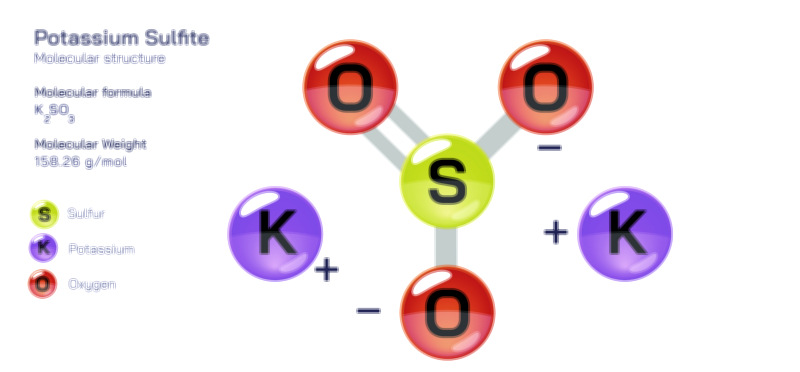
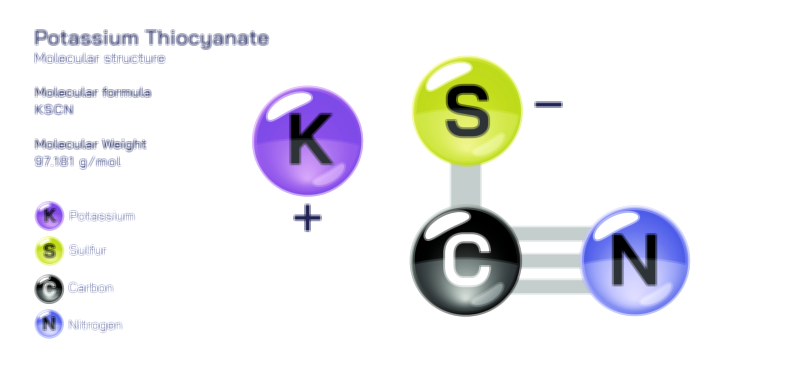
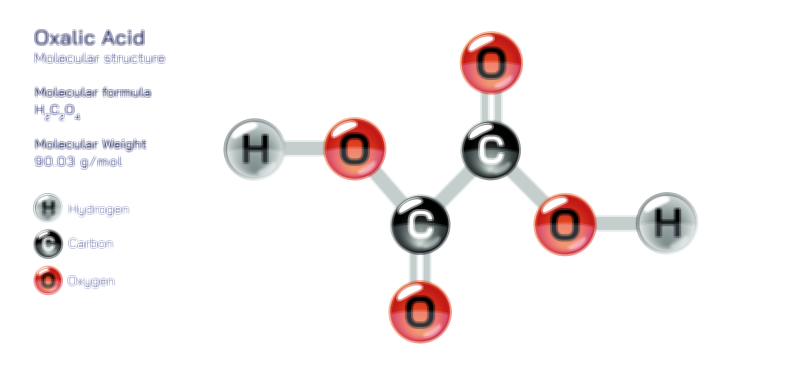
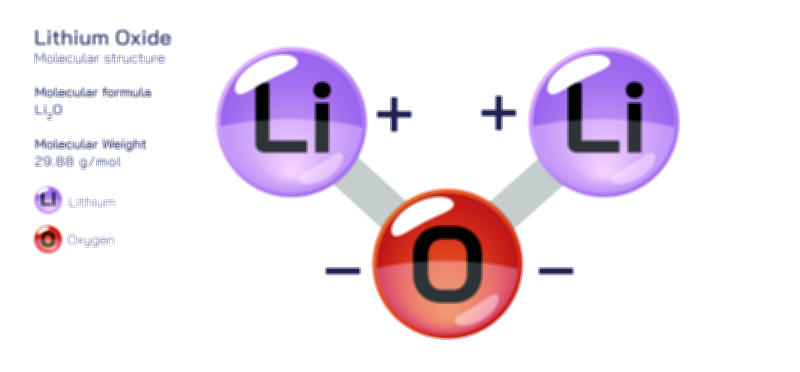
ØÉüØÉÜØɽØÉóØÉ«ØÉ” ØÉÄØÉ▒ØÉóØÉØØÉ× ŌĆö ØÉéØÉĪØÉ×ØÉ”ØÉóØÉ£ØÉÜØÉź ØÉéØÉ©ØÉ”ØÉ®ØÉ©ØÉ«ØɦØÉØ ØÉÜØɦØÉØ ØÉīØÉ©ØÉźØÉ×ØÉ£ØÉ«ØÉźØÉÜØɽ ØÉÆØÉŁØɽØÉ«ØÉ£ØÉŁØÉ«ØɽØÉ×.
