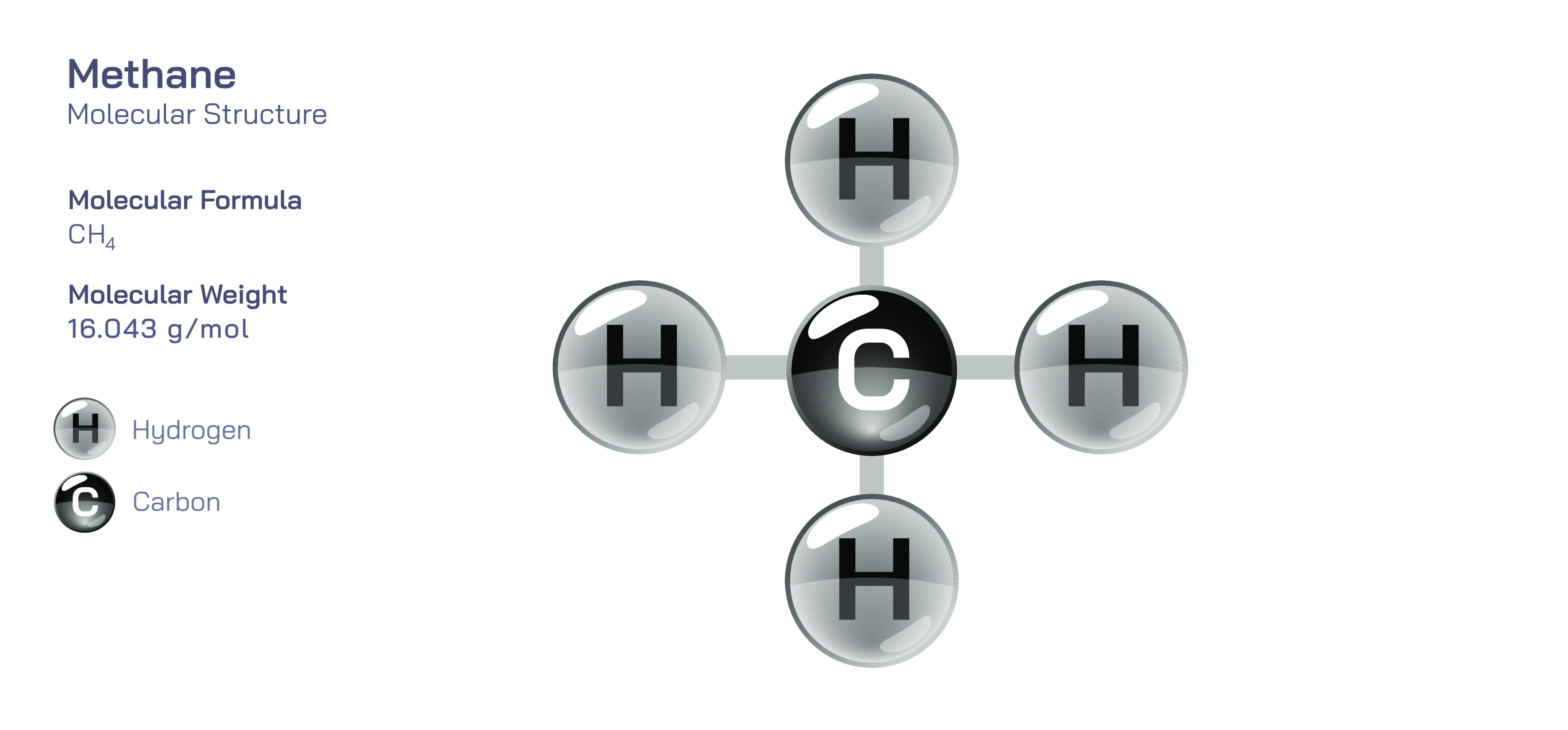
Methane — Molecular Structure and Composition
Methane is one of the simplest molecules in organic chemistry, yet its structure and composition give rise to immense scientific, environmental, industrial, and biological significance. Represented by the chemical formula CH₄, methane consists of one carbon atom at the center bonded to four hydrogen atoms in a precise tetrahedral geometry. Although the formula appears uncomplicated, the spatial organization of atoms in methane and the distribution of electrons around the carbon–hydrogen bonds produce a molecule that behaves with exceptional chemical stability, symmetry, and predictability under a wide range of physical conditions. Each C–H bond is formed by the overlap of sp³ hybrid orbitals from carbon with 1s orbitals from hydrogen, yielding four equivalent bonds oriented toward the corners of an imaginary tetrahedron. This three-dimensional symmetry evens out charge distribution across the entire molecule, leaving methane nonpolar and remarkably resistant to spontaneous chemical reactions in the absence of catalysts or high activation energy. Its compact structural simplicity allows methane to exist as a colorless, odorless gas at standard temperature and pressure, with low solubility in water and high miscibility with other nonpolar or weakly polar gases. Far from being a trivial molecule, methane stands at the core of energy production, earth science, atmospheric chemistry, planetary evolution, and even the search for extraterrestrial life, all rooted in the basic arrangement of four hydrogens around one carbon.
