Phenolphthalein Indicator – Chemical Structure, Acid–Base Behavior, Molecular Transition Mechanism, Color Change Properties, and Its Educational Importance in Analytical Chemistry
Phenolphthalein is one of the most widely used acid–base indicators in laboratories because of its dramatic and visually recognizable color change during titrations. Although it appears at first to be a simple dye added to solutions, its behavior is rooted in a fascinating molecular transformation triggered by changes in pH. The structure of phenolphthalein allows it to exist in multiple ionic and resonance-stabilized forms, each of which has distinctly different optical properties. When a solution shifts from acidic to basic, the arrangement of electrons within the molecule changes and alters which wavelengths of light are absorbed and reflected. The result is a sudden shift from colorless to pink or deep fuchsia, providing chemistry students and scientists a highly sensitive visual signal of the exact pH transition point. Understanding how and why this happens demonstrates the importance of functional group chemistry, conjugation, and acid–base equilibria in shaping observable properties of organic molecules.
Chemical Structure and Its Role in Indicator Function
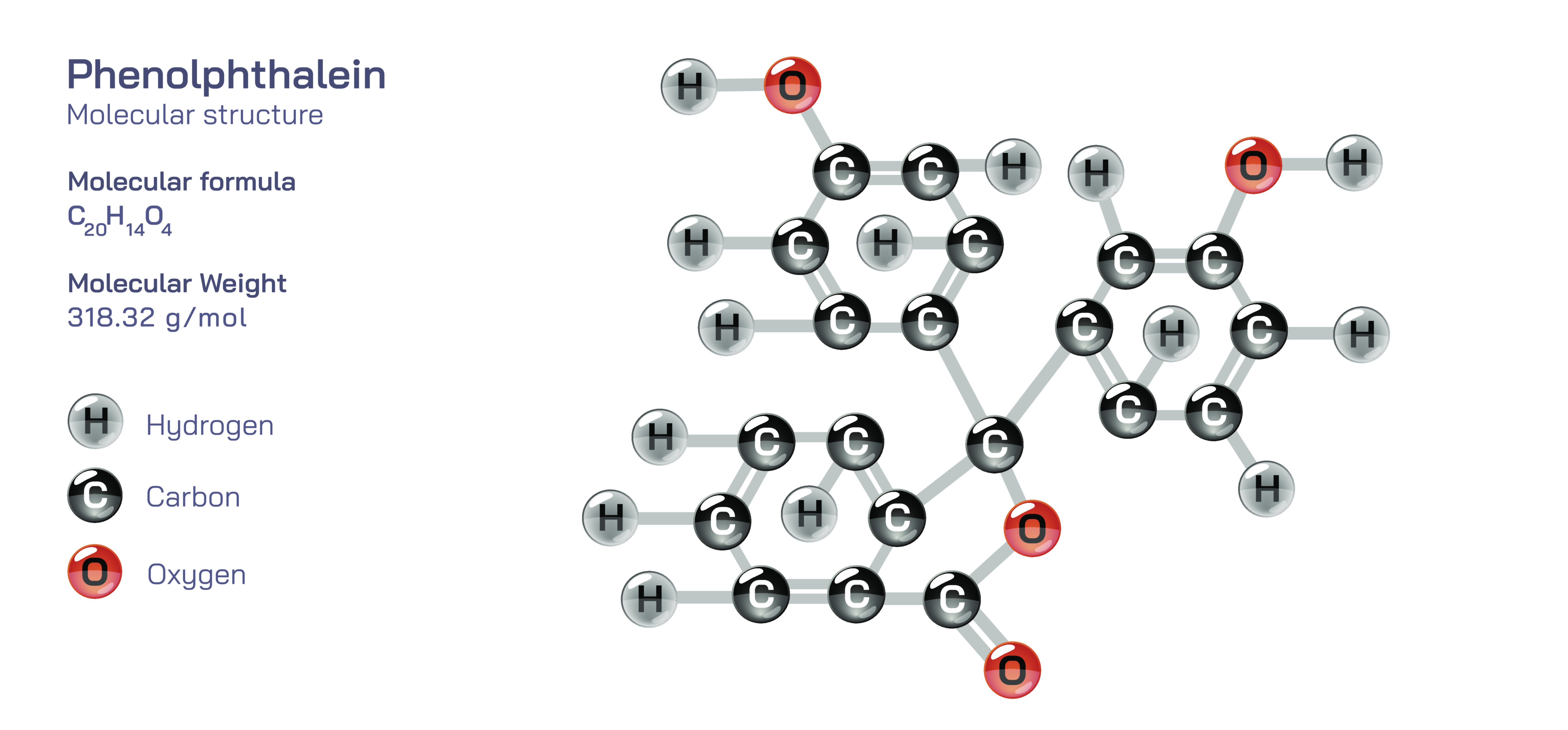
Phenolphthalein is an organic compound composed of three aromatic rings connected through a central carbon atom that binds to a lactone (cyclic ester) group. The molecular formula is C₂₀H₁₄O₄, and its structure includes phenyl groups that interact through extended conjugation — a system in which overlapping π-electrons spread across multiple aromatic rings. This extensive conjugation makes the compound highly sensitive to structural changes, particularly protonation and deprotonation. In acidic solution, phenolphthalein exists primarily in a closed lactone form in which the central ring remains intact. This molecular arrangement does not absorb visible light strongly, causing the solution to appear colorless. However, when the pH increases beyond a certain threshold, the molecule undergoes a transformation that opens the lactone ring and generates a new conjugated structure. This more extended and highly delocalized electronic configuration absorbs light differently, producing the characteristic pink or magenta color.