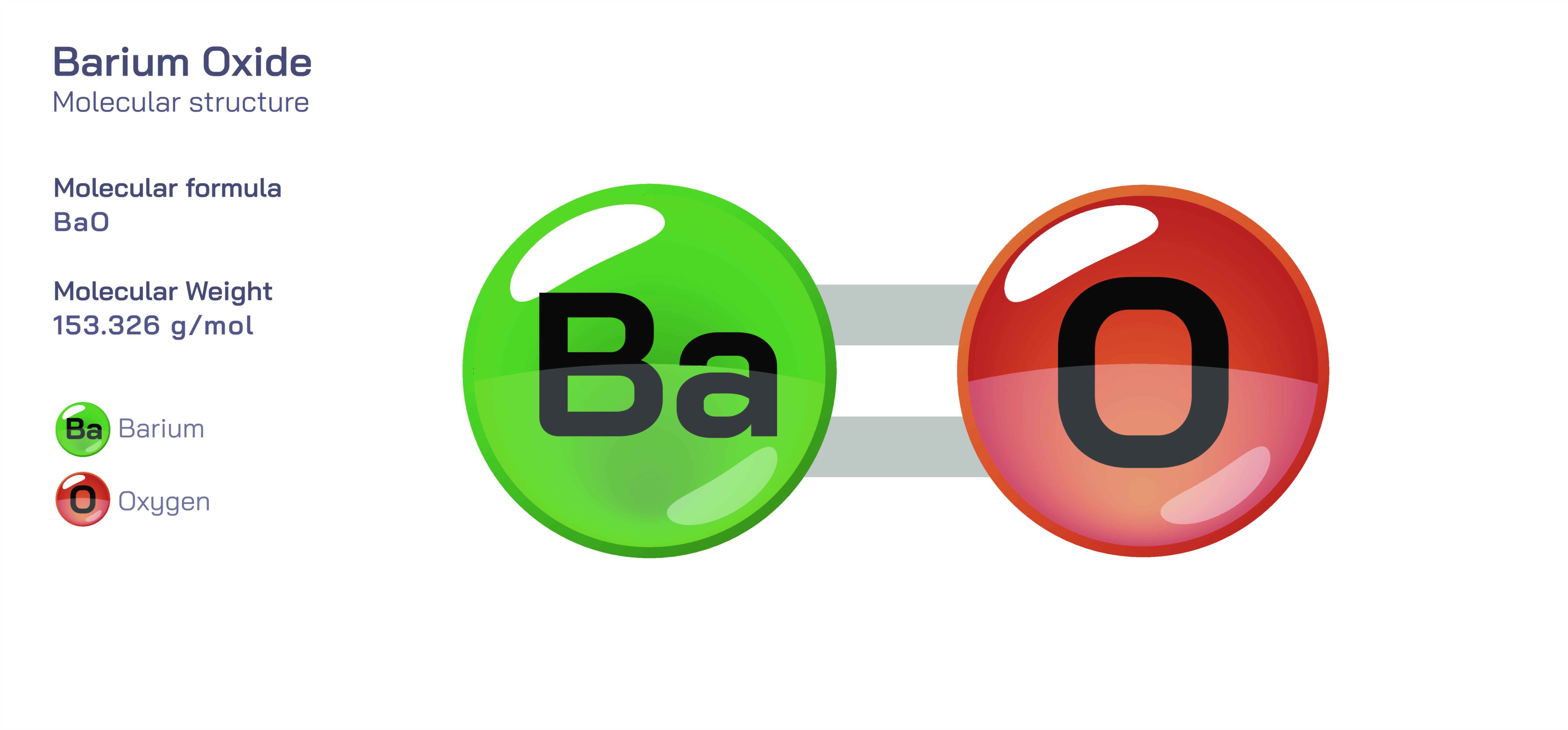
Barium oxide is an inorganic chemical compound composed of barium and oxygen, known scientifically by the formula BaO. It belongs to the family of alkaline earth metal oxides and stands out due to its strong ionic character, high melting point, robust basicity, and exceptional reactivity with moisture and carbon dioxide. The compound appears as a white to slightly yellow crystalline solid, but behind this simple appearance lies an intricate structural framework that governs all behaviours of the compound, including its solubility, reactivity, industrial applications, toxicity, and environmental sensitivity. At the atomic level, barium oxide consists of Ba┬▓Ōü║ cations and O┬▓Ōü╗ anions arranged in a strongly bonded ionic lattice. This structure represents one of the foundational forms of metal oxides, demonstrating the powerful electrostatic attraction between the highly electropositive barium ion and the highly electronegative oxide ion. The nature of the lattice determines physical features such as brittleness, thermal resistance, and density, while the chemical identity of the O┬▓Ōü╗ ion at the surface ensures extremely high reactivity toward proton donors such as water.
The ionic structure of barium oxide is the key to its molecular behaviour. In the solid phase, the Ba┬▓Ōü║ ions adopt a highly coordinated position within the lattice, typically surrounded symmetrically by oxide ions due to their large ionic radius. Likewise, O┬▓Ōü╗ ions occupy complementary positions in the lattice, creating a geometrically stable, repeating arrangement. Unlike covalent molecules that contain directional electron sharing and discrete molecular units, BaO exists as an extended structure in which ions form a continuous