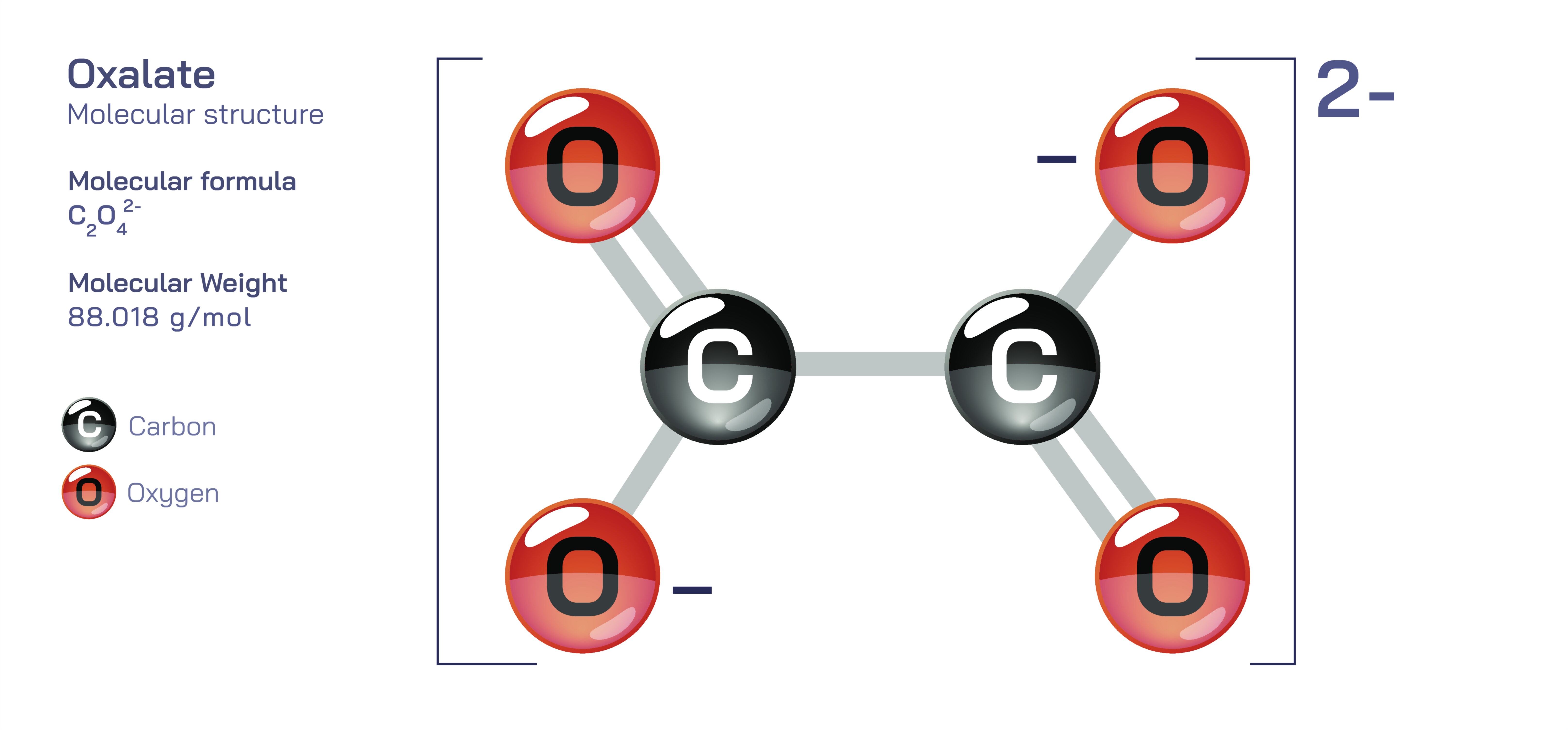
Oxalate – Molecular Structure, Bonding Characteristics, Charge Distribution, Coordination Chemistry, Biological Occurrence, Toxicological Significance, and Educational Insight into Organic and Inorganic Molecular Behavior
Oxalate is a chemically fascinating compound because it stands at the intersection of organic and inorganic chemistry, linking molecular structure, ionic bonding, biological metabolism, mineral formation, and toxicology into one unified topic of study. Represented by the formula C₂O₄²⁻, the oxalate ion is derived from oxalic acid and consists of two carbon atoms and four oxygen atoms bonded in a planar arrangement that reveals both symmetry and chemical complexity. At the structural level, oxalate consists of a central carbon-carbon linkage where each carbon atom is double-bonded to one oxygen and single-bonded to another oxygen that carries a negative charge, giving the ion its overall 2− charge. Although early diagrams portray a static arrangement of single and double bonds, the oxalate ion is actually stabilized by resonance, meaning electrons are delocalized across the entire structure rather than being confined to particular carbon–oxygen linkages. As a result, all C–O bonds in oxalate have similar character instead of being rigidly separated into single and double bonds. This delocalized electron distribution explains the notable stability of the oxalate ion in solution and its strong affinity for forming complexes with metal ions. For students learning molecular chemistry, oxalate provides a clear example of how resonance, charge distribution, and hybrid bonding patterns shape the behavior of polyatomic ions far beyond what a simple structural diagram may suggest.
