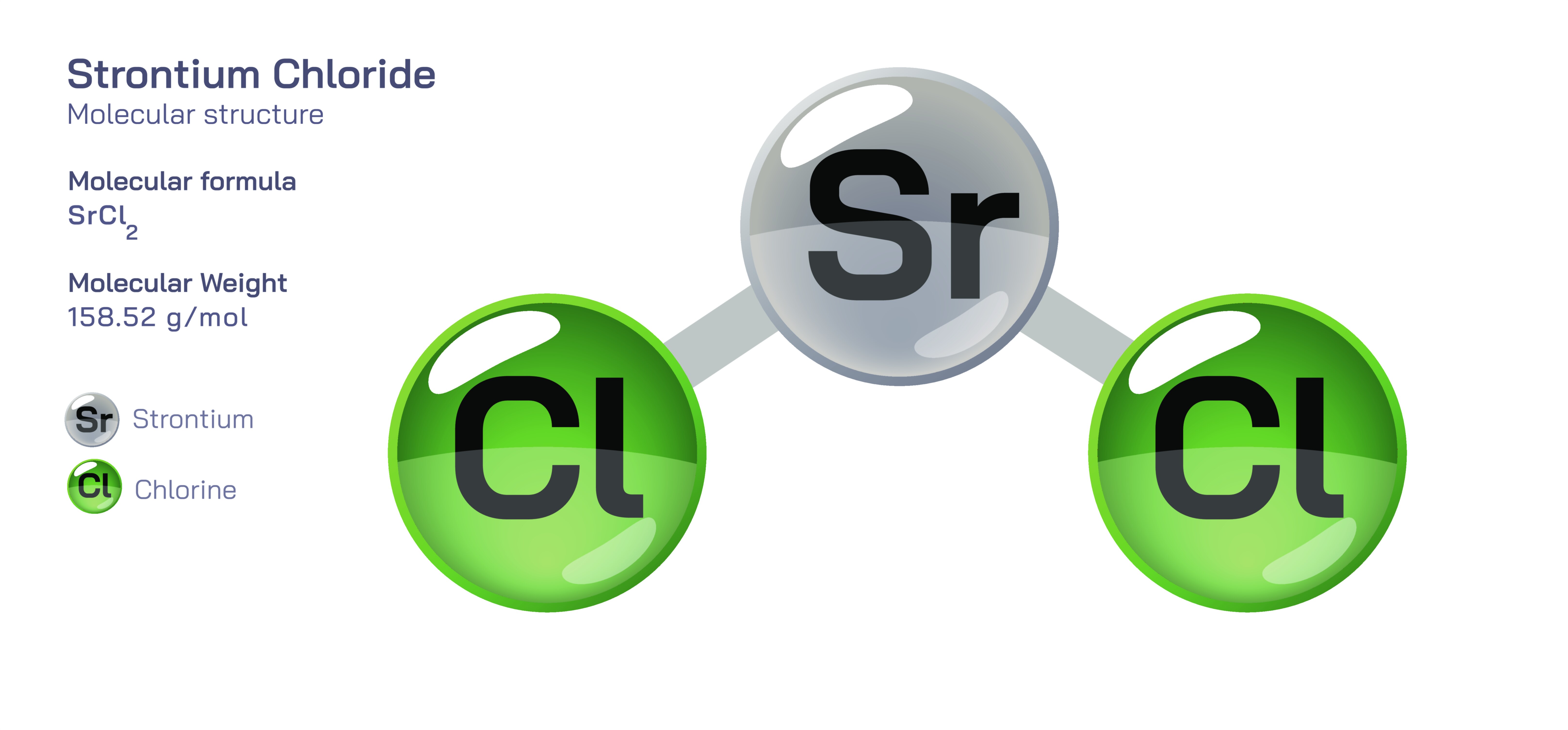
Strontium Chloride – Molecular Structure, Ionic Bonding, Physical Behavior, Aqueous Chemistry, and Educational Significance in Inorganic Chemistry
Strontium chloride is an important inorganic compound that provides an excellent platform for understanding the principles of ionic bonding and structure, the periodic behavior of alkali earth metals, and the relationship between atomic properties and bulk chemical characteristics. With the formula SrCl₂, strontium chloride is composed of two chloride anions and one strontium cation, forming a neutral salt in which electrical balance is achieved through the combination of a divalent alkaline earth metal ion with two monovalent halide ions. This stoichiometric arrangement arises directly from the periodic chemistry of strontium, a metal in Group 2 of the periodic table that naturally loses two electrons to achieve a stable electronic configuration, and chlorine, a Group 17 element that naturally gains one electron to achieve the same stability. The resulting ionic compound exemplifies the predictability of chemical bonding across the periodic table: divalent metals pair with two monovalent nonmetals to form electrically neutral substances. Understanding this electron-transfer relationship is a foundational step in chemistry education because it reinforces periodic trends, charge balancing, electronegativity principles, and the energetic preference of atoms to achieve noble gas configurations through bond formation.
