Sulfur Hexafluoride – Molecular Structure, Bonding Geometry, Electron Distribution, Physical Properties, Inert Chemical Behavior, Environmental Impact, and Educational Importance in Inorganic Chemistry
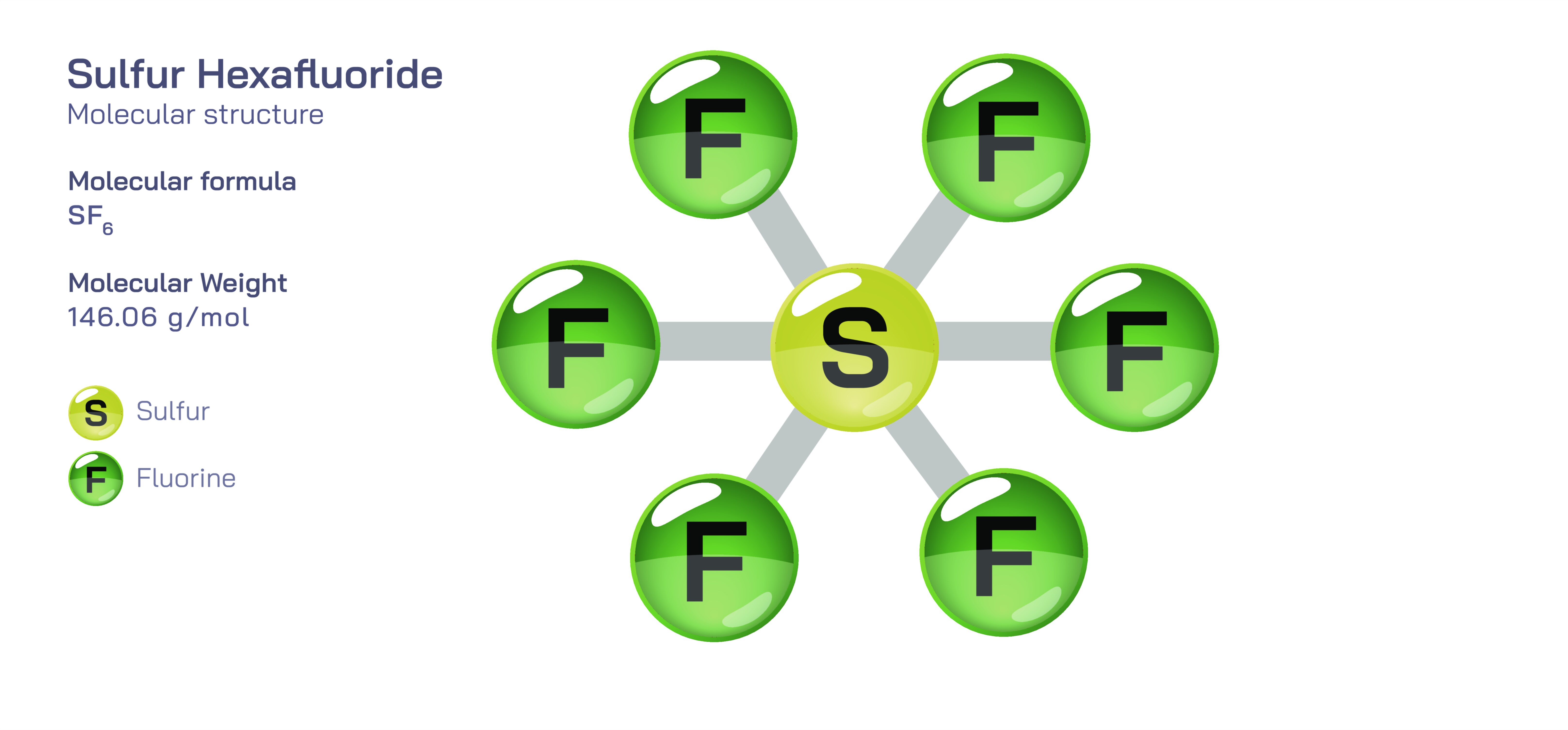
Sulfur hexafluoride, written chemically as SF₆, is a remarkable and widely studied compound because it highlights several core principles of molecular structure and provides a clear demonstration of how electron arrangement and geometry shape a molecule’s macroscopic behavior. At first glance, SF₆ may seem simple—one sulfur atom bonded to six fluorine atoms—but its structural precision produces physical and chemical characteristics that make it unique among covalent compounds. For chemistry learners, sulfur hexafluoride becomes a powerful example of the connection between atomic design and real-world properties, illustrating concepts ranging from valence shell expansion and octahedral geometry to molecular symmetry, polarity, electronegativity effects, and chemical inertness.
