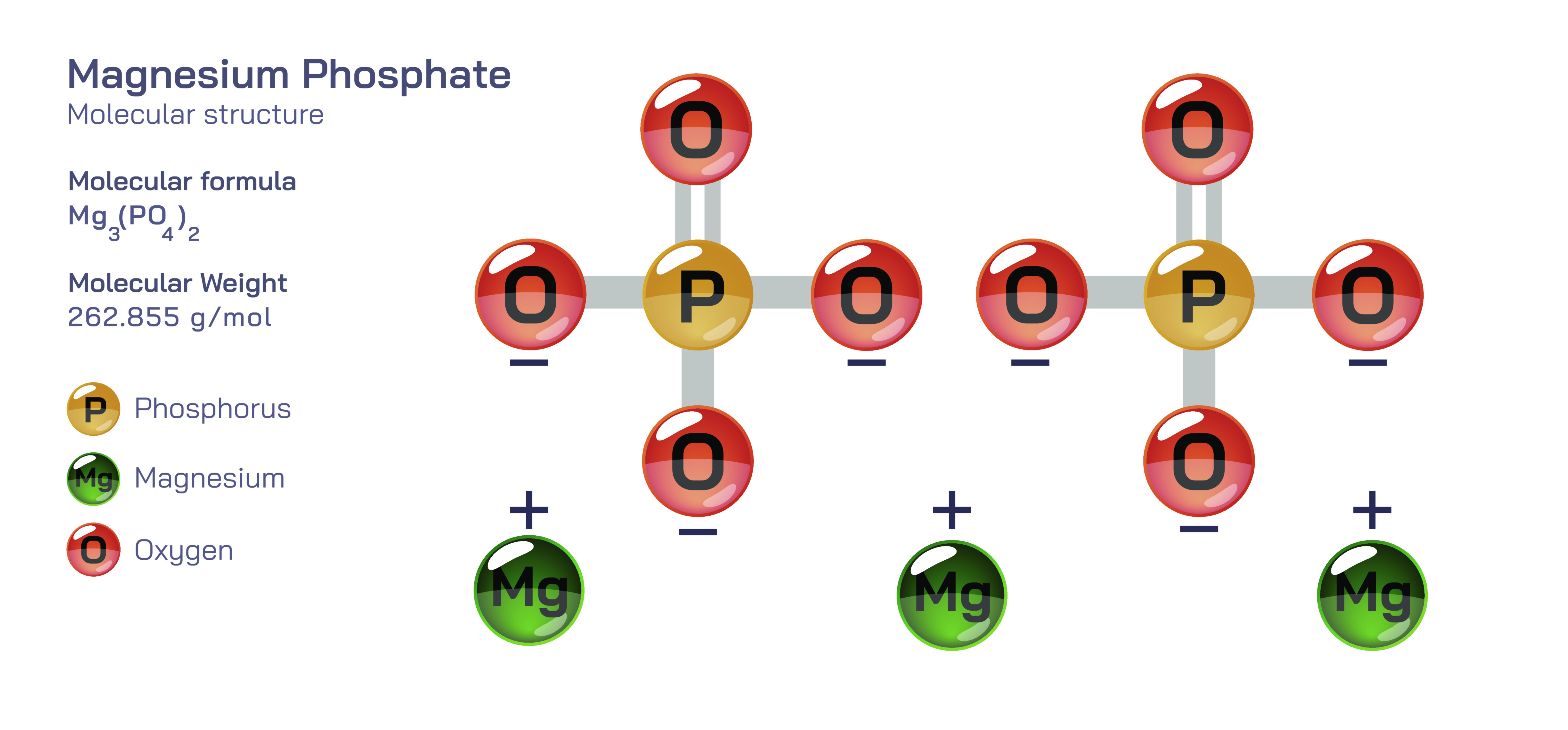
Magnesium Phosphate — Molecular Structure and Composition
Magnesium phosphate is an important family of inorganic compounds composed of magnesium ions and phosphate groups, and it occupies a central position in biological chemistry, nutritional science, industrial processes, and medical therapeutics due to the deep relationship between its molecular architecture and its physiological functionality. Although “magnesium phosphate” is often spoken of as a single substance, the term actually encompasses multiple structural variants, including monomagnesium phosphate, dimagnesium phosphate, and trimagnesium phosphate, each defined by the ratio between magnesium ions and phosphate units. The phosphate group itself is built from one phosphorus atom surrounded by four oxygen atoms arranged in a tetrahedral configuration, and this arrangement has an extraordinary capacity to form stable ionic bonds with magnesium, a divalent metal cation with a high affinity for oxygen-based ligands. When magnesium binds to phosphate groups, the result is a highly cohesive three-dimensional ionic network capable of forming crystalline solids that remain structurally robust under physiological and environmental conditions. The exact crystal pattern varies with composition and hydration, giving rise to forms that range from fully anhydrous to multi-hydrated molecular assemblies. These subtle structural differences determine solubility, bioavailability, acidity, stability, and functional behavior, which explains why magnesium phosphates have become widely distributed across biomedical formulations, food fortification programs, fertilizers, water systems, and tissue-engineering technologies.
