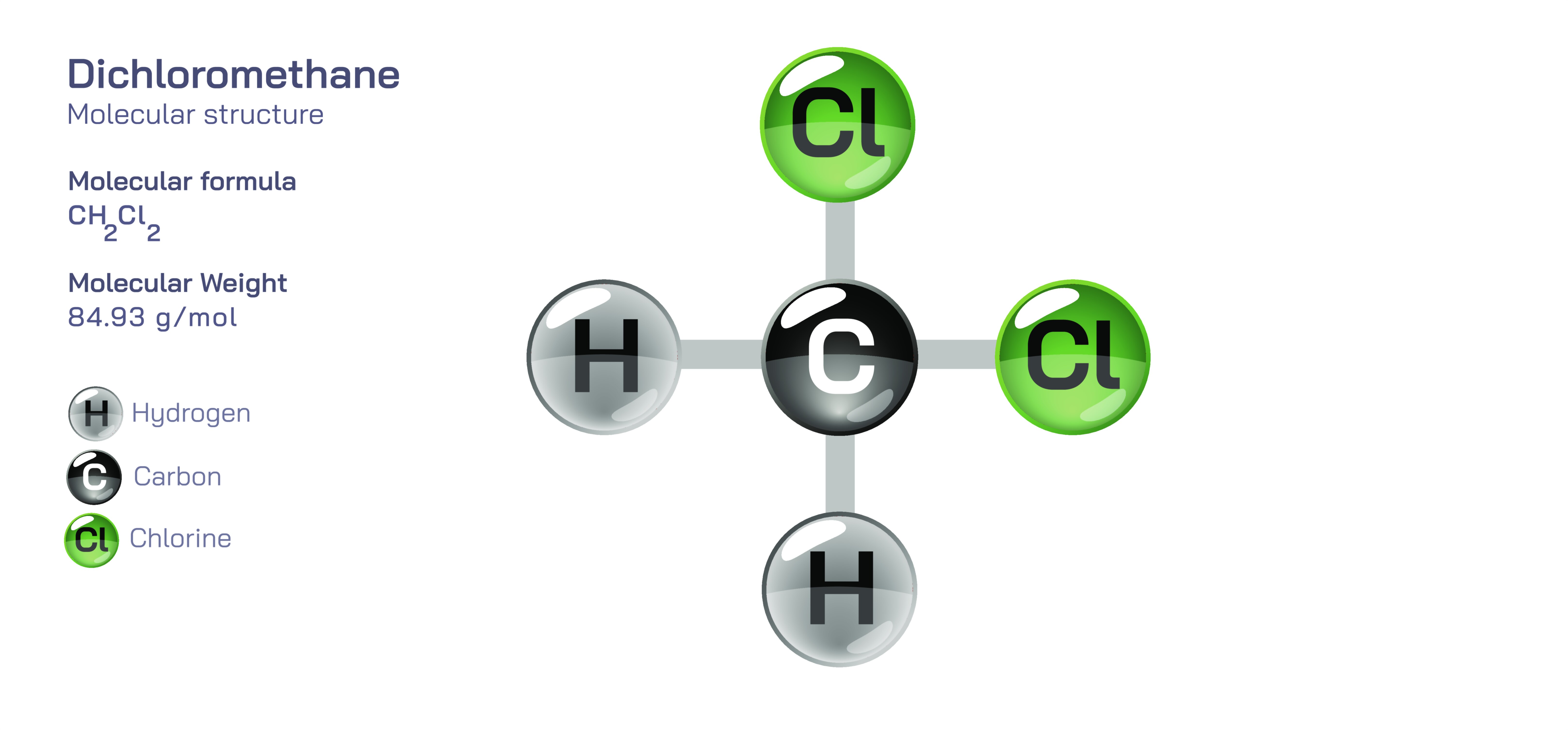
Dichloromethane — Organic Halogenated Solvent
Dichloromethane, commonly known as methylene chloride, occupies a distinctive and influential place in the broad category of halogenated organic solvents, combining volatility, strong dissolving power, and a molecular structure that lends itself to an unusually wide range of industrial, laboratory, and commercial applications. As a chlorinated hydrocarbon, dichloromethane is formed by replacing hydrogen atoms in methane with chlorine, creating a molecule that is stable enough to be handled in controlled conditions yet reactive enough to participate in numerous chemical processes. Its colorless, sweet-smelling, and highly volatile liquid form makes it immediately recognizable to chemists, engineers, and industrial workers. Dichloromethane’s ability to dissolve organic materials while remaining immiscible with water gives it a unique versatility that has made it a staple solvent in extraction, purification, paint stripping, pharmaceutical production, adhesives, coatings, and many other technologies. However, this same versatility is tightly intertwined with concerns about toxicity, environmental impact, workplace exposure, and long-term health effects, making dichloromethane both a powerful tool and a substance requiring rigorous safety management. Understanding dichloromethane demands a careful exploration of its molecular behavior, physical properties, industrial uses, and the growing emphasis on responsible handling in modern science and industry.
