Sulfur Dioxide – Molecular Structure, Bonding Characteristics, Electron Geometry, Polarity, Reactivity, Environmental Chemistry, and Educational Insight into Molecular Behavior
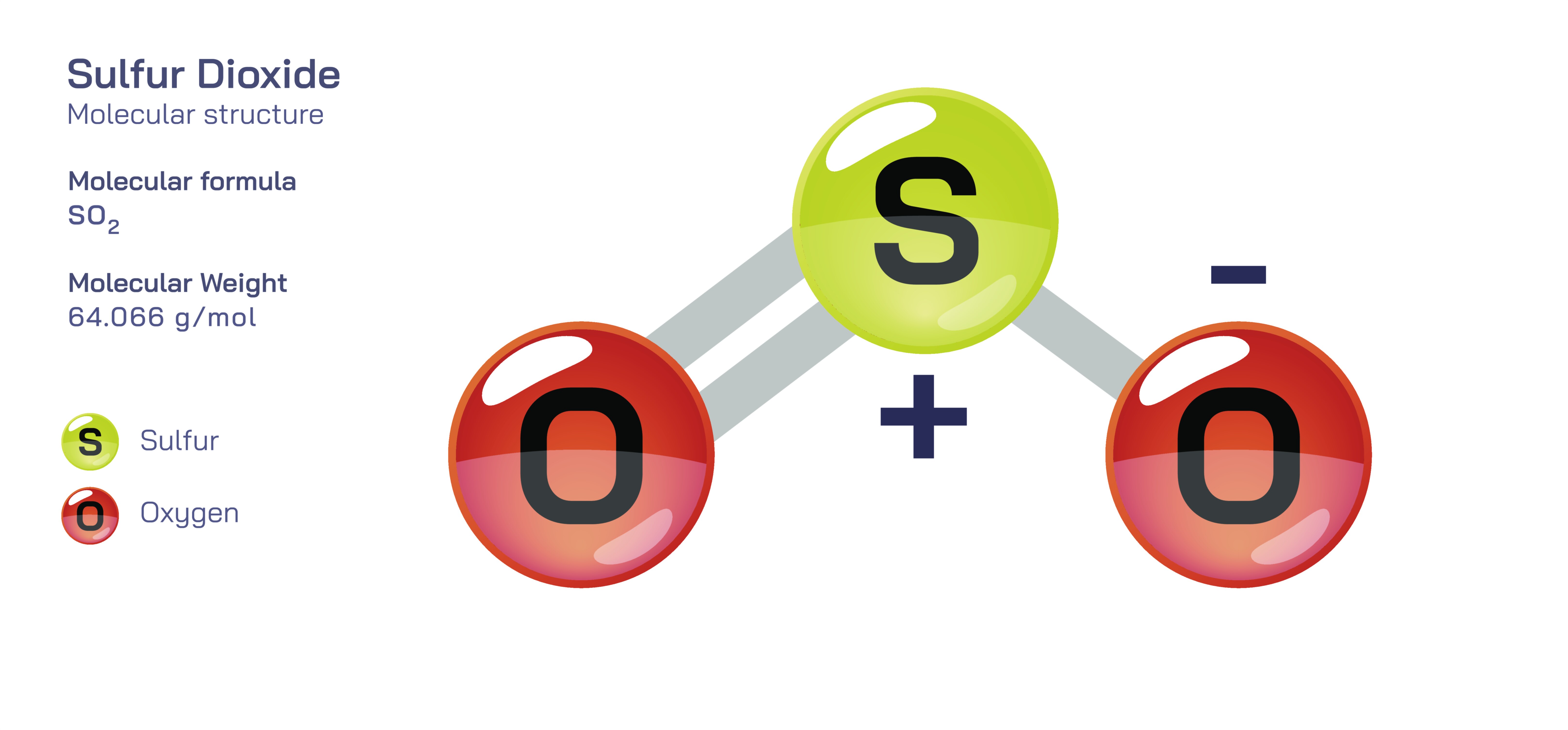
Sulfur dioxide, represented by the chemical formula SO₂, is a small yet deeply informative molecule that has earned an important place in chemistry education because its structure and properties highlight essential ideas such as covalent bonding, molecular geometry, resonance, polarity, oxidation state, acid–base behavior in water, redox processes, and environmental chemistry. Although simple in appearance, sulfur dioxide demonstrates how atomic arrangement and electron distribution can dramatically shape the behavior of a substance, giving rise to specific physical characteristics, types of chemical reactivity, and wide-ranging consequences for living systems and the atmosphere. Understanding SO₂ at a molecular level therefore teaches students how structural chemistry and real-world science are intertwined rather than separate academic domains.
The structural foundation of sulfur dioxide begins with its atomic composition: one sulfur atom bonded to two oxygen atoms. However, the arrangement is not linear, and the molecule does not adopt a pattern where the three atoms lie in a straight line. Instead, sulfur dioxide takes on a bent molecular shape, which reflects deeper electronic behavior. The key to this geometry lies in the distribution of electron pairs around the central sulfur atom. Sulfur is surrounded by two bonding electron domains, each linking sulfur to oxygen, plus one lone pair of electrons that remains on sulfur and does not participate in bonding. According to valence shell electron pair repulsion (VSEPR) theory, electron groups repel each other and seek spatial positions that minimize repulsion. In SO₂, the two bonding pairs and one lone pair arrange themselves in a trigonal planar electron geometry, but because one position is occupied by a nonbonding pair rather than an atom, the observable molecular geometry becomes bent. The presence of this lone pair compresses the O–S–O bond angle below the ideal 120°, giving the molecule its angular shape. This structural arrangement is not just a textbook illustration but the reason SO₂ exhibits dipole behavior, solubility patterns, and specific interactions with other molecules.
