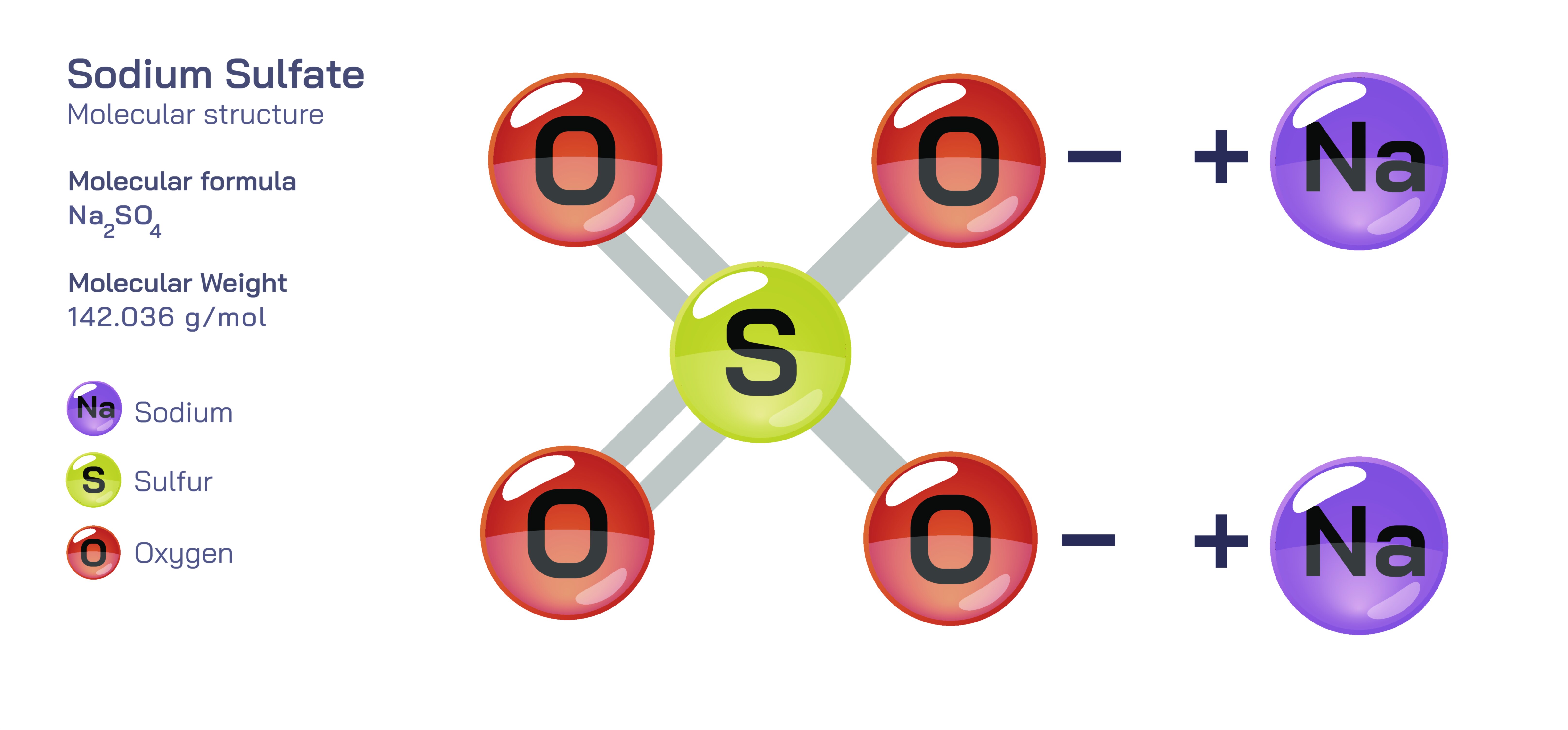
Sodium Sulfate – Molecular Structure, Chemical Characteristics, and Educational Overview
Sodium sulfate is an inorganic compound that plays a significant role in industrial chemistry and aqueous ionic interactions, and its molecular structure offers an excellent foundation for understanding ionic bonding and salt chemistry in general. When studying this compound, it becomes clear that its structure and properties arise primarily from the electrostatic forces that link sodium cations and sulfate anions, creating a stable configuration that remains consistent across a variety of physical states. The compound is commonly represented by the formula Na₂SO₄, reflecting the presence of two monovalent sodium ions associated with a single sulfate ion, and this numerical arrangement provides insight into how charge balance works when cations and anions form neutral chemical substances. Each sodium ion carries a positive charge, while the sulfate group possesses an overall negative charge made possible by the tetrahedral orientation of oxygen atoms around a central sulfur atom, giving the complete compound a predictable stoichiometry and arrangement. Understanding sodium sulfate’s structure becomes particularly enriching when students visually imagine the tetrahedral geometry of the sulfate anion surrounded by sodium ions attracted to the electron-rich oxygen atoms, forming a stable ionic lattice in the solid crystalline state.
