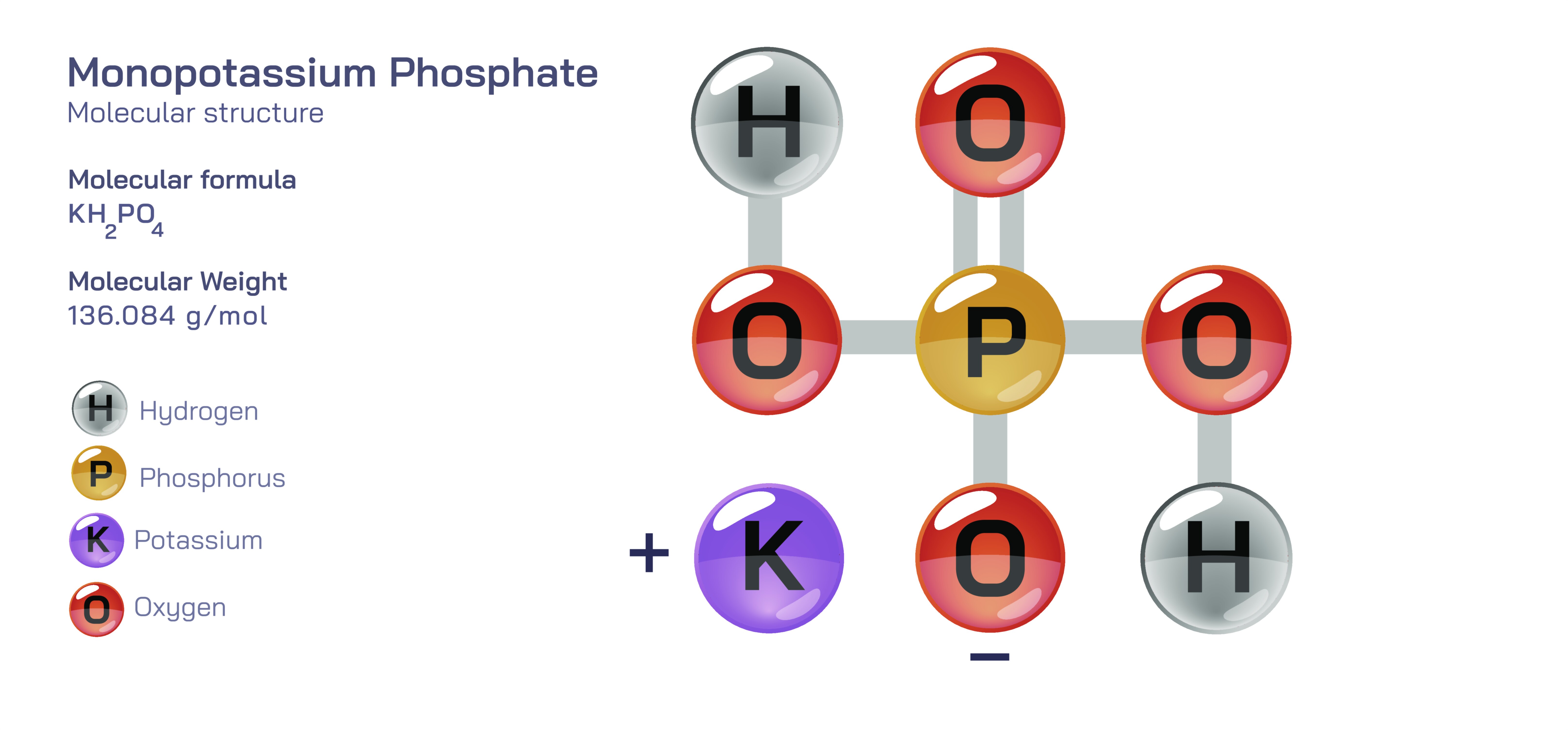
Monopotassium Phosphate — Molecular Structure and Composition
Monopotassium phosphate is an inorganic compound whose molecular architecture lies at the heart of its important roles in agriculture, plant nutrition, food chemistry, biochemistry, industrial processing, and laboratory science. Known chemically as KH₂PO₄, the compound consists of one potassium ion (K⁺) paired with a dihydrogen phosphate anion (H₂PO₄⁻). Although the formula appears simple, the internal structure of the phosphate unit and its network of hydrogen bonding define everything about the compound—from its solubility and dissociation in water to its buffering behavior and biological significance. The phosphate anion is based on a tetrahedral framework in which a central phosphorus atom is bonded to four oxygen atoms. Two of these oxygen atoms carry acidic hydrogens, another is double-bonded to phosphorus, and the fourth is singly bonded and carries the negative charge that attracts and pairs with the potassium cation. This arrangement produces a molecule with strong polarity and high ionic strength, allowing monopotassium phosphate to dissolve readily in water and release potassium ions and phosphate species essential to biological and chemical systems. That simple tetrahedral geometry is the foundation of the compound’s massive importance, because phosphate—with its ability to bind metals, buffer chemical conditions, and transfer energy—has been integral to Earth’s chemistry and life since the earliest stages of biological evolution.
