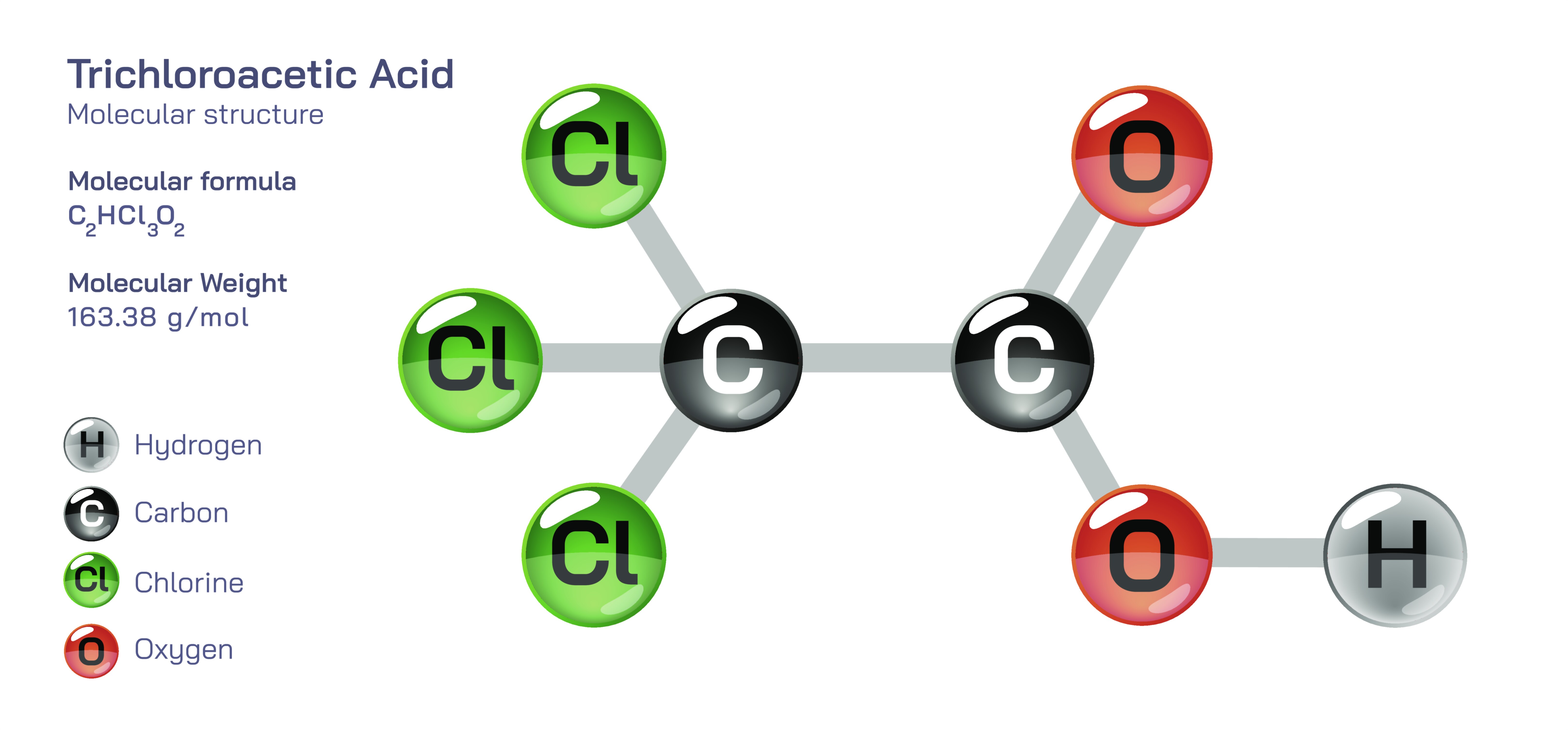
Trichloroacetic Acid – Molecular Structure, Bonding Characteristics, Electron-Withdrawing Effects, Acidity Strength, Reactivity in Organic Chemistry, and Its Educational Importance in Understanding Substituted Carboxylic Acids
Trichloroacetic acid, commonly written as CCl₃COOH, is one of the strongest and most reactive members of the carboxylic acid family, and its molecular structure offers a powerful lesson in how substituents attached to a molecule can dramatically shape its acidity and chemical behavior. Even though trichloroacetic acid shares the same functional group as ordinary acetic acid, the replacement of three hydrogen atoms in the methyl group with three chlorine atoms transforms the entire electron distribution within the molecule. This transformation enhances polarity, increases the electron-withdrawing power of the molecule, and intensifies the dissociation of the acidic proton of the –COOH group. Trichloroacetic acid therefore becomes a highly electrophilic and strongly acidic compound whose structure makes it an excellent educational model for exploring how substitutions on a carbon chain modify acidity, reactivity, solvation, and biochemical interactions.