The central structural feature of sulfur hexafluoride is its octahedral molecular geometry. One sulfur atom occupies the center of the molecule, and six fluorine atoms arrange themselves symmetrically around it, each positioned at the corners of an octahedron. The S–F bonds extend outward in six equivalent directions, giving the molecule perfect three-dimensional symmetry. This geometry is predicted by valence shell electron pair repulsion (VSEPR) theory, which states that electron regions around a central atom will arrange themselves as far apart as possible to minimize repulsion. In SF₆, there are six bonding electron pairs and no lone pairs on the sulfur atom, making octahedral geometry the most stable configuration. This atom-centered symmetry is not merely geometric elegance—it determines how the molecule interacts with electric fields, radiation, and surrounding matter.
One of the most educational aspects of SF₆ is that sulfur forms six equivalent covalent bonds, exceeding the conventional “octet rule” taught for second-period elements. While carbon, nitrogen, and oxygen are constrained to eight valence electrons, sulfur belongs to the third period of the periodic table and can expand its valence shell. Whether this expansion literally uses 3d orbitals is debated in modern quantum chemistry, but the molecule undeniably accommodates 12 electrons around sulfur because of accessible electron-delocalized bonding models. SF₆ therefore becomes a key teaching example showing that heavier main-group elements can form stable structures that do not follow the simple octet rule. Students encountering SF₆ deepen their understanding that bonding models must be flexible enough to describe elements beyond the second period.
Although each S–F bond is highly polar (because fluorine is significantly more electronegative than sulfur), sulfur hexafluoride as a whole is nonpolar. This result often surprises learners until they consider the molecular symmetry. The equal and opposite pull of each fluorine on the central sulfur atom cancels vectorially, producing no net dipole moment. This cancellation illustrates a core principle in molecular chemistry: polarity depends not only on the polarity of individual bonds but also on their spatial arrangement. Because the bonds are symmetrically distributed, the molecule remains electronically balanced in all directions. This concept helps students distinguish between bond polarity and molecular polarity and is foundational for predicting physical properties such as solubility and intermolecular forces.
The physical properties of SF₆ derive directly from its structural symmetry and strong covalent bonding. The molecule is extremely stable, chemically inert, non-flammable, and thermally robust. It does not react easily with acids, bases, metals, or oxidizing agents under normal conditions because the sulfur atom is already in one of its most oxidized and energetically stable states. The large size and spherically symmetric electron distribution create weak intermolecular attractions, allowing SF₆ to behave as an almost ideal gas. The gas is also exceptionally dense—about five times heavier than air—because it contains heavy elements and has a compact molecular structure. Demonstrations often show SF₆ filling a container like a “liquid-like gas,” allowing lightweight objects to float on it, a striking educational display that underscores the relationship between molecular mass and gas density.
Another property rooted in its electronic structure is SF₆’s excellent dielectric strength and electrical insulating ability. When an electric field is applied, the symmetrical electron distribution resists polarization, preventing electrical breakdown. For this reason, sulfur hexafluoride has been widely used in high-voltage electrical systems, transformers, circuit breakers, and gas-insulated switchgear. These technological applications trace back directly to fundamental molecular features: strong covalent bonds, perfect symmetry, minimal polarity, and chemical stability.
Despite its many useful properties, sulfur hexafluoride also plays a significant role in environmental chemistry, helping students appreciate how molecular characteristics scale to global consequences. SF₆ is an extremely potent greenhouse gas, with a global warming potential many thousands of times greater than carbon dioxide and with an atmospheric lifetime that can exceed thousands of years. Its strength comes from the same features that make it stable and non-reactive: the molecule does not break down easily in the atmosphere, does not dissolve efficiently in water, and does not photodecompose readily under sunlight. Thus, while SF₆ is non-toxic and safe in controlled environments, its long atmospheric persistence emphasizes how beneficial chemical stability in industry can create challenges for environmental sustainability. This reinforces an important message: evaluating a molecule requires both microscopic chemical understanding and macroscopic ecological responsibility.
In educational chemistry, sulfur hexafluoride brings together multiple lessons:
It demonstrates how VSEPR theory predicts three-dimensional structure.
It highlights valence shell expansion beyond the octet rule for heavier elements.
It exemplifies the difference between bond polarity and overall molecular polarity.
It shows how symmetry governs intermolecular forces and macroscopic properties.
It illustrates how molecular stability enables technological usefulness yet requires environmental caution.
Beyond memorizing formulas, students gain insight into how molecular architecture shapes real-world outcomes. The octahedral symmetry of SF₆ explains its inertness, the inertness explains its industrial value, and the same inertness explains why it persists in the atmosphere. This chain of reasoning turns chemistry from abstract symbolism into a unified science that explains the world at every scale.
Ultimately, sulfur hexafluoride demonstrates one of the most important principles in molecular science:
structure determines properties, and properties determine applications and consequences.
By studying SF₆ in depth, learners not only understand a single compound but also develop analytical skills that apply to advanced inorganic chemistry, materials science, environmental science, and industrial chemical design.