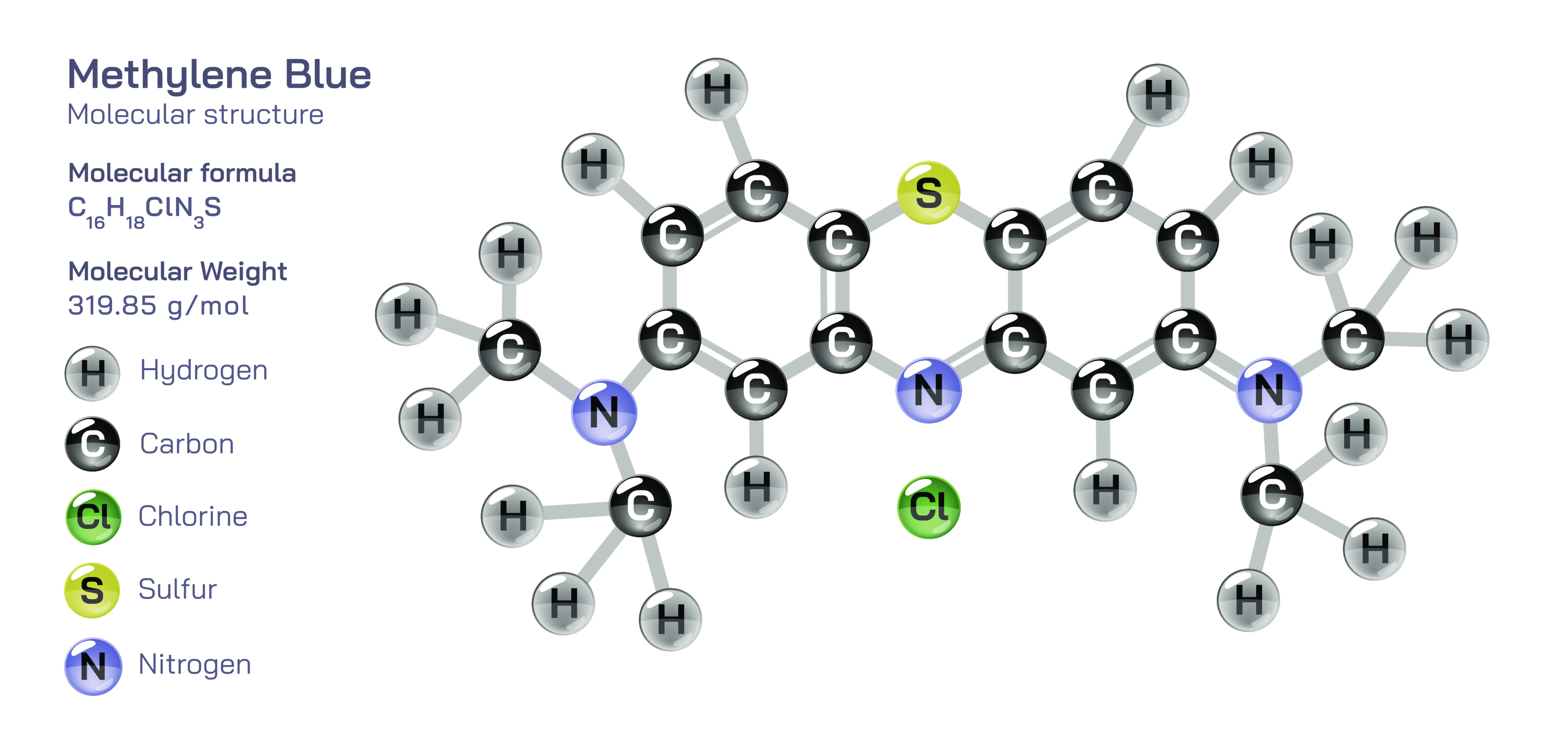
Methylene Blue — Molecular Structure and Composition
Methylene blue is an organic compound whose molecular structure and elemental composition have given it an extraordinary range of functions in medicine, biology, chemistry, materials science, and even energy research. Known chemically as a phenothiazinium dye, methylene blue contains a tricyclic aromatic core arranged around a sulfur and nitrogen heterocycle, giving the molecule a richly conjugated electronic system that absorbs light strongly in the visible range and produces the deep blue color for which it is named. Its molecular formula, C₁₆H₁₈ClN₃S, reflects not only the carbon, hydrogen, nitrogen, sulfur, and chlorine present in the compound but also the underlying ionic nature of the salt. In biological and chemical applications, methylene blue typically exists as a chloride salt composed of a positively charged dye cation and a chloride anion that balances electrical charge. The core of the molecule consists of linked benzene rings whose π-electron system extends across the phenothiazine structure, enabling strong electron delocalization. This delocalization is responsible for methylene blue’s deep coloration, its fluorescence, and its capacity to participate in electron transfer reactions. The nitrogen substituent attached to the aromatic system allows the molecule to carry a positive charge, making it water soluble while permitting it to associate selectively with negatively charged biological structures such as nucleic acids, cellular membranes, and mitochondrial components. Every medical, biochemical, and industrial use of methylene blue emerges from this intricate molecular architecture, which fuses aromatic stability, ionic solubility, light absorption, and redox chemistry into a single compound.