At the center of the molecular structure is the carboxyl group –COOH, which is responsible for the acidic character of all carboxylic acids. In trichloroacetic acid, this group consists of a carbonyl (C=O) bonded to a hydroxyl (–OH), forming a functional unit capable of releasing a proton in solution. However, what distinguishes this compound from simple carboxylic acids is the presence of a trichloromethyl group (–CCl₃) attached directly to the carbonyl carbon. Each chlorine atom is highly electronegative and powerfully withdraws electron density through the sigma bonds toward itself. Because there are three chlorine atoms acting simultaneously, the electron-withdrawing force on the carboxyl carbon and the oxygen atoms becomes exceptionally strong. This withdrawal stabilizes the conjugate base formed after deprotonation — the trichloroacetate anion — by dispersing negative charge across the structure. Stabilization of the conjugate base is the key reason trichloroacetic acid is far more acidic than acetic acid; the molecular structure is actively encouraging proton release in order to form a more stabilized anion. Students examining this compound quickly understand that acidity is not governed solely by the –COOH group but by the substituents around it and how they influence electron distribution.
The electron-withdrawing effect of the chlorine atoms creates additional structural and energetic consequences. Because of reduced electron density near the carbonyl carbon, trichloroacetic acid exhibits enhanced electrophilicity, making it highly reactive in esterification, substitution, and peptide-cleaving reactions. The polarization of bonds increases hydrogen bonding in solution and raises the dielectric interaction with water, which helps explain why the compound dissolves readily in water despite its halogen-rich hydrocarbon fragment. The molecule’s geometry further supports this polarity: the carbonyl group maintains its usual planar trigonal arrangement, while the three chlorine atoms adopt spatial positions around the central carbon of the trichloromethyl group that maximize electron repulsion while stabilizing the overall structure. This geometry produces a molecule that is compact yet heavily polarized, demonstrating that shape and electronic characteristics are inseparable.
In organic synthesis and biochemical research, trichloroacetic acid serves as a model compound that demonstrates how structural changes modify acidity and reactivity. One of its well-recognized applications occurs in biochemical laboratories, where it is used to precipitate proteins from solution. This effect arises from the extreme acidity of trichloroacetic acid, which protonates and disrupts the charge balance on protein surfaces, causing them to aggregate and separate from aqueous environments. This behavior directly reflects the molecule’s ability to donate protons aggressively and alter electrostatic interactions, a direct outcome of its electron-withdrawing chlorine substituents. Trichloroacetic acid also appears in synthetic chemistry as an activating and oxidizing agent, as well as in the formation of esters and anhydrides. Its unique position between simple organic acids and more complex halogen-substituted intermediates highlights how structure dictates function across different branches of chemistry.
From an educational standpoint, trichloroacetic acid is particularly valuable because it illustrates several fundamental concepts simultaneously: inductive electron withdrawal, resonance stabilization of conjugate bases, the structural determinants of acidity, halogen influence on reactivity, and structure–property relationships. When comparing trichloroacetic acid with acetic acid, students observe firsthand that substituting hydrogens with electronegative atoms dramatically increases acid strength. This makes the compound a keystone example in learning how acidity scales with structural modifications. It also teaches that the behavior of molecules in water depends not only on the presence of polar functional groups but on the overall distribution of electron density throughout the molecule. As learners progress into advanced organic chemistry, the principles visible in trichloroacetic acid become the basis for understanding pharmaceuticals, metabolic pathways, polymer chemistry, and catalysis, where electron-withdrawing or electron-donating substituents reappear repeatedly as tools for modifying molecular effects.
Ultimately, the molecular structure of trichloroacetic acid embodies the essential relationship between structure and reactivity that lies at the heart of chemistry. By taking a familiar functional group and attaching highly electronegative substituents, the molecule transforms into a powerful acid with distinct solvation behavior, reactivity, and industrial significance. Its behavior shows that even simple structural modifications — in this case, replacing hydrogens with chlorine atoms — generate profound consequences at the atomic, molecular, and macroscopic scales. Through this compound, students gain a deeper understanding of how electron flow, substituent effects, and molecular geometry combine to shape the chemistry of acids, making trichloroacetic acid not only an important industrial compound but also a cornerstone of chemical education.