and its health implications. In nature and industry alike, oxalic acid exemplifies how the geometry of a molecule shapes how it interacts with the world.