One of the defining characteristics of methylene blue is its reversible redox behavior. The molecule can readily cycle between its oxidized blue form and its reduced colorless form, known as leucomethylene blue. This transformation does not destroy the compound; rather, it shifts electrons within the aromatic ring system, altering optical properties without affecting fundamental structural integrity. This reversible redox capacity is the reason methylene blue became one of the earliest redox indicators used in chemistry and physiology. In microbiology laboratories, its behavior serves as a visual marker for oxygen availability because the dye turns colorless when electrons accumulate under anaerobic conditions and regains its blue color when oxygen becomes available to reoxidize it. In biochemical pathways, the compound can intervene in mitochondrial electron transport and cellular respiration by acting as an alternative electron carrier, a property that has given it therapeutic relevance in conditions involving oxidative stress and impaired metabolism. When methylene blue participates in these redox cycles, the molecular structure does not fragment; instead, electrons move across the conjugated aromatic system in a reversible fashion that preserves the core. This reversible flow of electrons is built into the very geometry of the molecule and explains why methylene blue acts simultaneously as a dye, a redox agent, and a metabolic modulator.
At the biological level, the positive charge carried by methylene blue plays an equally important role in shaping its interactions with cells and tissues. Charged molecules navigate biological environments differently than uncharged ones because they are attracted to specific molecular surfaces, organelles, and charged biological macromolecules. Methylene blue associates strongly with anionic cellular components, especially nucleic acids and elements of mitochondrial membranes. This affinity has been exploited for more than a century in microscopy and staining techniques, where the dye binds to cellular structures and makes them visible under light microscopes. In hematology, the molecule stains reticulocytes and other blood cells for diagnostic purposes, and in histopathology it highlights tissue features, microbial organisms, and structural elements that would otherwise be difficult to observe. Its deep absorption band in the visible spectrum allows tiny amounts of dye to produce intense coloration, making the molecule highly efficient for diagnostic staining. Because the charged aromatic core penetrates biological membranes in a controlled manner, methylene blue also displays selective localization in certain cellular compartments, particularly mitochondria, which underlies therapeutic and toxicological properties alike.
In medicine, methylene blue has an unusually broad therapeutic profile because of the interaction between its redox chemistry and its electrostatic affinity for biological systems. The compound is the primary treatment for methemoglobinemia, a condition in which hemoglobin becomes oxidized to methemoglobin and loses its ability to transport oxygen. Methylene blue, once reduced to its colorless leuco form within red blood cells, donates electrons to convert methemoglobin back to functional hemoglobin, restoring oxygen-carrying capacity. This process demonstrates how electron transfer embedded in the molecular design can be harnessed for life-saving intervention. The same properties allow methylene blue to act as a powerful antidote in specific cases of poisoning involving agents that interfere with mitochondrial respiration or disrupt redox balance. In controlled low doses, methylene blue has been studied for its ability to support mitochondrial efficiency, counteract oxidative stress, and modulate neurotransmission through pathways involving nitric oxide, monoamine oxidase, and guanylate cyclase. These effects arise not from synthetic modification of biological structures but from subtle redox cycling within living systems, anchored to the phenothiazinium core.