The biochemical relevance of magnesium phosphate is profound because magnesium and phosphate together take part in many of the most fundamental pathways of life. Inside living organisms, the interaction between magnesium ions and phosphate groups governs the behavior of nucleic acids, energy metabolism, muscle contraction, and enzyme activation. ATP, the primary energy currency of the cell, is not biologically stable unless coordinated with magnesium, and this coordination is only possible because of the strong but flexible bond between magnesium and phosphate groups. Without this relationship, the energy-transfer machinery that drives cell division, nutrient transport, nerve signaling, and metabolic regulation would collapse. Magnesium phosphate also plays a crucial structural role in bones and teeth, where it forms part of a mineral matrix integrated with calcium phosphate, creating strong yet adaptable tissue that can withstand mechanical force while remaining biologically dynamic. Although calcium is more abundant in skeletal frameworks, magnesium is essential for stabilizing the mineral microstructure, influencing crystal size, bone density, resistance to fracture, and regenerative remodeling over the human lifespan. Deficiency of magnesium, therefore, does not only affect biochemical reactions but can also influence skeletal health by disturbing the formation of the phosphate-rich mineral lattice that gives bones their strength.
Different forms of magnesium phosphate can be formulated for medical and nutritional applications depending on their solubility and biochemical effects. Trimagnesium phosphate, which contains a relatively high proportion of magnesium to phosphate, dissolves slowly and is frequently used as a gentle mineral supplement because it allows gradual magnesium release without sudden digestive discomfort. In contrast, monomagnesium phosphate and dimagnesium phosphate dissolve more readily and can be used when faster ionic availability is needed inside the body or in pharmaceutical formulations. Because magnesium phosphate behaves as a mild alkaline compound, it is incorporated into antacid preparations to neutralize excess stomach acidity while simultaneously providing a bioavailable source of magnesium. Its buffering characteristics reduce gastrointestinal irritation, making it suitable for individuals who cannot tolerate harsher basic compounds. In addition to digestion-related applications, magnesium phosphate has long been used in homeopathic traditions to support neuromuscular relaxation, although the effectiveness of such approaches depends on complex factors and should be interpreted responsibly within the context of modern evidence-based medicine. Nevertheless, due to its safe chemical nature and direct biological compatibility, magnesium phosphate remains a widely trusted therapeutic mineral in both mainstream and alternative health practices.
Beyond biomedical use, magnesium phosphate is valuable in agricultural science because it offers dual nutrient support by supplying both magnesium and phosphorus, two of the most critical elements in plant growth. Magnesium fuels chlorophyll production and energy transport in leaves, while phosphorus contributes to root formation, cellular reproduction, seed development, and carbohydrate metabolism. The phosphate chemistry of magnesium phosphate provides a slow-release nutrient profile that supports crop growth over time without causing the sudden nutrient spikes associated with highly soluble fertilizers that can wash away quickly or trigger algae-induced eutrophication in water systems. In hydroponics and controlled-environment agriculture, magnesium phosphate is sometimes used as a mineralizing agent due to its predictable dissolution behavior and its ability to stabilize nutrient solutions through pH buffering. The ecological safety of the compound has contributed to its use in sustainable farming programs that prioritize soil structure, long-term fertility, and environmentally responsible mineral delivery.
