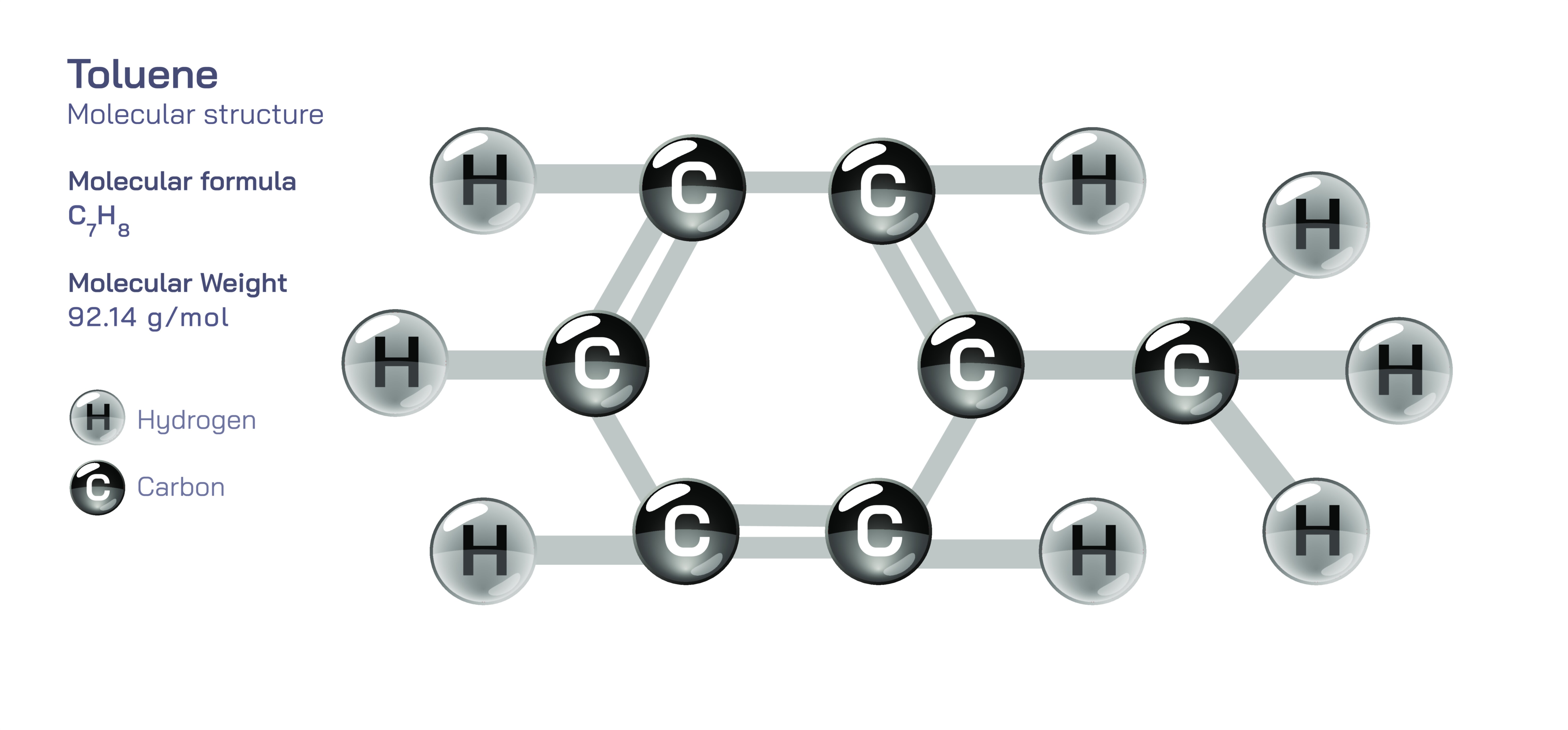
Toluene – Molecular Structure, Bonding Behavior, Aromatic Ring Characteristics, Substitution Patterns, Industrial Importance, and the Educational Value of Understanding Aromatic Hydrocarbon Chemistry
Toluene represents one of the most fundamental aromatic compounds in organic chemistry, and studying its molecular structure provides deep insight into how aromaticity, electron distribution, and chemical substituents govern the reactivity and behavior of hydrocarbons. With the molecular formula C₆H₅CH₃, toluene consists of a six-membered benzene ring bonded to a single methyl group. Although the addition of a methyl substituent might appear subtle at first, it introduces changes that distinguish toluene from benzene in both physical properties and chemical behavior. The methyl group delivers electron-releasing effects to the aromatic ring, amplifying electron density and enhancing the compound’s ability to undergo reactions typical of electrophilic aromatic substitution. Because of this, toluene becomes a valuable instructional bridge between simpler aromatic systems like benzene and more complex substituted aromatic molecules that populate pharmaceutical chemistry, dyes, polymers, and fuel components. Understanding toluene’s structure therefore equips chemistry learners with foundational knowledge that extends into applied and industrial chemistry.