Methylene blue’s structure also determines its photochemical activity, which forms the basis for another major area of use: photodynamic therapy and antimicrobial photoinactivation. The extended aromatic ring system allows methylene blue to absorb visible light and enter an excited electronic state. From this state, the dye can transfer energy to surrounding oxygen molecules, generating reactive oxygen species such as singlet oxygen. These reactive species damage microbial cell walls, nucleic acids, and metabolic enzymes, producing an antimicrobial effect that does not rely on conventional antibiotics. Because only illuminated regions are affected, this property enables targeted therapy for localized infections, wound disinfection, dental treatments, and biofilm reduction. The photodynamic capability originates directly from the conjugated heterocyclic structure of the molecule, which allows it to interact with light and oxygen in a cyclic, catalytic manner without decomposition. This light-dependent antimicrobial behavior illustrates the remarkable way in which chromophore geometry can determine biological function.
Outside biological systems, the molecular composition of methylene blue shapes its behavior in analytical chemistry, water treatment, and environmental science. Its intense coloration and sensitivity to redox changes make it a reliable indicator for oxidation–reduction potential and electron flow in chemical solutions. In environmental testing, methylene blue provides a rapid method for measuring the presence of surfactants and organic contaminants in water, because those compounds interact with the dye at the molecular level, causing measurable changes in absorbance. The stability of the dye in aqueous environments allows it to function without rapid decomposition, and its charge enables straightforward spectrophotometric detection. In water purification and research systems, methylene blue serves as a model contaminant for studying adsorption mechanisms onto activated carbon, clays, nanocomposites, and biopolymers. Because the dye binds strongly to these surfaces, it becomes a valuable probe for characterizing pollutant removal efficiency in wastewater treatment studies. This widespread research use stems from fundamental chemical properties rooted in the molecular structure: the extended aromatic system favors adsorption through π–π interactions, and the positive charge creates electrostatic attraction to negatively charged surfaces.
The compound’s electrochemical and photophysical behavior has also positioned it at the forefront of innovative research fields such as molecular electronics, photochemical energy storage, photocatalysis, and artificial photosynthesis. In these contexts, methylene blue acts as a mediator capable of shuttling electrons between molecular components or amplifying solar-driven reactions. The dye’s resonance-stabilized aromatic core enables rapid and reversible charge transfer, while its cationic nature facilitates controlled electron mobility in electrolytes and polymer films. As material scientists explore organic semiconductors, dye-sensitized systems, and redox-active polymers, methylene blue provides a molecular template for tuning energy flow within complex chemical architectures. It is another case where a molecule developed long before modern electronics now serves surprising purposes because its structural logic aligns with cutting-edge scientific goals.
The same features that give methylene blue its utility also necessitate responsible handling. In high concentrations or inappropriate clinical situations, the dye can interfere with oxygen transport, mitochondrial function, or neurotransmitter signaling. Because its structure allows rapid cellular penetration and redox interaction, improper exposure may lead to oxidative stress rather than protection. Its charge state influences how it distributes within the body, and patients with certain enzyme deficiencies, such as glucose-6-phosphate dehydrogenase deficiency, cannot safely metabolize it into its reduced form. Such toxicology considerations remind us that molecular power must be applied with precision—compounds that support life in the right context can undermine it in another.
Across all of these fields—diagnostic staining, emergency medicine, microbiology, photodynamic therapy, analytical chemistry, wastewater treatment, electrochemical engineering, and energy science—the explanation for methylene blue’s wide impact always returns to the same principle: the structure and composition of the molecule define its behavior. The tricyclic aromatic system allows strong light absorption and high electron delocalization. The redox-active core enables reversible electron flow. The positive charge permits targeted interactions with biological systems. The chloride counterion provides solubility and mobility in water. These chemical features are not independent attributes; they are interconnected consequences of how atoms are arranged inside a single molecule.
Methylene blue stands as a vivid example of how chemistry creates influence far beyond the molecular scale. A compound synthesized in the nineteenth century became one of the earliest synthetic medicines, one of the foundational biological stains, one of the most important redox dyes in science, one of the earliest photodynamic therapeutics, and one of the modern probes for environmental and materials research. Its longevity across time, disciplines, and technologies highlights the power of molecular architecture in shaping the trajectory of science. Through its structure, methylene blue continues to bridge laboratory chemistry, clinical medicine, life sciences, and technological innovation, demonstrating that the arrangement of atoms determines not only chemical reactivity but also the impact a molecule can have on the world.