The bonding in sulfur dioxide offers another valuable educational lesson because it presents a situation that is more nuanced than simple single or double bonds. In many introductory depictions, sulfur is shown double-bonded to both oxygen atoms simultaneously, creating the appearance of a symmetrical structure. But electron distribution in SO₂ is not fixed that way in reality; instead, sulfur dioxide exhibits resonance, meaning that the electrons in the pi-bonding system are delocalized across the molecule rather than locked into one rigid picture. It can be drawn as sulfur double-bonded to one oxygen and single-bonded to the other, but the actual resonance hybrid shows that both S–O bonds have an intermediate bond order between a single and a double bond. The equal bond lengths of the two sulfur–oxygen bonds observed experimentally confirm this delocalization. The resonance model teaches students that some molecules cannot be fully described by a single Lewis structure, and that stability is enhanced when electrons are spread across multiple bonding arrangements rather than concentrated in one. This idea is central to higher-level chemistry and provides continuity between simple bonding models and quantum mechanical descriptions of molecular orbitals.
Sulfur dioxide also illustrates the importance of molecular polarity in determining behavior and reactivity. Because the molecular structure is bent rather than linear and because oxygen is significantly more electronegative than sulfur, the charge distribution in SO₂ is uneven. The molecule has a net dipole moment, meaning that electrons tend to be drawn toward the oxygens, creating partial negative charges on them and a partial positive charge on the sulfur atom. This polarity affects many physical properties: SO₂ dissolves readily in polar solvents such as water, contributes to strong intermolecular interactions when acting as a gas or liquid under pressure, and influences the way the molecule interacts with biological tissue and atmospheric components. Understanding polarity through SO₂ helps students see how geometry and electronegativity work together to produce observable chemical patterns rather than remaining abstract concepts on paper.
The behavior of sulfur dioxide in water is another major point of chemical education, especially because it bridges inorganic chemistry and environmental science. When SO₂ dissolves in water, it forms sulfurous acid (H₂SO₃), although the acid does not exist as a pure isolated compound outside solution. The reversible hydration equilibrium demonstrates principles of acid–base chemistry, showing that sulfur dioxide itself acts as an acidic oxide. The resulting solution contains bisulfite (HSO₃⁻) and sulfite (SO₃²⁻) ions in varying proportions depending on conditions. These transformations teach students how molecular species shift identity during hydration and how pH and equilibrium influence the relative amounts of different ions. Because the equilibrium favors hydration in water-rich environments such as clouds and atmospheric droplets, the behavior of SO₂ becomes significant in the chemistry of rainwater and atmospheric transport cycles.
Sulfur dioxide also participates actively in redox chemistry. Sulfur in SO₂ has an oxidation state of +4, which places the compound in a middle position between sulfate, where sulfur is in the +6 oxidation state, and hydrogen sulfide or elemental sulfur, where the oxidation state is lower. This intermediate value enables SO₂ to behave as either an oxidizing agent or a reducing agent depending on reaction partners. In some reactions, it donates electrons and is oxidized to sulfate; in others, it accepts electrons and is reduced. This dual capability makes sulfur dioxide a vivid example of how oxidation states govern chemical reactivity, and helps learners recognize that redox behavior depends not only on absolute electron count but on the chemical environment and available pathways.
The environmental chemistry of sulfur dioxide further reinforces the principle that molecular structure can have large-scale consequences. SO₂ is released naturally through volcanic eruptions and geothermal activity, but large amounts also enter the atmosphere from industrial sources, heavy fuel combustion, and metal smelting. Through atmospheric hydration and oxidation processes, sulfur dioxide forms acidic components that can lead to acid rain, which affects soil chemistry, aquatic systems, vegetation, and building materials. These transformations are not unrelated to what students learn at the molecular level; the same hydration that produces sulfurous acid in the laboratory is responsible for acid formation in clouds. Understanding the molecule helps explain how human activities influence environmental cycles, demonstrating why atmospheric chemistry is not a separate field but an extension of basic chemical principles.
In controlled settings, sulfur dioxide plays important roles as a preservative, disinfectant, and antioxidant. In the food and beverage industries—especially in winemaking—SO₂ prevents microbial growth and slows oxidation that would deteriorate flavor and quality. This antimicrobial action stems from the molecule’s reactive nature toward biological functional groups, allowing it to disrupt pathways that would otherwise promote spoilage. The same antioxidant behavior explains why sulfur dioxide has been historically used to prevent discoloration in dried fruit and to sterilize barrels and equipment. These applications counterbalance its hazardous nature at high concentrations and teach students that toxicity depends on dose, context, and exposure rather than being an inherent quality of a molecule.
A comprehensive educational appreciation of sulfur dioxide emerges when thinking of it not as an isolated formula but as a showcase of core chemistry concepts. It demonstrates how electron arrangement determines shape, how shape determines polarity, how polarity influences solubility and intermolecular forces, how resonance affects bond strength and stability, how oxidation state guides reactivity, and how molecular processes scale up into environmental and industrial outcomes. In studying SO₂, learners gain not only knowledge about a specific molecule but also the ability to interpret chemical formulas as blueprints for behavior. By understanding sulfur dioxide deeply rather than superficially, students develop insights they can transfer to wider areas of chemistry ranging from covalent bonding to environmental stewardship, reminding them that the microscopic structure of a molecule has macroscopic consequences for the world around them.