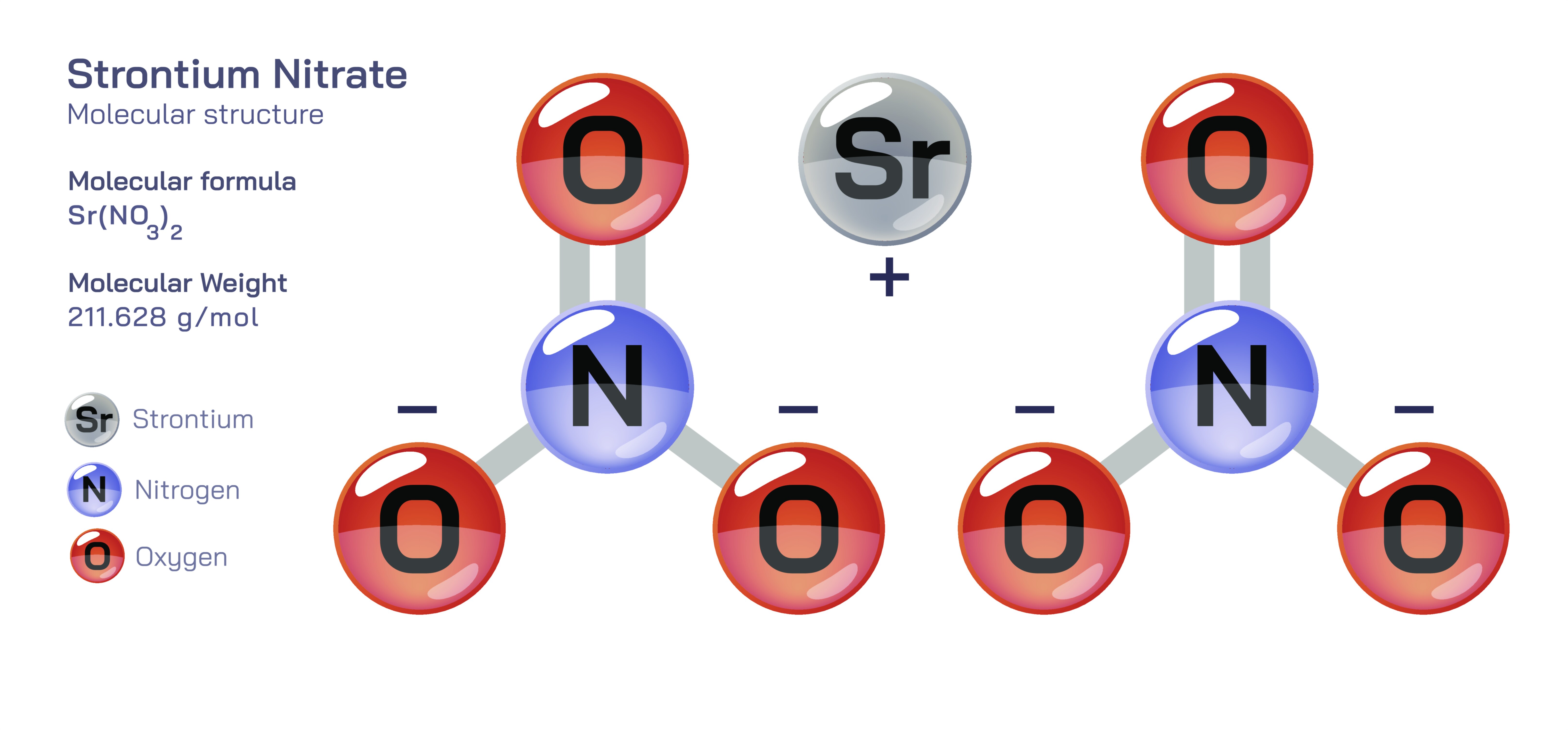
Strontium Nitrate – Molecular Composition, Ionic Structure, Physical Characteristics, Chemical Behavior, Thermal Decomposition, Reactivity in Aqueous Solution, and Educational Importance in Inorganic Chemistry
Strontium nitrate is an excellent compound for learning the structural and conceptual foundations of inorganic chemistry because it combines principles from ionic bonding, polyatomic anion chemistry, solubility, thermochemical reactions, and flame emission spectroscopy. Represented by the chemical formula Sr(NO₃)₂, the compound consists of one strontium cation (Sr²⁺) and two nitrate anions (NO₃⁻), forming a neutral ionic salt. Each component of this formula reveals an important lesson about periodic behavior and bonding. Strontium belongs to Group 2 of the periodic table and therefore naturally loses two electrons to achieve a stable noble-gas configuration. Nitrate is a polyatomic ion composed of nitrogen centrally bonded to three oxygen atoms in a trigonal planar arrangement, and it carries a single negative charge because of delocalized electrons in resonance among the oxygen atoms. Two nitrate ions are required to balance the +2 charge of one strontium ion, illustrating the fundamental rule of charge conservation in ionic compounds: the total positive charge must equal the total negative charge for the solid to be electrically neutral. This fundamental balancing rule is true across all ionic chemistry and helps students build predictable formulas, whether or not they have memorized specific compounds.
