Sulfate Ion – Molecular Structure, Bonding Arrangement, Charge Distribution, Resonance Stability, Physical and Chemical Behavior, and Educational Importance in Inorganic Chemistry
The sulfate ion, represented by the formula SO₄²⁻, is one of the most important and widely studied polyatomic ions in chemistry. It appears in minerals, biological systems, fertilizers, industrial processes, atmospheric cycles, and laboratory reactions. Understanding the sulfate ion at the molecular level reveals foundational concepts in bonding, resonance, molecular geometry, acid–base chemistry, and solubility patterns. Because sulfate contains both covalent and ionic character depending on the compound in which it is found, it helps learners develop a clear understanding of how atoms share and transfer electrons in complex ions rather than forming simple two-atom molecules.
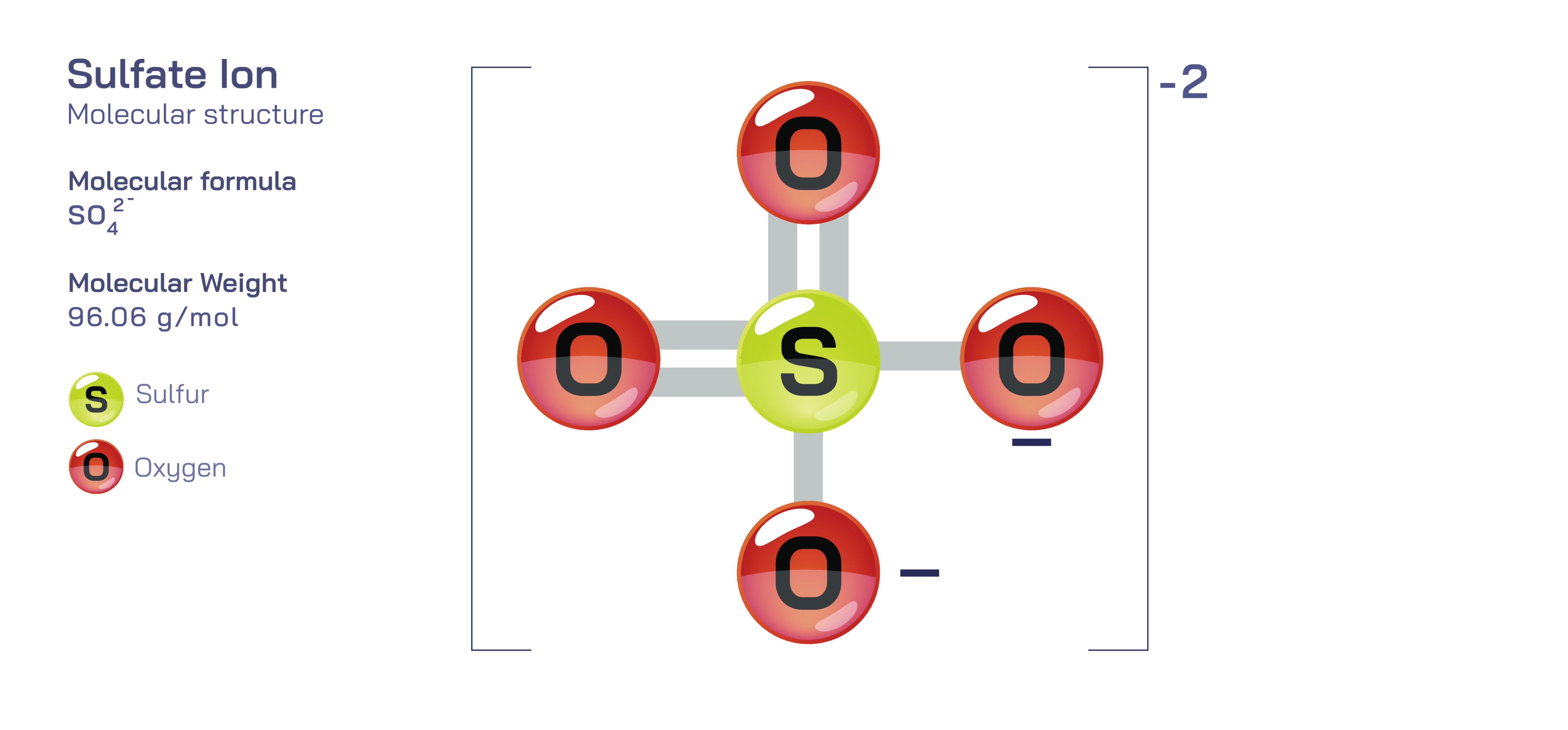
Structurally, the sulfate ion consists of one central sulfur atom bonded to four oxygen atoms, forming a tetrahedral geometry. The sulfur sits at the center of the tetrahedron and each oxygen occupies a vertex at nearly equal distances from the sulfur. This arrangement arises because sulfur in the +6 oxidation state forms four equivalent S–O bonds. Although Lewis diagrams often depict sulfate as having two double bonds and two single bonds, modern chemical understanding shows that the real structure is not a static mix of single and double bonds. Instead, sulfate exhibits resonance, meaning the electrons involved in bonding are delocalized over the entire ion. Rather than four unequal S–O bonds, all four bonds are equal in length and strength due to electron sharing distributed across multiple resonance structures. This delocalization greatly stabilizes the sulfate ion and illustrates why resonance is not simply an artistic drawing convention, but a physical phenomenon that influences molecular properties and reactivity.