Methane’s prevalence in nature highlights the enduring importance of its structural simplicity. It is the primary component of natural gas and arises from the anaerobic decomposition of organic matter in deep geological formations, wetlands, sediments, permafrost, swamps, and the digestive systems of ruminant animals. In all of these environments, methane is generated when microorganisms called methanogenic archaea break down carbon-based substrates in oxygen-free settings. These microbes complete one of the oldest metabolic pathways on Earth, relying on methane formation as a way to extract biochemical energy from hydrogen and carbon dioxide or from other simple organic molecules. This biogenic methane shows that the tetrahedral CH₄ molecule is not only a product of geological pressure over millions of years but also a living output contained in the metabolic networks of Earth’s oldest evolutionary lineages. The molecule’s chemical stability ensures that once methane is formed, it can persist in trapped reservoirs or natural release zones, rising slowly through rock pores or emerging in cold seeps on ocean floors. Methane hydrates—a unique class of crystalline water–methane systems—trapped inside sediments of ocean margins and polar permafrost further illustrate how the composition of methane enables it to form solid clathrate structures under high pressure and low temperature. These giant methane reservoirs store more carbon than all known fossil fuel deposits on Earth combined and play a major role in planetary carbon cycling and climate history.
Beyond biology, methane influences Earth’s environmental systems in both positive and negative ways due to its molecular behavior in the atmosphere. When released into air, methane is a highly efficient greenhouse gas because its molecular vibrations interact strongly with infrared radiation, trapping heat in the lower atmosphere. Ton for ton, methane warms the planet far more effectively than carbon dioxide over short time intervals, even though it does not remain in the atmosphere as long. This heat-trapping capability arises from the rotational and vibrational energy states permitted by the tetrahedral arrangement of carbon and hydrogen. As methane molecules absorb infrared radiation, they undergo transitions involving bending and stretching motions of the C–H bonds, and these vibrational events delay the escape of thermal radiation into space. Over time, methane is oxidized in the atmosphere, primarily by hydroxyl radicals (·OH), converting first to formaldehyde, then to carbon monoxide, and finally to carbon dioxide and water vapor. Understanding the structural basis of methane’s infrared absorption patterns is crucial for climate science because it helps explain why relatively small concentration increases can contribute significantly to global warming. The molecule that is the backbone of natural gas and biological metabolism is also at the center of one of the defining environmental challenges of the modern world.
Human civilization depends heavily on methane as a fuel source, a dependence that is again rooted in molecular structure. When methane undergoes complete combustion in the presence of excess oxygen, it produces carbon dioxide and water while releasing a large amount of heat. This high energy yield per carbon atom is due to the strong, stable nature of the C–H bonds that store considerable potential energy. Upon ignition, these bonds break and form stronger O–H and C=O bonds in the combustion products, resulting in a net release of energy that powers electricity generation, household heating, industrial kilns, gas turbines, and engines. Methane’s status as the simplest hydrocarbon makes it central to the petrochemical industry as well; steam reforming of methane produces hydrogen—an important reactant in ammonia synthesis, fuel cells, metal refining, and emerging hydrogen economies. Methane can also undergo partial oxidation or catalytic conversion into methanol, formaldehyde, or longer-chain hydrocarbons, demonstrating how its fundamental tetrahedral structure serves as a building block for more complex chemical products. The molecule therefore links microscopic bonding characteristics to macroscopic economic systems, with the energy held inside C–H bonds forming the backbone of industrial heat, electricity, and chemical manufacturing.