Once KH₂PO₄ dissolves in water, the interactions between the ionic components extend into a dynamic network of hydrogen-bonded structures. The dihydrogen phosphate anion does not remain static in solution; instead, it participates in acid–base equilibria that shift based on pH. Depending on conditions, H₂PO₄⁻ may donate one of its acidic protons, forming hydrogen phosphate (HPO₄²⁻) while releasing a hydronium ion. This dissociation does not modify the core tetrahedral geometry of the phosphate group but changes the charge distribution around the oxygen atoms, providing one of the key reasons phosphate salts—including monopotassium phosphate—function as buffers. In environments where pH tends to fall, the molecule accepts excess hydrogen ions by converting HPO₄²⁻ back to H₂PO₄⁻; in conditions where pH tends to rise, the anion donates a proton to preserve chemical balance. This reversible proton shifting is one of the most important buffering systems in biological science and enables phosphate-containing compounds to maintain stable reaction conditions in metabolic pathways, enzyme systems, laboratory media, industrial formulations, and even living cells.
Monopotassium phosphate has become one of the most valued fertilizers in the world because its molecular composition delivers two nutrients that plants require for optimal growth—potassium and phosphorus—without adding nitrogen or altering soil chemistry dramatically. The potassium ion contributes to osmotic regulation, enzyme activation, carbohydrate metabolism, and stress tolerance in plants, while the phosphate species contribute to root development, flowering, fruit formation, ATP-based energy transfer, and photosynthetic efficiency. Because KH₂PO₄ dissolves quickly and does not leave insoluble residues, it delivers these nutrients in a form that plant roots can immediately absorb. The fact that the phosphate is already in the dihydrogen form means that it exists in a plant-compatible stage of acidity, bypassing the slow dissolution and soil-bound losses associated with rock phosphate or less soluble forms. The molecular structure therefore influences agricultural value directly: potassium remains mobile in solution while phosphate remains plant-available without excessive binding to soil particles such as calcium, magnesium, aluminum, or iron. This characteristic makes monopotassium phosphate indispensable in fertigation systems, hydroponic agriculture, and precision-nutrient programs where controlled solubility, nutrient purity, and predictable uptake determine crop productivity.
In food and pharmaceutical systems, the buffering and ionic properties of monopotassium phosphate allow it to stabilize acidity, support fermentation, preserve texture, and regulate biochemical reactions. Because the molecule dissociates smoothly and does not introduce strong off-flavors, it has become a preferred source of phosphate in dairy products, bakery formulas, and beverages, where pH must be controlled to preserve flavor stability and microbial safety. In yeast-based fermentation, the orthophosphate group plays a critical biochemical role because phosphate is essential to the phosphorylation steps that support glycolysis and energy metabolism, while the potassium ion supports yeast viability. This makes KH₂PO₄ far more than a passive additive—it becomes a nutrient that shapes reaction rates in food and beverage production. In pharmaceutical science, the same chemical logic applies: monopotassium phosphate appears in oral formulations, nutritional products, parenteral solutions, and hydration therapies where pH buffering and electrolyte balance determine product safety and biological compatibility. The tetrahedral phosphate core and the ease of dissociation into charged ions are central to its role in stabilizing solutions for drug delivery and biochemical assays.