three-dimensional network. The simplicity of the formula BaO therefore does not reflect separate barium-oxide molecular pairs, but rather an immense ionic crystal in which the Ba┬▓Ōü║ and O┬▓Ōü╗ ions repeat throughout the solid. The strong electrostatic attraction in the lattice accounts for the compoundŌĆÖs high melting and boiling point and its thermal stability at elevated temperatures, making BaO suitable for high-temperature industrial environments. The broad spatial electron distribution around the oxygen ion gives the lattice basicity, which is why BaO reacts vigorously with acids and moisture.
The basic character of barium oxide originates from the oxide ion, a strong proton acceptor. When exposed to water, BaO reacts exothermically to form barium hydroxide, Ba(OH)Ōéé, releasing heat and demonstrating a powerful affinity for hydrogen. This reaction exemplifies the fundamental difference between simple hydration and chemical combination, because the oxide ion in BaO does not merely dissolve but actively reacts to generate a new compound. This strong interaction with water makes barium oxide a highly hygroscopic substance, meaning it absorbs moisture quickly from the air. Even in the absence of liquid water, BaO can gradually react with atmospheric moisture to form barium hydroxide, explaining why the solid must be stored in airtight environments. In addition to moisture sensitivity, barium oxide also reacts with carbon dioxide to form barium carbonate, BaCOŌéā. This reaction illustrates the compoundŌĆÖs capacity to function as a chemical absorber of both water and carbon dioxide, making it useful in gas purification systems and carbon dioxide scavenging in vacuum tubes, where oxygen- and carbon dioxide-free atmospheres are required for proper functioning.