Even though the chemical formula looks compact, the bonding within strontium nitrate has two distinct character types that help clarify the difference between ionic and covalent interactions. Within each nitrate ion, nitrogen and oxygen atoms are held together by covalent bonds in which electrons are shared internally among the atoms. However, the nitrate ion as a whole does not form a covalent bond with the strontium ion. Instead, nitrate ions and strontium ions interact through electrostatic ionic attraction. This dual nature—covalent bonding inside the polyatomic ion and ionic bonding between ions in the lattice—offers an important instructional opportunity. It demonstrates to students that the term “ionic compound” does not imply that all bonds inside the structure are ionic; instead, ionic compounds often contain internal covalent units embedded in a larger ionic lattice. The nitrate group behaves like a discrete charged cluster, maintaining its internal structure even when the compound dissolves or reacts, further reinforcing the stability of resonance-stabilized polyatomic ions.
In its solid state, strontium nitrate arranges itself into a repeating ionic lattice rather than discrete molecular units. Each Sr²⁺ ion is surrounded by multiple nitrate ions, and each nitrate ion is electrostatically attracted to strontium ions in nearby positions. This geometric pattern continues indefinitely in three dimensions, reflecting the energetic preference of oppositely charged ions to maximize attraction and minimize repulsion. Because of this rigid arrangement, strontium nitrate appears as a crystalline solid with high density, high melting point, and significant brittleness—common features among ionic compounds. Examining these structural properties gives chemistry students an intuitive sense of how bonding type and packing arrangement influence macroscopic behavior. Unlike covalent molecular solids, which soften with heat and melt at relatively low temperatures because their molecules are held together only by weak intermolecular forces, ionic solids like strontium nitrate remain intact until substantial thermal energy breaks the ionic framework.
One of the most important teaching features of strontium nitrate is its behavior in aqueous solution. Because water molecules strongly attract ions, the rigid ionic lattice readily dissociates when strontium nitrate dissolves. In solution, Sr²⁺ ions and NO₃⁻ ions separate from each other and disperse uniformly, each becoming surrounded by hydration shells of water molecules. The complete dissociation of the compound makes strontium nitrate a strong electrolyte, meaning it conducts electricity efficiently when dissolved because of the free mobility of ions. This feature allows the salt to be used in conductivity experiments and in demonstrations of how ionic concentration affects the magnitude of electrical conduction. Additionally, the nitrate ion plays a central role in learning solubility rules, since nitrates of nearly all metals are soluble in water. This predictable behavior enables chemistry students to determine whether ionic compounds will precipitate when mixed in solution and to perform qualitative analysis in the laboratory. When a solution of strontium nitrate is combined with sulfate or carbonate ions, for example, an insoluble strontium salt precipitates, allowing students to detect the presence of Sr²⁺ ions even when invisible to the eye.
Strontium nitrate is particularly significant educationally because of its thermal decomposition behavior, which helps illustrate how some ionic compounds undergo redox reactions when heated. When strontium nitrate is exposed to high temperature, it breaks down into strontium oxide (SrO), nitrogen dioxide (NO₂), and oxygen gas (O₂). During this breakdown, the nitrate anion is reduced and oxidized within the same reaction, making this a classic example of an intramolecular redox rearrangement. The production of oxygen gas explains why strontium nitrate acts as an oxidizing agent in pyrotechnics and combustion reactions: it supplies oxygen to sustain burning when other sources of oxygen may be limited. Understanding this decomposition mechanism teaches students how electron transfer chemistry, oxidation–reduction processes, and energetic decomposition can occur even in apparently stable ionic solids.
Another visually striking property of strontium nitrate is its flame coloration. When heated in a flame, strontium ions emit a deep crimson or bright red color, which corresponds to the release of energy as electrons transition from excited states back to their ground state. This effect is used in fireworks, signal flares, and pyrotechnic displays. From a teaching standpoint, strontium nitrate is therefore a gateway to learning about emission spectroscopy, a technique in which different elements are identified by the characteristic wavelengths of light they emit when excited. The red emission of strontium distinguishes it sharply from yellow sodium, green barium, or violet potassium, illustrating that flame tests are not arbitrary demonstrations but rather direct evidence of quantized electronic structure in atoms. When students connect the red color in fireworks to the electronic configuration of Sr²⁺, the atomic world becomes directly observable and memorable.
Strontium nitrate also supports learning in broader contexts of environmental, industrial, and material chemistry. It is used as a precursor for synthesizing other strontium compounds, a component of luminous and signal devices, and a reagent for laboratory precipitation analysis. It plays a role in ceramics and glass manufacturing, where it influences mechanical and optical properties. Each of these applications reinforces the link between microscopic structure and real-world uses: the nitrate anion carries oxygen that makes the compound an oxidizer, the strontium ion produces characteristic flame emission, and the ionic form dissolves easily in water to permit controlled chemical reactivity.
From an educational standpoint, strontium nitrate is not valuable only because of its properties, but because these properties emerge from fundamental chemical principles that students must learn to master. It ties together ionic bonding, covalent bonding within polyatomic ions, geometric resonance stabilization, solid-state lattice structure, solubility rules, electrolyte dynamics, precipitation reactions, thermal decomposition, redox processes, and quantum-level light emission. When chemistry learners see all of these linked within a single compound, the discipline becomes less fragmented and more integrated. Strontium nitrate becomes a teaching example that shows how chemical formulas are not simply symbols on paper but fingerprints of structure, charge distribution, and energetic behavior.
This compound thus represents a powerful bridge between theory and application. By understanding strontium nitrate deeply—from the arrangement of ions in the lattice to the color of its flame—students learn to extrapolate structure–property relationships and to recognize that chemical understanding is cumulative. Strontium nitrate demonstrates how the periodic table, bonding rules, electron behavior, solubility, and spectroscopy come together to shape the observable world, preparing learners for more advanced exploration of inorganic chemistry, materials science, and analytical instrumentation.