Color Change Process and pH Range
0. Below pH 8.2 — in acidic and weakly neutral solutions — phenolphthalein remains in its protonated closed-ring form and appears completely colorless. As the solution becomes mildly basic and the pH rises into the transition interval, hydroxide ions extract protons from the molecule, prompting structural rearrangement into the open-ring quinoid form. It is this quinoid structure that absorbs wavelengths in the visible spectrum, reflecting pink. When the solution becomes strongly basic (with very high pH), the molecule deprotonates further and forms a different ionic structure in which the pink color can fade slightly toward a colorless state again due to hyper-deprotonation reducing conjugation. However, under typical laboratory titration conditions, the pink form is stable and reliable for detecting the equivalence point in reactions involving strong bases.
Acid–Base Equilibria and Structural Interconversion
The indicator behavior of phenolphthalein illustrates the concept of acid–base equilibrium at the molecular level. Adding a small amount of indicator to a solution does not alter the pH significantly because only a small fraction of the indicator molecules convert between protonated and deprotonated forms. Instead, the indicator responds to the proton concentration already present, reflecting the state of equilibrium between acidic and basic species. In its protonated (H₂In) form, phenolphthalein is colorless. After losing one proton (HIn⁻), the quinoid form responsible for the pink coloration appears. The equilibrium between these forms depends directly on hydrogen ion concentration. Therefore, phenolphthalein provides a visual signal that corresponds to a precise chemical transition — the moment when the concentration of hydroxide ions overtakes that of hydrogen ions in the titration system. This explains why phenolphthalein is ideal for titrating strong acids with strong bases or weak acids with strong bases, but less suitable for titrating strong acids with weak bases, where the endpoint would fall below its effective color-change interval.
Why Phenolphthalein Appears to Give a “Sudden Change”
In practical laboratory observation, phenolphthalein’s color often seems to appear abruptly rather than gradually. This rapid shift occurs because the titration curve for strong acid–strong base systems climbs steeply in the pH range where phenolphthalein changes color. For many drops of titrant, the solution remains acidic and colorless; then within seconds the pH crosses the transition zone and the pink color locks into the quinoid form. This visual sharpness makes it ideal for identifying the endpoint of titration with high precision. Additionally, the sensitivity of phenolphthalein to structural resonance amplification ensures that even a small proportion of the quinoid form generates a strong visible signal.
Solubility and Use in Laboratory Solutions
Phenolphthalein is only sparingly soluble in water, so laboratory indicator solutions are usually prepared by dissolving phenolphthalein in ethanol, followed by dilution with water. This combination ensures complete dispersion and rapid color response during titrations. The compound is stable under ordinary laboratory conditions but loses color over long exposure to strong light or oxidizing agents. It should be stored in an airtight amber bottle to maintain long-term reliability.
Historical and Biological Context
Phenolphthalein was first synthesized in the late 19th century by German chemist Adolf von Baeyer and quickly became a standard indicator in analytical chemistry. Interestingly, phenolphthalein was once used as a laxative due to its stimulation of intestinal movement, but this medical use has largely been discontinued because long-term consumption raised safety concerns. Today, phenolphthalein remains primarily a laboratory tool and a teaching molecule that helps students visualize equilibrium principles.
Importance of Phenolphthalein in Education and Analytical Science
Phenolphthalein is more than a color-changing dye — it is a molecular demonstration of how structure, resonance, and protonation drive physical properties. Its behavior provides a memorable and intuitive way to understand pH, titration curves, ionic equilibria, and conjugated electron systems. For students encountering organic chemistry and analytical chemistry for the first time, phenolphthalein links the microscopic world of electrons and molecules to macroscopic observations visible to the naked eye. For professionals in chemistry, pharmaceuticals, and environmental testing, it continues to ensure accuracy in laboratory measurements.
Ultimately, phenolphthalein exemplifies a profound scientific truth: molecules act as dynamic systems whose structure and properties transform depending on their chemical environment. The characteristic color change of phenolphthalein — from invisible to vivid pink — reveals the invisible movement of protons and electrons taking place within the solution, turning an abstract molecular process into a clear visual message.