At the molecular level, the defining feature of toluene is its benzene ring, which consists of six carbon atoms arranged in a planar hexagon with alternating single and double bonds. However, because of resonance — a phenomenon where electron density is delocalized across all six carbon atoms — the ring does not actually contain discrete single and double bonds but rather six equivalent carbon-to-carbon bonds of intermediate character. This resonance stabilization produces the unique concept of aromaticity, a hallmark of compounds containing a cyclic, conjugated π-electron system that follows Hückel’s rule. The benzene ring in toluene maintains its flat geometry to allow full π-orbital overlap, ensuring that electrons circulate throughout the ring rather than remaining localized. This aromatic electron cloud creates stability, prevents typical addition reactions that would disrupt aromaticity, and directs the ring toward substitution pathways that preserve the delocalized electron system. Thus, even before considering the methyl group, the aromatic component of toluene establishes a strong structural and energetic framework that determines the compound’s behavior.
The methyl group attached to the benzene ring distinguishes toluene structurally from benzene in a way that has profound consequences. By containing carbon–hydrogen σ-bonds capable of hyperconjugation — an effect where electrons from σ-bonds interact with the π-system of the ring — the methyl group acts as an electron-donating substituent. This donation increases electron density within the aromatic ring, making toluene more reactive than benzene toward electrophilic species. Electrophilic aromatic substitution reactions such as nitration, sulfonation, halogenation, and Friedel–Crafts alkylation or acylation occur more readily with toluene because the methyl substituent activates the ring electronically. It also influences orientation by directing incoming substituents toward the ortho (positions adjacent to the methyl group) and para (position opposite the methyl group) sites, rather than the meta positions. This directing effect is central to understanding how functional groups influence the regiochemistry of aromatic reactions, and toluene serves as a model that educators frequently use when explaining substituent effects in organic chemistry. The behavior reveals a major pedagogical theme: not all aromatic rings behave the same, and small structural variations can shift reactivity dramatically.
The three-dimensional structure of toluene remains largely planar in the region of the benzene ring, but the methyl group can rotate freely around the carbon–carbon bond without disturbing aromaticity. This rotational freedom does not significantly change chemical reactivity because the benzene ring’s electron distribution dominates the compound’s behavior. However, methyl substitution subtly affects physical properties. Toluene is a colorless liquid at room temperature with a characteristic sweet odor, a boiling point of approximately 110°C, and greater hydrophobicity than benzene due to the increased nonpolar surface area from the methyl group. These physical characteristics contribute to toluene’s functionality as a widely used solvent in paints, resins, adhesives, and coatings. Many organic compounds dissolve easily in toluene because its aromatic character and moderate nonpolarity allow it to solvate both weakly polar and nonpolar molecules effectively. This solvating ability stems from the molecule’s balance between hydrophobic interactions and electron-rich aromatic orbital overlap, which create favorable attractions with solute species ranging from polymers to oils.