Industrial and technological fields use magnesium phosphate in specialized ways arising from its thermal behavior, mechanical strength, and compatibility with ceramic systems. Magnesium phosphate cements have gained attention as a rapid-setting and durable class of construction materials that differ from traditional Portland cement by forming mineral bonds based on phosphate chemistry rather than calcium silicate hydration. These cements exhibit impressive strength development, adhesion to various substrates, and resistance to chemical corrosion, which makes them especially useful in emergency repairs, cold-weather construction, and industrial flooring exposed to corrosive materials. Because magnesium phosphate cement cures quickly and emits significantly lower carbon dioxide during production, it serves as a more sustainable alternative in certain civil engineering applications. In advanced ceramics and composite materials, magnesium phosphate contributes to the formation of microstructures capable of withstanding high temperatures, suppressing thermal expansion, and improving mechanical integrity. Such characteristics are useful in protective coatings, refractory materials, electronic substrates, and fire-resistant architectural components.
In water treatment systems, magnesium phosphate is utilized in processes that depend on phosphate-based coordination to remove unwanted metal ions and control water hardness. Its affinity for calcium ions enables controlled precipitation that prevents scaling inside pipes and boilers, where mineral buildup can drastically reduce efficiency and increase operational costs. In ecological water systems, carefully formulated magnesium phosphate compounds help stabilize mineral equilibrium and reduce undesirable shifts in pH that might threaten aquatic organisms. Although phosphate chemistry must always be managed responsibly to prevent over-enrichment of water bodies, magnesium phosphate is considered relatively safe because of its ability to remain in stable mineral form without excessive rapid leaching.
Magnesium phosphate is also known for its contributions to food science and consumer product formulations. It acts as a stabilizer, emulsifier, and mineral fortifier in bakery products, dairy items, nutritional powders, and beverages. Its mild taste and non-reactive behavior allow it to maintain the structural and sensory qualities of food without producing off-flavors. In powdered supplements and pharmaceutical tablets, magnesium phosphate functions as both an active mineral ingredient and a flow aid that improves the texture and uniformity of blended products. Personal-care manufacturers use magnesium phosphate as an absorbent, deodorizing, and pH-regulating ingredient that is skin-friendly and free from harsh reactive tendencies, making it suitable for sensitive-skin products, mineral cosmetics, and gentle everyday formulations.
Although magnesium phosphate is safe, non-toxic, and environmentally compatible, it requires proper handling to preserve its performance characteristics. Exposure to excessive moisture may alter its hydration state, which can change both its weight and solubility profile. In industrial settings where precise stoichiometry is necessary, magnesium phosphate should therefore be stored in dry, sealed environments to avoid unintended hydration or clumping. In agricultural applications, overapplication must be avoided to prevent nutrient saturation in soil, and balanced mineral management remains essential for supporting sustainable ecosystem health.
Magnesium phosphate ultimately demonstrates how molecular structure and composition shape real-world functionality in a profound way. The interaction between magnesium and the tetrahedral phosphate group produces a mineral system that is stable yet biologically compatible, strong yet soluble under specific conditions, and versatile enough to support human physiology, plant development, industrial manufacturing, and cutting-edge material science simultaneously. Whether strengthening bones and teeth, stabilizing cellular energy cycles, enhancing soil fertility, enabling advanced ceramic technology, fortifying therapeutic supplements, or contributing to rapid-setting sustainable construction materials, magnesium phosphate stands as a clear example of how foundational mineral chemistry continues to support scientific progress, environmental responsibility, and human wellbeing across multiple dimensions of modern life.