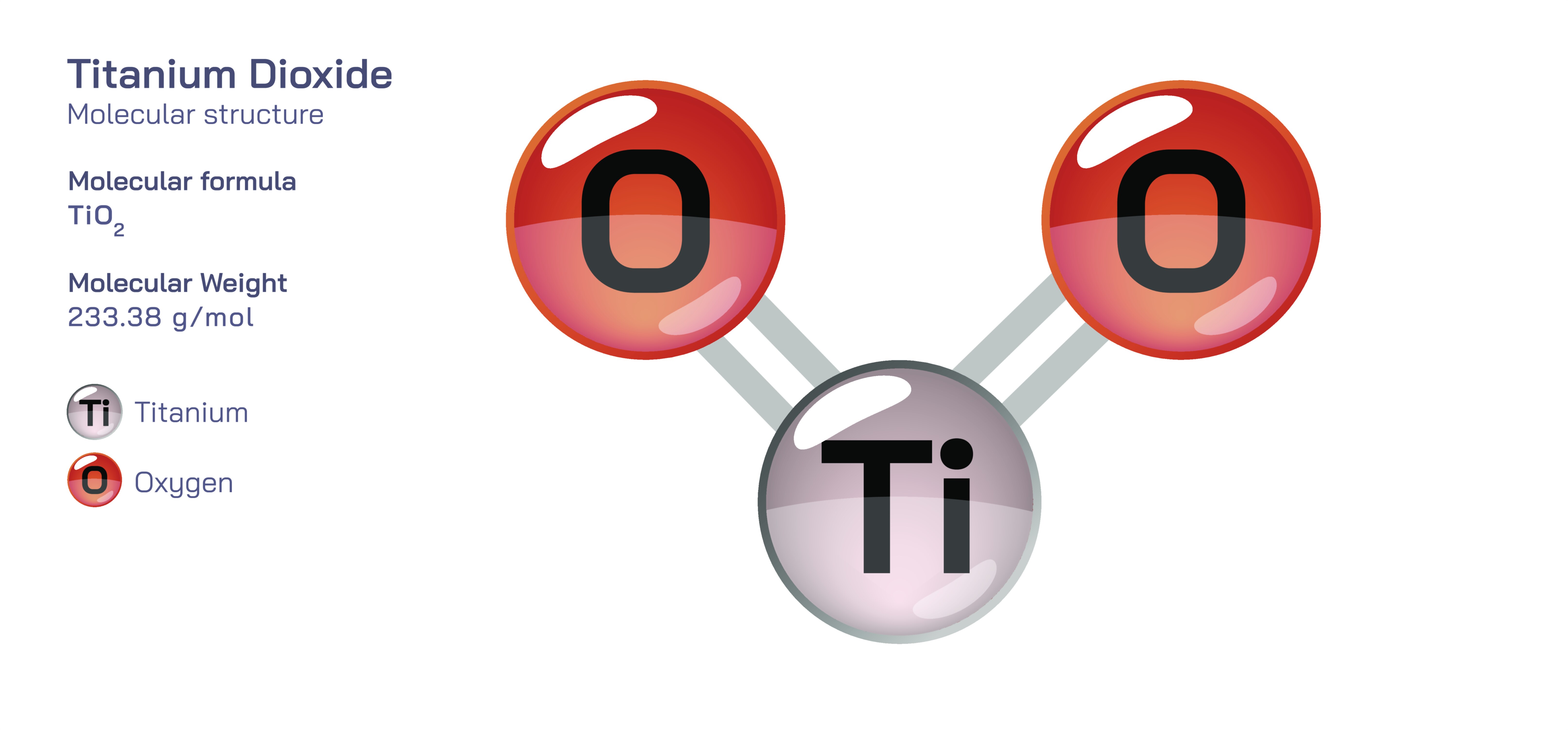
Titanium Dioxide – Molecular Structure, Crystallographic Arrangement, Bonding Characteristics, Electronic Behavior, Surface Chemistry, and Its Transformational Role in Modern Materials Science and Industrial Chemistry
Titanium dioxide, widely known by its chemical name TiO₂, is one of the most scientifically and industrially influential inorganic compounds due to its unique molecular structure and the extraordinary physical and chemical properties that arise from that structure. To understand why this compound is so valuable across fields ranging from nanotechnology and renewable energy to pigments and cosmetics, it is essential to start with its atomic-scale architecture. Titanium dioxide is composed of titanium atoms bonded to oxygen atoms in a rigid network that forms extended crystalline structures rather than discrete molecules. Even though its empirical formula appears simple, the arrangement of atoms in space and the nature of bonding determine how the compound behaves when interacting with light, electrons, energy, and chemical environments. TiO₂ exists primarily in three crystalline polymorphs — rutile, anatase, and brookite — each sharing the same chemical composition but differing in the geometric arrangement of titanium and oxygen atoms. These variations in structure control optical reflectivity, photocatalytic activity, stability, electronic band gap, and surface behavior, demonstrating that a chemical formula alone cannot capture the complexity of solid-state materials.
The most stable and widely occurring polymorph of titanium dioxide is rutile. In this structure, each titanium atom is surrounded by six oxygen atoms arranged in a distorted octahedral coordination, forming a compact tetragonal lattice in which atoms pack efficiently and maximize structural density. The distortions slightly elongate certain titanium–oxygen distances, producing subtle variations in bond strength that have major consequences for the rigidity and refractive properties of the crystal. This dense atomic arrangement creates a material exceptionally resistant to thermal and chemical attack, explaining why rutile TiO₂ survives extreme conditions such as high heat, UV radiation, and exposure to reactive substances. Its ability to reflect and scatter visible light strongly — owing to the high refractive index of its crystal lattice — makes rutile the dominant white pigment in paints, plastics, and coatings. The intense brightness and opacity of rutile have replaced toxic white pigments such as lead carbonate, linking crystallography directly to public health and safety.