In the context of industrial chemistry, the molecular structure of toluene makes it a key building block for chemical synthesis. Because the aromatic ring is activated toward electrophilic substitution, toluene serves as a precursor to compounds such as TNT (trinitrotoluene), benzoic acid, benzaldehyde, cresols, and xylenes. Under controlled laboratory or industrial conditions, nitration of toluene introduces nitro groups predominantly in the ortho and para positions, enabling the synthesis of specialized dyes, explosives, and chemical intermediates. Oxidation of the methyl group transforms the side chain into a carboxylic acid group, producing benzoic acid, an important compound used as a food preservative and in plasticizer production. Alternatively, selective oxidation may generate benzaldehyde, useful in perfumery and flavoring industries. These examples demonstrate how the lone methyl carbon attached to the aromatic ring becomes a functional handle that chemists can manipulate to produce dramatic chemical transformations. Toluene therefore reflects a larger concept within organic synthesis: a single molecular feature can represent an entry point to create a vast landscape of derivatives.
While the aromatic ring defines the electronic properties of toluene, the compound’s ability to undergo side-chain reactions demonstrates additional pathways of chemical interest. The benzylic position — the carbon directly attached to the aromatic ring — experiences unique reactivity due to resonance stabilization of intermediates. When free radicals or carbocations form at this benzylic carbon, neighboring π-electrons in the benzene ring delocalize into the side chain, stabilizing the reactive intermediate. As a result, benzylic hydrogen atoms are more reactive than hydrogens in typical aliphatic methyl groups. Free-radical halogenation, for example, occurs rapidly at the benzylic position to produce benzyl chloride, benzyl bromide, or benzyl iodide depending on reaction conditions. These reactions introduce functionality that enables further transformations, such as substitution, elimination, or coupling. The special stability of benzylic intermediates makes toluene a teaching tool for understanding how resonance extends beyond the aromatic ring into adjacent atoms, modifying reactivity beyond what pure aromatic or aliphatic compounds display independently.
Because of its aromaticity and activated ring structure, toluene also plays a role in materials science and polymer chemistry. In the production of polyurethane foams, adhesives, sealants, and elastomers, toluene diisocyanate (TDI) — derived from nitration and phosgenation of toluene — is a central monomer that reacts with polyols to create flexible or rigid polymeric materials. These materials span industries ranging from construction and automotive manufacturing to bedding and insulation. In gasoline formulations, toluene functions as an octane booster, increasing the resistance of fuel to premature detonation in internal combustion engines. This fuel-enhancing property arises from the stability of the aromatic ring and its high energy content, which allow toluene to burn smoothly rather than explosively under compression. Although the connection between molecular structure and fuel performance may not seem obvious initially, it exemplifies the overarching theme that molecular architecture directly influences macroscopic utility.
Educationally, toluene is essential for building conceptual understanding of organic chemistry because it connects key topics — aromaticity, resonance, directing effects, hyperconjugation, intermediate stability, substitution vs. side-chain reactivity, and structure–property relationships. Students who analyze toluene learn how electron-rich substituents activate aromatic rings, how reaction mechanisms depend on electron flow rather than simple memorization, and how the stability of reactive intermediates explains product formation. Toluene introduces learners to the logic that governs organic chemistry rather than isolated facts: when structure changes, reactivity changes, and those changes can be predicted by analyzing electron distribution. This makes toluene a conceptual stepping stone toward more complex aromatic systems that populate biological molecules, pharmaceuticals, polymers, agrochemicals, and advanced materials.
Ultimately, toluene illustrates how much meaning can be contained in a single molecule. Its structure blends the stability of an aromatic ring with the reactivity of a substituent that modifies electron distribution and directs chemical transformations. From paints and gasoline to polymers and fine chemicals, toluene’s applications trace directly back to its atomic-level organization. By understanding the molecular structure of toluene, students learn not only how aromatic compounds behave but also how careful mathematical and conceptual thinking about electrons, geometry, and resonance allows chemists to predict and control the outcome of reactions. In this way, toluene becomes not only a practical industrial substance but an educational doorway to the deeper language of organic chemistry.