At the molecular level, dichloromethane’s structure is simple yet profoundly consequential. It consists of a carbon atom bonded to two hydrogen atoms and two chlorine atoms, creating an asymmetric, polar molecule. This polarity arises because chlorine atoms draw electron density toward themselves more strongly than hydrogen atoms, producing an uneven distribution of charge. The molecule’s moderate polarity makes it an excellent solvent for a wide range of polar and nonpolar substances, allowing dichloromethane to dissolve oils, resins, polymers, organic compounds, and certain inorganic substances. Its molecular weight and structural compactness contribute to its relatively low boiling point, allowing it to evaporate quickly. This rapid evaporation is one of the reasons dichloromethane is widely used in processes requiring the quick removal of solvent without high heat, such as paint stripping, adhesive removal, and thin-film processing. The interplay between polarity, volatility, and low viscosity creates a solvent with an impressive balance of strength, efficiency, and ease of use.
One of the most important roles of dichloromethane is as an extraction solvent. In chemical laboratories and industrial facilities, it is frequently used in liquid–liquid extractions to separate organic compounds from aqueous mixtures. Because dichloromethane is denser than water, it forms the bottom layer in a separation funnel, which allows chemists to easily drain off a purified organic phase containing the dissolved target compound. This density difference, combined with its ability to extract a wide variety of substances, has made dichloromethane a fundamental solvent in organic chemistry. In pharmaceutical production, dichloromethane plays a central role in extracting active pharmaceutical ingredients from reaction mixtures or natural sources, helping purify substances that require careful control of solubility and solvent compatibility. Its ability to selectively dissolve certain compounds while leaving others behind is essential in producing medicines, fragrances, agrochemicals, and fine chemicals. Even though modern green chemistry efforts encourage the search for safer and more sustainable alternatives, dichloromethane remains prominent due to its unique combination of solubility characteristics and ease of separation.
Dichloromethane’s role in paint stripping and coating removal is one of its most well-known industrial uses. When applied to surfaces covered with paint, varnish, or hardened coatings, dichloromethane penetrates the polymer matrix, causing it to swell, soften, and detach from the underlying surface. This makes it highly effective for removing old layers of paint in industrial maintenance, furniture restoration, automotive refinishing, and construction work. However, the same physical properties that make it effective—high volatility, strong solvency, and the ability to penetrate materials—also make it hazardous to workers if proper ventilation, protective equipment, and exposure controls are not maintained. In recent years, regulatory agencies in many countries have restricted or banned consumer-grade paint strippers containing dichloromethane due to accidental poisonings and workplace fatalities linked to improper use. These incidents highlight the need for safe handling protocols and increased public awareness of the risks associated with volatile chlorinated solvents.
Beyond paint stripping, dichloromethane is used extensively in the production of adhesives, particularly those employed in plastics, construction materials, and specialty coatings. It dissolves polymers such as polystyrene, polycarbonate, and acrylic materials, allowing them to be molded, welded, or joined together through solvent bonding. This method of bonding is common in manufacturing plastic components for electronics, automotive parts, household goods, and packaging materials. Dichloromethane evaporates quickly once exposed to air, leaving behind a hardened joint formed by the fused polymer surfaces. Its role in adhesive production also extends to the pharmaceutical and cosmetic industries, where it is used to dissolve components in formulations that require uniform dispersion before solvent removal. The predictable evaporation of dichloromethane ensures that these applications achieve consistent quality and performance.