The oxalate ion also highlights the important concept of charge stabilization in molecular systems. Each negatively charged oxygen atom holds only part of the total charge because resonance spreads the electrons across the ion, creating a balanced electrostatic profile. This arrangement not only prevents disproportionate buildup of charge that would destabilize the molecule but also increases oxalate’s capacity to act as a ligand in coordination chemistry, meaning it can bind to metal ions through two donor oxygen atoms. Because of its bidentate nature—binding through two coordination sites—the oxalate ion can form chelate complexes, creating stable ringlike structures around metal centers. These complexes are important in analytical chemistry and industrial applications because the oxalate ion’s ability to wrap around a metal ion increases binding strength and selectivity. A classic example is the formation of iron oxalate complexes, where oxalate coordinates with Fe³⁺ or Fe²⁺ in highly stable structures that demonstrate how electron-rich ligands latch strongly onto electron-deficient metal ions. This coordination ability is not only central to laboratory chemistry but also influences mineral formation in nature, helping explain why oxalate-containing rocks and geological deposits form under particular environmental conditions.
Oxalate additionally provides an opportunity to connect molecular structure with biological processes. In living organisms, oxalate is produced as a metabolic by-product, particularly during the breakdown of compounds such as glyoxylate and ascorbic acid. Under normal physiological conditions, small amounts of oxalate are harmless and are filtered out of the bloodstream through the kidneys into the urine. However, the chemical properties of oxalate make it prone to forming insoluble salts when it encounters certain metal ions—especially calcium. When calcium ions in urine interact with oxalate ions, the result can be the formation of calcium oxalate crystals, which accumulate into kidney stones. These stones are among the most common forms of nephrolithiasis and cause severe pain because the sharp crystalline structures scrape against urinary tract tissues. The connection between oxalate chemistry and human health therefore illustrates how seemingly simple molecular behavior can produce profound physiological effects. Foods rich in oxalate—including spinach, beet greens, rhubarb, nuts, and tea—can contribute to elevated oxalate levels in susceptible individuals, though biological risk is influenced not only by dietary intake but by hydration, calcium balance, gut microbiome activity, and liver metabolism. For educational purposes, oxalate becomes a strong example of how chemical structure translates into biological relevance and why nutrition science cannot be separated from molecular biochemistry.
Beyond kidney stone formation, oxalate also plays a role in plant biology. Many plants store oxalate in the form of calcium oxalate crystals as part of their defense mechanisms. The sharp needle-like crystals, called raphides, irritate the mouths and digestive tracts of herbivores, deterring feeding. Some plants, like members of the Araceae family, also use calcium oxalate to regulate cellular calcium levels and maintain ionic balance. In botany, oxalate crystals can even be used for plant classification because different species and genera produce distinct crystal shapes, positioning oxalate as a chemical tool for both protection and identity at the evolutionary scale. These biological functions highlight the dual nature of oxalate: chemically simple yet biologically sophisticated.
In environmental chemistry, oxalate appears naturally in soil and water because of plant decomposition and microbial activity. Soil bacteria decompose organic matter, releasing oxalate, which interacts with minerals and influences soil acidity. Some microorganisms metabolize oxalate as their carbon source, meaning oxalate supports microbial ecosystems and contributes to nutrient cycling. Light-driven atmospheric reactions involving oxalate also influence particulate chemistry in clouds and aerosols. Even though these processes occur far beyond the scale of a single molecule, they are influenced by oxalate’s structural properties—especially its ability to bind metals and precipitate insoluble compounds. This environmental role reinforces the lesson that molecular chemistry produces effects that cascade upward into large-scale ecological outcomes.
The educational value of studying oxalate expands further when considering analytical chemistry. Oxalate acts as both a reducing agent in some chemical reactions and a complexing agent in others. For example, in redox titrations, oxalate can be oxidized to carbon dioxide, releasing electrons that are detected through changes in reagents. The reaction between oxalate and potassium permanganate, often taught in undergraduate chemistry laboratories, demonstrates the principles of oxidation-reduction reactions, activation energy, and catalytic pathways. The fact that oxalate can participate in redox chemistry while also forming chelate complexes makes it a versatile teaching subject for students learning how molecules interact, transfer electrons, and stabilize charge within chemical systems. The same versatility makes oxalate valuable in industrial applications ranging from metal cleaning and rust removal to textile processing and photochemistry. These applications emphasize that chemistry is not confined to laboratory bottles—it shapes manufacturing, environmental systems, and biological function.
Understanding oxalate also offers a gateway into discussions about toxicity and safety, because substances that are naturally occurring are not always harmless. Pure oxalic acid, the parent molecule of oxalate, is corrosive and can interfere with calcium metabolism if consumed in excessive amounts. Even foods high in natural oxalate are safe for most people but potentially harmful for individuals predisposed to kidney stone formation or disorders of mineral balance. This nuance reinforces the educational lesson that toxicity depends on dose, biological context, and personal health, rather than on whether a substance is “natural” or “synthetic.” In practical healthcare, managing oxalate-related risks involves hydration, balancing calcium intake rather than eliminating it, and maintaining gut microbiome conditions that support oxalate-degrading bacteria. Even these medical recommendations trace back to the original chemical reality that oxalate forms insoluble salts that precipitate under particular ionic conditions.
Ultimately, the oxalate ion serves as an exemplary subject for chemistry education because it allows learners to see how molecular structure controls physical behavior, how behavior influences biological systems, and how those biological consequences scale upward to medical, ecological, and industrial relevance. Studying oxalate teaches that chemistry is not isolated but connected: resonance influences charge distribution, charge distribution influences solubility, solubility influences biological crystallization, crystallization influences disease development, and disease influences diet, medicine, and lifestyle. In this way, oxalate becomes a single molecule that demonstrates how deeply chemical principles shape living systems and human experience. Through the lens of oxalate, students begin to grasp one of the most important truths of molecular science—understanding the structure of a molecule is not merely an academic exercise, but a direct key to understanding how matter behaves, how life functions, and how human health and the environment are intertwined through the quiet logic of chemistry.