Monopotassium phosphate also plays a powerful role in laboratory research. Phosphate-buffered systems are among the most widely used aqueous environments in molecular biology, microbiology, and biochemistry because they maintain stable pH values across physiological ranges without interfering with most enzymes, proteins, or cell membranes. The buffering action derived from the H₂PO₄⁻ ↔ HPO₄²⁻ equilibrium allows reaction conditions to remain stable during metabolic studies, PCR protocols, protein purification, electrophoresis, enzyme kinetics, and cell culture. In vitro systems often rely on KH₂PO₄ to provide a predictable phosphate supply without the presence of sodium, which may disrupt certain ion-sensitive experiments. Even in animal physiology, the biological compatibility of potassium phosphate solutions has made them useful in electrolyte replacement therapies because the molecular structure mirrors the ionic species that appear in natural intracellular fluids. The same tetrahedral geometry and proton-transfer logic that govern phosphate in living organisms make monopotassium phosphate an ideal laboratory buffer.
In material science and industrial chemistry, the compound’s ionic strength and thermal behavior come into play. Heating drives off water and eventually converts monopotassium phosphate into pyrophosphates and polyphosphates, creating long chains of phosphate units that have their own important applications—including high-temperature fluxing agents, metal-treatment compounds, flame retardants, and components of fire-extinguishing powders. These transformations occur because the tetrahedral phosphate groups share oxygen atoms when dehydrated, linking into P–O–P chains without collapsing the phosphorus–oxygen framework. Even in these altered states, the structural logic of the phosphate core persists. Meanwhile, in water treatment and detergent chemistry, monopotassium phosphate offsets hardness-causing ions by stabilizing calcium and magnesium in solution, preventing scaling and improving performance of soaps and industrial cleaners. Although environmental regulations have reduced phosphate use in consumer detergents, the same underlying chemistry remains important in specialized industrial treatments where scale inhibition and water conditioning must be tightly controlled.
The large-scale significance of monopotassium phosphate is inseparable from the deep biological importance of phosphorus. Phosphate is one of the pillars of life: it forms the backbone of DNA and RNA, anchors cell membrane phospholipids, connects the high-energy bonds in ATP, and supports enzyme structure and signaling pathways. The compound KH₂PO₄ is not exotic or chemically unusual—it is important largely because it acts as a stable, water-soluble, and biologically compatible form of phosphate and potassium, both of which are essential for living systems. The tetrahedral P–O arrangement makes phosphorus one of the few elements capable of enabling energy transfer and information storage at the scale of molecular biology. Monopotassium phosphate therefore participates in global nutrient cycles, agricultural systems, biochemical processes, and industrial applications not because of accidental usefulness but because its structure is aligned with the fundamental logic of how life organizes and transfers energy.
Even its environmental role reflects molecular structure. In responsible use, monopotassium phosphate supports plant development, food production, and nutrient cycling. When mismanaged, phosphate runoff contributes to eutrophication in freshwater ecosystems, stimulating excessive algal growth and disrupting ecological balance. The same solubility that makes the compound efficient in agriculture makes it mobile in soil water. This is not an argument against its value but an acknowledgment that structural power requires responsible stewardship. The mobility of phosphate ions and their affinity for biological systems underscore why nutrient management strategies—not chemical avoidance—form the basis of sustainable use.
Across all areas—agriculture, food science, medicine, laboratory chemistry, industrial processing, and environmental systems—the influence of monopotassium phosphate always returns to the same unifying theme: the arrangement of atoms determines the behavior of matter. The potassium ion remains freely dissociated in water because its electron configuration does not promote covalent bonding in this context. The tetrahedral phosphate anion gains its buffering ability from the proton-transfer capacity embedded in its oxygen substructure. Solubility, pH control, nutrient availability, buffering capacity, and biological functionality are not abstract characteristics but direct expressions of molecular geometry.
Monopotassium phosphate stands as a powerful example of how a compound can play roles of global importance not because it is chemically complex, but because its structure is exquisitely aligned with the needs of living systems and industrial processes. A single potassium ion and a single dihydrogen phosphate unit—once dissolved and distributed—participate in energy metabolism, cell signaling, food production, laboratory analysis, water treatment, and material transformation. Through its composition and structure, KH₂PO₄ continues to shape the chemical, biological, and agricultural foundations of life and technology, illustrating the enduring truth that the microscopic architecture of molecules governs the macroscopic world we build and inhabit.