In analytical and laboratory chemistry, dichloromethane is widely used for chromatography, spectroscopy sample preparation, and film casting. Because it dissolves a broad range of organic molecules, it is often used to prepare samples for analysis by gas chromatography, mass spectrometry, infrared spectroscopy, and other instrumental techniques. In polymer science, dichloromethane helps researchers cast thin films by dissolving a polymer and then allowing it to spread and dry on a surface as the solvent evaporates. These films are used for testing material properties, fabricating membranes, studying optical behavior, or preparing electronic materials. The solvent’s ability to evaporate evenly and quickly is crucial for forming uniform films without bubbles or defects, making it a preferred choice in research involving polymers and nanomaterials.
Another important area where dichloromethane plays a significant role is pharmaceutical formulation, especially in the production of microcapsules, nanoparticles, and controlled-release drug systems. Many of these technologies rely on solvent evaporation or phase separation techniques in which dichloromethane dissolves a polymer that later solidifies around a drug as the solvent leaves the system. Its volatility and miscibility with key organic components make it suitable for encapsulation processes that require careful control of particle size, coating thickness, and drug distribution. Although these applications provide tremendous medical benefits, ensuring that trace levels of dichloromethane do not remain in finished pharmaceutical products is essential. Strict purification procedures and regulatory limits ensure that any residual solvent meets safety standards before a medication can be approved for use.
The toxicological profile of dichloromethane is complex and requires careful consideration. Although not immediately corrosive, dichloromethane poses serious health risks through inhalation, skin contact, or improper handling. The solvent can depress the central nervous system at moderate concentrations, causing dizziness, headaches, confusion, and impaired coordination. At higher exposures, it can cause unconsciousness or death. One of the most concerning aspects of dichloromethane is that the body metabolizes part of it into carbon monoxide, a toxic gas that binds to hemoglobin and reduces the blood’s ability to carry oxygen. This metabolic pathway increases the danger of exposure, especially in confined spaces or poorly ventilated environments. Long-term exposure has been associated with elevated risks of certain cancers, leading several health agencies to classify dichloromethane as a potential carcinogen. These concerns have prompted regulatory bodies to establish strict workplace exposure limits, promote protective gear, and encourage the development of safer alternatives where feasible.
Environmental considerations also influence the use of dichloromethane. Although it does not persist as long as some other halogenated solvents, dichloromethane does contribute to environmental pollution when released into air, water, or soil. Its volatility means that most releases occur into the atmosphere, where it can remain for months before breaking down. Some of its degradation products may participate in atmospheric reactions that influence air quality, although it does not have the same long-term ozone-depleting potential as older chlorinated compounds like chlorofluorocarbons. In water systems, dichloromethane can harm aquatic life if present in significant concentrations, and improper disposal from industrial facilities can contaminate groundwater. Modern environmental regulations require industries to capture, recycle, or neutralize dichloromethane emissions, and many companies now employ closed-loop systems designed to minimize environmental impact. These efforts reflect a broader trend in chemical industries toward sustainability and responsible solvent management.
Despite these challenges, dichloromethane remains deeply embedded in the infrastructure of scientific and industrial processes. Its ability to dissolve diverse materials, its physical characteristics, and its compatibility with numerous chemical reactions make it difficult to replace in many situations. Alternative solvents often lack the same combination of volatility, solubility, and chemical stability. Nevertheless, researchers continue to explore greener substitutes and develop safer technologies that reduce reliance on dichloromethane. The evolution of solvent usage reflects a broader transformation in chemical sciences as industries strive to balance performance with environmental responsibility and worker safety.
Conceptually, dichloromethane serves as an important example of how molecular structure dictates behavior. The presence of chlorine atoms not only alters the polarity and solvency of the molecule but also influences its toxicity, stability, and environmental impact. Its moderate polarity creates a solvent that bridges the gap between nonpolar hydrocarbons and highly polar solvents like alcohols or water, allowing it to serve as a universal medium for countless reactions and processes. Its volatility demonstrates how intermolecular forces shape phase behavior. Its industrial relevance illustrates how chemistry permeates the modern world through manufacturing, purification, construction, and analysis. At the same time, its hazards remind us that the power of chemical substances comes with responsibilities—understanding risks, enforcing safety standards, and ensuring that benefits do not overshadow potential harms.
Ultimately, dichloromethane represents both the promise and the complexity of chemical technology. It embodies how a simple molecular structure can unlock vast possibilities while simultaneously requiring careful control and informed judgment. Through dichloromethane, one can appreciate the intersection of chemistry, engineering, safety science, environmental stewardship, and modern innovation, gaining insight into how solvents shape the processes and products that define contemporary life.