The overall 2− charge of sulfate is not concentrated on one atom but is distributed across the ion. Each oxygen atom carries a partial negative charge because oxygen is more electronegative than sulfur, and sulfate stability relies on this balanced distribution rather than a localized charge. As a result, the sulfate ion does not act as four independent negatively charged oxygens but as a unified chemical species capable of interacting with cations such as sodium, calcium, magnesium, barium, strontium, and many others. These ionic interactions form crystalline salts in which the sulfate ion remains chemically intact, demonstrating the durability of resonance-stabilized polyatomic ions in both solid and dissolved states.
Because of its charge distribution and resonance stabilization, the sulfate ion has a strong tendency to combine with positively charged ions, forming a wide variety of salts. Many sulfates are highly soluble in water, such as sodium sulfate and magnesium sulfate, whereas others such as barium sulfate, lead sulfate, and strontium sulfate are poorly soluble and precipitate out of solution. These patterns are an important part of classical solubility rules, used widely in analytical chemistry to identify unknown ions in solution. For example, when a soluble sulfate is added to a solution containing barium ions, the formation of a white precipitate of barium sulfate is a reliable indicator of Ba²⁺. Through this kind of qualitative testing, students learn how sulfate participates in double displacement reactions and why predictable solubility patterns make sulfate useful in laboratory analysis.
In acid–base chemistry, the sulfate ion provides insight into dissociation and proton transfer. Sulfate forms when sulfuric acid (H₂SO₄) loses both of its acidic hydrogen ions through dissociation. The first proton is released completely in water, forming the hydrogen sulfate ion (HSO₄⁻), and the second proton is released when HSO₄⁻ loses an additional hydrogen to form SO₄²⁻. This two-step dissociation explains why sulfuric acid is a strong diprotic acid and why sulfate acts as the conjugate base of HSO₄⁻. Observing this progression helps students see how polyprotic acids can lose more than one proton and why the resulting ions differ in both charge and reactivity.
The electronic structure of sulfate also makes it an excellent example for introducing the concept of expanded valence shells. In the sulfate ion, sulfur forms more than the typical octet of electrons seen in second-period elements like carbon, nitrogen, or oxygen. Because sulfur belongs to the third period of the periodic table, it has access to 3d orbitals that allow expanded bonding capacity. Whether or not these orbitals are actively used has been debated in modern computational chemistry, but from a teaching standpoint, sulfate demonstrates that elements beyond the second period can stabilize structures involving more than eight electrons around the central atom. This conceptual flexibility helps students understand why heavier main-group elements frequently form highly coordinated oxyanions like sulfate, phosphate, and nitrate.
Sulfate also supports learning about molecular geometry and VSEPR theory. With four substituent atoms and no lone pairs on the central sulfur, the electron-pair geometry and molecular geometry of sulfate are both tetrahedral. This symmetry means sulfate has no permanent dipole moment as a whole, even though individual S–O bonds are polar. This reinforces the concept that polarity depends not only on the presence of polar bonds but also on their spatial arrangement.
Beyond its structural and chemical principles, the sulfate ion plays important roles in environmental and biological systems. Sulfates occur naturally in seawater, minerals, soil, volcanic emissions, and industrial effluents. They are essential for plant nutrition because sulfur is needed to synthesize certain amino acids and enzymes. In the atmosphere, sulfate aerosols influence cloud formation and climate interactions. In living organisms, sulfate participates in detoxification reactions within the liver and in the formation of structural biomolecules such as glycosaminoglycans. These biological and environmental applications teach students that inorganic ions are not confined to laboratory chemistry but drive global and physiological processes.
From a chemical education perspective, the sulfate ion unifies multiple core topics: covalent bonding within polyatomic ions, ionic bonding between ions, resonance stabilization, electron delocalization, tetrahedral geometry, acid–base dissociation, solubility rules, qualitative analysis, and environmental chemistry. By studying sulfate, learners gain a deeper understanding of why polyatomic ions behave consistently across different reactions and why apparently simple formulas contain layers of structural meaning.
Ultimately, sulfate brings together the central message of molecular chemistry: the arrangement and distribution of electrons determine the structure, charge, stability, and reactivity of a species. When students understand SO₄²⁻ not just as a symbol but as a tetrahedral, resonance-stabilized ion with clear patterns of interaction, they develop foundational insight that extends far beyond sulfate itself—providing stepping stones toward mastery of inorganic chemistry, analytical science, and environmental molecular principles.