Anatase, another major polymorph of titanium dioxide, contains the same octahedral titanium–oxygen coordination as rutile but in a more open and less densely packed arrangement. This modified connectivity results in greater surface energy, increased photocatalytic reactivity, and a wider electronic band gap compared with rutile. Anatase tends to form nanoscale crystals that expose a higher proportion of surface atoms relative to total volume, making their external surfaces unusually chemically active. When anatase absorbs ultraviolet light, electrons in the lattice become energetic enough to jump into conduction states, leaving behind positively charged vacancies known as holes. These electrons and holes migrate to the surface where they interact with water, oxygen, or organic molecules, generating highly reactive species capable of breaking down pollutants and destroying microorganisms. The photocatalytic efficiency of anatase has transformed TiO₂ into a key component of self-cleaning surfaces, air-purifying coatings, and wastewater treatment technologies. It also gives the compound a central role in dye-sensitized solar cells, where the movement of electrons through anatase nanoparticles converts sunlight into electricity, highlighting how molecular geometry becomes the basis of alternative energy strategies.
Brookite, the least common natural polymorph of titanium dioxide, has an orthorhombic crystal structure that differs significantly from rutile and anatase. Although it shares the octahedral coordination around the titanium atom, the three-dimensional connectivity of these octahedra produces a more complex and less symmetrical lattice. Brookite exhibits strong interactions between atomic layers, generating high surface charge density and unique electronic dynamics that researchers continue to study. While brookite has not found the same scale of industrial use as rutile and anatase, it plays a major role in advanced research related to heterogeneous catalysis, composite materials, and hybrid polymorph mixtures where combining multiple TiO₂ phases increases photocatalytic efficiency beyond what any single phase can achieve. This points to a central concept in materials science: subtle differences in lattice geometry can lead to dramatic differences in physical performance.