Yet methane’s significance stretches beyond Earth. It is a recurring subject in planetary science because its presence in extraterrestrial atmospheres can signal dynamic geological or biological processes. The discovery of methane on Mars has intrigued researchers because the molecule should break down relatively quickly in the Martian atmosphere unless it is being continually replenished. Scientists debate whether Martian methane originates from geological pathways—such as serpentinization reactions between water and ultramafic rocks—or from microbial metabolism that might persist in subsurface environments. On Saturn’s moon Titan, methane is so abundant that it forms lakes, clouds, and rainfall in a methane-based hydrological cycle analogous to Earth’s water cycle. Titan’s methane rivers carve channels, its methane clouds precipitate seasonal weather patterns, and its methane lakes shape a landscape suggesting that simple hydrocarbon chemistry may support complex surface processes across the solar system. In these worlds, methane becomes more than a fuel or greenhouse gas—it becomes a solvent, a climate regulator, and a sculptor of geology. The simplicity of the CH₄ structure allows it to exist as liquid, gas, or solid under extraterrestrial conditions, again demonstrating how the arrangement of atoms controls the behavior of matter on the grandest scales.
Within technology and material science, methane continues to unlock important innovations. Controlled pyrolysis of methane produces solid carbon nanomaterials—nanotubes, graphene flakes, and nanofibers—because the molecule contains exactly the right carbon–hydrogen ratio to release elemental carbon without undesired by-products when processed correctly. Plasma-assisted decomposition and catalytic methane cracking supply high-purity hydrogen alongside nanostructured carbon, supporting research into hydrogen energy systems that minimize carbon dioxide emissions. Even semiconductor technology relies on methane in certain high-precision processes, where methane serves as the carbon source in chemical vapor deposition to create diamond-like coatings used in cutting tools, wear-resistant surfaces, quantum sensing materials, and optical systems. In these applications, methane’s role is not about heat or energy but about atomic precision and purity—its structure allows carbon deposition without the contamination risks associated with larger, more complex hydrocarbons.
Despite its versatility, methane presents a duality in global sustainability thought. As a fuel, it produces significantly less carbon dioxide per unit of energy than coal or oil and releases fewer particulates and pollutants upon combustion, making it a comparatively cleaner energy source. Yet leakage of unburned methane during extraction, processing, and transport can negate these advantages because its greenhouse potency in the atmosphere far outweighs the emissions reductions achieved during combustion. This paradox has reshaped global energy strategies, prompting technological development focused on methane leak detection, pipeline integrity systems, flare efficiency improvements, methane-to-hydrogen conversion facilities, and biological methane capture using methanotrophic bacteria capable of oxidizing methane at ambient temperatures. These innovations are an implicit acknowledgment that methane is not inherently “good” or “bad”; its environmental impact depends not on the molecule itself but on how society interacts with it.
At every level of analysis—from atomic structure, to industrial application, to planetary climate dynamics, to astrobiological speculation—methane demonstrates how the architecture of a molecule governs its destiny. With its four C–H bonds arranged in perfect tetrahedral symmetry, methane achieves remarkable chemical stability while retaining enormous energetic potential. Its composition enables its biological formation in oxygen-deprived habitats, its geological storage in hydrate deposits, its atmospheric presence as a greenhouse gas, its commercial use as a fuel and chemical feedstock, and its planetary role on worlds beyond Earth. The molecule that sits at the foundation of organic chemistry also sits at the crossroads of modern energy policy, environmental science, and the search for life in the universe. In methane, we see a clear reminder that the simplest molecules can exert the most profound influence—and that the behavior of matter at the molecular level shapes both the workings of nature and the trajectory of civilization.