Delving deeper into the molecular geometry, the sulfate anion consists of a sulfur atom centrally bonded to four oxygen atoms in a symmetric tetrahedral shape, and this arrangement creates a strong and evenly distributed electronic environment that contributes to the stability of the anion. The sodium ions do not form directional bonds with the sulfate group like covalent species do; instead, they electrostatically interact with the negatively charged oxygen atoms, and the resulting ionic network builds extensive crystalline structures that reflect orderly long-range patterns. These geometric and electronic influences shape many of the physical properties of sodium sulfate, including its high melting point, good solubility in water, and well-defined transition forms under different temperature conditions. When dissolved in water, the sodium ions disperse freely through the solvent, while the sulfate ion separates and becomes fully solvated, illustrating the behavior of dissociation and hydration shells that students often encounter when studying electrolyte interactions in aqueous solutions. This makes sodium sulfate a useful demonstration compound for lessons involving ionic dissolution, conductivity, and precipitation reactions, because it consistently behaves in line with theoretical expectations for strong electrolytes.
Beyond structure and geometry, sodium sulfate serves as an excellent educational example for understanding polymorphism, where the same chemical composition can exist in different structural forms depending on environmental conditions such as temperature. The most widely known hydrated form is decahydrate sodium sulfate, sometimes called Glauber’s salt, which incorporates water molecules into its crystalline lattice. Heating this hydrated form releases the associated water and produces anhydrous sodium sulfate, reinforcing concepts of hydration, crystallization, and reversible thermal transformations in salts. These transitions illustrate how the molecular arrangement adapts to physical changes, altering density, crystalline shape, and thermal behavior without reorganizing the fundamental ionic bonding scheme. Students exploring thermal chemistry, phase transitions, or thermodynamics can therefore gain a clear example of how energy input modifies chemical systems without requiring chemical decomposition or redox processes.
The chemical behavior of sodium sulfate further provides an excellent case study for understanding spectator ions in aqueous reactions, since it plays a passive role in many processes. The sodium and sulfate ions do not readily participate in redox changes or form precipitates with most common ions, and for that reason the compound is often used in controlled experiments requiring neutral ionic background conditions. It demonstrates how some ionic species remain stable and unchanged even when introduced into complex reaction environments, providing a comparison point for ions that are much more chemically active. Teachers and students often employ sodium sulfate in double-displacement reaction experiments to evaluate solubility rules, where most products involving sodium or sulfate remain soluble, thus helping to reinforce solubility predictions and dissolution patterns. By practicing with known nonreactive ions, learners gain confidence before exploring compounds that yield more complex or unexpected products.
From an industrial and environmental perspective, sodium sulfate also enriches chemistry education by showing how molecular structure influences technological and natural applications. The predictable dissolution and re-crystallization properties make it useful for detergents, glass manufacturing, and pulp processing, and these applications highlight how characteristics arising from molecular structure scale into real-world utility. Water treatment processes sometimes involve sodium sulfate indirectly, as its ions can adjust ionic strength without creating unwanted precipitates, thus demonstrating how controlled ion behavior contributes to industrial scalability and environmental decision making. The compound’s stability and resistance to oxidation or reduction enable its safe handling and long-term storage in laboratory environments, and its relatively low cost makes it highly accessible for classroom and research use, further encouraging its role in education.
For learners focusing on chemical bonding, sodium sulfate demonstrates the complementary roles of ionic and covalent interactions within a single species. The sodium–oxygen attraction represents classic ionic bonding behavior, formed through electrostatic attraction between charged species, while the sulfur–oxygen connections inside the sulfate anion remain covalent in nature despite the overall negative charge of the group. This dual-bonding arrangement helps students distinguish between bond types within polyatomic ions and reinforces the idea that covalent bonds can exist inside an ion that behaves overall as a charged unit in ionic compounds. As a teaching topic, sodium sulfate bridges multiple conceptual pillars in chemistry: ionic theory, solubility, structural geometry, crystallography, aqueous behavior, phase transition, and industrial practice. It supports conceptual learning by remaining consistent and predictable across a wide range of experiments, a quality that strengthens theoretical lessons and encourages hands-on exploration.
Ultimately, studying sodium sulfate from a molecular perspective encourages an integrated understanding of how chemical structure determines macroscopic properties and experimental behavior. Every characteristic—from its crystalline lattice to its dissolution pattern—reflects the fundamental interplay of charges, geometry, and intermolecular forces, making the compound a remarkably clear representative of foundational chemistry principles. Whether used in classrooms, laboratories, or industrial environments, sodium sulfate consistently reinforces key concepts in chemical education and provides an approachable yet intellectually rich subject for learners seeking to deepen their understanding of how atoms and ions assemble into stable compounds that shape both scientific theory and practical everyday applications.