The chemical bonding within TiO₂ governs many of the properties that make it indispensable. Titanium exists in the +4 oxidation state and oxygen in a −2 oxidation state, and the electrostatic attraction between the positively charged titanium centers and the negatively charged oxygen atoms forms strong ionic character within the lattice. However, the overlap of titanium d-orbitals with oxygen p-orbitals also contributes a significant covalent character, producing robust directional bonding that stabilizes the three-dimensional network. This hybrid nature of bonding — neither purely ionic nor purely covalent — explains why TiO₂ behaves as a semiconductor rather than a simple salt. The electrons in titanium dioxide occupy energy levels that, though tightly bound, can be excited into higher-energy conduction states under ultraviolet illumination. This balance of rigidity and electronic flexibility allows TiO₂ to withstand corrosive conditions while also interacting dynamically with light and charge carriers.
As technology moves deeper into the nanoscale, the surface chemistry of titanium dioxide has become one of its most intriguing features. At the atomic surface, titanium atoms often lack the full coordination they experience in the bulk lattice, leaving them highly reactive toward adsorbing water, ions, organic molecules, or oxygen species. In aqueous environments, surface oxygen vacancies — locations where an oxygen atom is missing — dramatically increase reactivity and govern how ions and molecules bind at the surface. This environment becomes a platform for catalytic reactions where pollutants are decomposed, dyes are broken down, and fuel intermediates are oxidized or reduced. In many photocatalytic systems, TiO₂ does not serve as a reactant that is consumed; instead, it acts as a catalyst that accelerates chemical change through electron transfer. These catalytic reactions demonstrate how the marriage of light absorption and surface bonding dynamics make TiO₂ an unparalleled tool for environmental and industrial chemistry.
Beyond environmental applications, titanium dioxide affects daily life and consumer products through its optical properties. Its high reflectivity across the visible spectrum creates exceptional whiteness and brightness, and its UV-blocking capability makes it a standard ingredient in sunscreens and cosmetic formulations. The molecular structure of TiO₂ determines how it interacts with light — its crystal lattice scatters visible wavelengths while strongly absorbing ultraviolet radiation. In sunscreen, nanosized anatase and rutile particles protect skin by absorbing and redirecting harmful UV light without penetrating the skin barrier. In paints and coatings, TiO₂ prevents degradation of pigments and polymers by blocking photochemical pathways initiated by UV exposure. This makes the compound essential not only for visual appearance but for material longevity, durability, and safety.
Even with its widespread use, titanium dioxide continues to stand at the center of cutting-edge scientific research. Scientists are investigating how doping TiO₂ with metals and nonmetals changes its band structure, enabling it to absorb visible light rather than only ultraviolet light. This shift could revolutionize solar energy and environmental cleanup by allowing the compound to harvest a larger portion of the solar spectrum. The ability of TiO₂ to interface with biological tissues without substantial toxicity has led to its incorporation in biomedical implants and drug delivery systems, where its surface chemistry promotes cell adhesion and long-term stability. Researchers are also working to combine rutile, anatase, and brookite into engineered composites because mixtures of polymorphs improve charge separation and extend photocatalytic lifetime, revealing that the future of titanium dioxide lies not in a single crystal structure but in the synergy between multiple forms.
Ultimately, the molecular structure of titanium dioxide showcases the intricate interplay between atomic arrangement and macroscopic properties. Whether functioning as a semiconductor, UV absorber, pigment, catalyst, or nanoscale electronic component, TiO₂ derives its performance from the geometric and electronic framework built from titanium and oxygen atoms. Its continued impact on modern science demonstrates how the study of solid-state chemistry opens doors to sustainable energy, environmental purification, materials engineering, and human health. In this single compound, the connection between crystallography, bonding, light–matter interactions, and industrial importance becomes undeniably clear — showing that understanding structure at the molecular level is essential to shaping the future of chemistry and technology.