When examined structurally, strontium chloride does not consist of discrete molecular units as covalent compounds do. Instead, its structure consists of an extended three-dimensional ionic lattice where each strontium ion is surrounded symmetrically by chloride ions, and each chloride ion is likewise surrounded by multiple strontium ions. The electrostatic attraction between these oppositely charged ions forms a strong and rigid structure that contributes to the compound’s high melting point, crystalline appearance, and considerable thermal stability. This distinguishes ionic solids from molecular solids, where discrete molecules are held together by weaker intermolecular forces. In the case of strontium chloride, the repeating lattice network builds indefinitely in space, demonstrating how ionic compounds are not composed of “molecules” in the traditional sense, but instead of solid-state arrangements governed by electrostatic forces. This distinction helps students understand why ionic compounds behave differently from covalent compounds in terms of melting behavior, solubility, and electrical conductivity.
The crystalline environment around the strontium ion also allows educators to introduce the concept of ionic radii and coordination geometry. Strontium, a relatively large cation due to its electron configuration and its position lower in the group below calcium, adopts a coordination arrangement in which multiple chloride ions surround it in a symmetrical pattern. This spatial distribution is driven by energy minimization within the lattice: ions organize themselves in a way that maximizes attraction and minimizes repulsion. Through this arrangement, learners begin to appreciate how atomic size, charge density, and lattice energy collectively influence the stability and physical properties of ionic solids. Comparing strontium chloride with magnesium chloride or calcium chloride, for example, helps demonstrate how lattice strength and melting point vary with ion size and charge. These concepts are not abstract; they are observable in laboratory experiments and computational modeling, reinforcing how structure governs material behavior.
In aqueous chemistry, strontium chloride offers valuable insight into solubility, dissociation, and electrolyte behavior. When dissolved in water, the ionic bonds between ions do not remain intact; instead, the lattice separates, and strontium and chloride ions become individually surrounded by water molecules through hydration interactions. This dissolution process allows strontium chloride to act as a strong electrolyte, freely generating charged species that conduct electricity in solution. For students learning about electrolyte classification, strontium chloride provides a textbook example of complete dissociation, in contrast to weak electrolytes or non-electrolytes, which dissociate incompletely or not at all. The free mobility of ions in water is the basis for conductivity tests commonly performed in introductory chemistry laboratories, where strontium chloride solutions display strong conduction due to the abundance of dissolved ions. This reinforces the link between ionic structure, solubility behavior, and measurable physical consequences, such as conductivity and colligative effects.
Strontium chloride further serves as an educational model for understanding chemical reactivity of Group 2 metals in solution. When dissolved, the strontium ion does not generally form complex ions with water, meaning it remains relatively simple chemically compared with many transition metal ions. However, the chloride ions can participate in precipitation reactions when introduced to solutions containing metal ions that form insoluble chlorides. Conversely, strontium ions can be precipitated out of solution by sulfate ions or carbonate ions, creating a useful demonstration of solubility and precipitation rules. Such reactions show how ionic compounds interact in predictable ways during double displacement processes, allowing students to determine the identities of unknown ions in solution based on the presence or absence of precipitates. This makes strontium chloride an important reagent in analytical chemistry and one that appears frequently in laboratory exercises designed to teach solubility patterns and qualitative ionic analysis.
In thermochemistry and flame emission studies, strontium chloride offers additional educational value, especially in illustrating how metal ions emit characteristic colors when heated. When strontium ions are introduced to high-temperature sources such as a Bunsen flame, electrons within the ion become excited to higher energy levels and then release energy as red light when they return to their original state. This phenomenon is responsible for the deep crimson-red flame coloration often associated with fireworks containing strontium salts. This real-world application demonstrates how atomic structure and quantized energy transitions drive observable macroscopic effects. It also supports teaching about emission spectroscopy, an analytical technique in which atoms and ions emit light of specific wavelengths that can be used to identify them. Strontium chloride allows learners to see how spectroscopy connects quantum-level behavior to practical applications in materials science, entertainment technology, and forensic chemical analysis.
Strontium chloride also finds relevance in industrial, environmental, and biological contexts that help learners appreciate how inorganic chemistry extends beyond the laboratory. In industry, it is used in the production of other strontium compounds, metal refining, pyrotechnics, and corrosion control. In biological and biochemical research, strontium ions behave similarly to calcium ions due to their comparable charge and size, allowing researchers to substitute strontium for calcium in studies of cellular signaling and bone mineralization. These applications give context to the earlier discussion of ionic radius and periodic trends: similarity between ions in the same group influences substitution behavior in natural and technological systems.
From an educational perspective, strontium chloride functions as a bridge between theory and observation. It embodies many fundamental chemistry principles simultaneously: the predictable charges of ions based on periodic position, the ionic lattice arrangement of solid salts, the relationship between lattice energy and physical properties, the dissociation of ionic compounds in water, the conduction of electricity in solution, the formation of precipitates in double displacement reactions, and the emission of characteristic wavelengths in flame spectroscopy. Because all of these behaviors arise from the same underlying structural characteristics, students learn that the molecular composition of a compound is not just a symbolic formula but a blueprint that determines the compound’s behavior across chemical contexts.
Ultimately, understanding strontium chloride at the molecular and structural level reinforces one of the most important lessons in chemistry: the arrangement of atoms and ions dictates the properties and applications of a material. When learners study strontium chloride in detail, they strengthen not only their knowledge of a single compound but also their ability to interpret how ionic bonding, periodic trends, aqueous chemistry, and energy changes shape the identity of inorganic substances. By seeing how a formula such as SrCl₂ expands into a world of structural, physical, and chemical insights, students develop the analytical thinking skills essential for advancing into more complex areas of chemistry, materials science, and applied research.