The structural characteristics of BaO also determine its solubility behaviour. Unlike many barium salts that dissolve readily, BaO does not dissolve in water but reacts with it, producing hydroxide ions, which dramatically increase basicity of the resulting solution. The presence of mobile Ba┬▓Ōü║ and OHŌü╗ ions in the resulting barium hydroxide solution makes the liquid electrically conductive and strongly alkaline. This chemical transformation confirms that the oxide ion behaves not as a passive spectator but as a reactive species that energetically seeks proton acquisition. The reaction pathway reveals a deeper truth about the relationship between ionic solids and their affinity for water: some ionic compounds dissolve; others undergo chemical transformation. Barium oxide belongs to the latter category because the lattice structure is not stable in aqueous environments due to the dominance of proton affinity at the oxide surface.
Industrial and scientific applications of barium oxide reflect its structural reactivity and thermodynamic properties. In the glass and ceramics industry, BaO is incorporated into specialized glass formulations to enhance thermal resistance, refractive index, and electrical insulating properties. Barium oxide helps create high-quality optical glasses used in lenses and precision optical instruments. In electronics and vacuum technology, BaO plays a critical role in cathode coatings for vacuum tubes, electron guns, and cathode-ray devices. When heated, BaO can release electrons more easily than many other oxides, improving electron emission efficiency. In older television and radio tubes, barium oxide coatings significantly improved brightness and signal amplification due to this electron emissivity. In modern catalytic chemistry, BaO is used in the synthesis of advanced materials, in NOx storage catalysts, and in processes requiring high-temperature basic catalysis. In metallurgical operations, BaO sometimes serves as a fluxing agent to assist in removing unwanted silica and other impurities.
Barium oxide is also historically tied to the development of gas absorption systems. Before modern materials replaced the technology, BaO was frequently used to maintain low-carbon dioxide atmospheres inside vacuum tubes, laboratory chambers, and submarine air filtration systems. The ability of BaO to remove COŌéé permanently by forming BaCOŌéā made it a reliable gas-cleaning agent, though handling challenges eventually led to the adoption of safer alternatives. Yet even today, the study of BaO remains central to research in gas sorbents because of its thermodynamic affinity for acidic oxide species.
Despite its usefulness, barium oxide must be handled carefully due to its biological toxicity. The danger does not originate from the oxygen component but from the Ba┬▓Ōü║ cation. When BaO reacts with water, it produces soluble barium ions, which can enter the bloodstream if ingested or inhaled in dissolved form. Barium ions interfere with potassium ion transport in nerves and muscles, disrupting physiological electrical impulses. Symptoms of barium poisoning include muscle weakness, vomiting, breathing difficulty, cardiac arrhythmia, and in severe cases paralysis and cardiac failure. In addition, BaOŌĆÖs strong alkalinity can cause burns upon contact with skin or eyes. Fortunately, its toxicity is avoidable through proper laboratory protocolsŌĆögloves, sealed containers, dry storage, and avoidance of ingestionŌĆöand barium compounds that are insoluble, such as barium sulfate, do not pose the same risk because they do not release Ba┬▓Ōü║ ions in the body.
The structural simplicity of BaO highlights a deeper theme in chemistry: even chemically simple formulas can conceal complex behaviours governed by ionic structure. The compound does not act like a neutral, inert solid but like a powerful base embedded in a crystal lattice. The absence of shared electrons leads BaO to behave according to pure electrostatic forces in the solid state but as a chemically reactive proton acceptor in the presence of water. Every major property of barium oxideŌĆöits strong basicity, ability to absorb water and carbon dioxide, transformation into hydroxide or carbonate, electron emission properties, industrial value, and toxicityŌĆöflows directly from its fundamental ionic structure made of Ba┬▓Ōü║ and O┬▓Ōü╗ ions.
In summary, barium oxide is a compelling example of how chemical structure defines chemical identity. Its lattice of strongly bonded ions gives it thermal stability, but the high reactivity of the oxide ion controls its behaviour in aqueous and atmospheric environments. It is a compound with dual significanceŌĆöhighly valuable in industry and science, yet hazardous when mismanaged. Understanding barium oxide therefore requires a structural perspective rather than a superficial formula, because the arrangement and charge of ions determine precisely how the compound interacts with materials, living systems, and the environment.