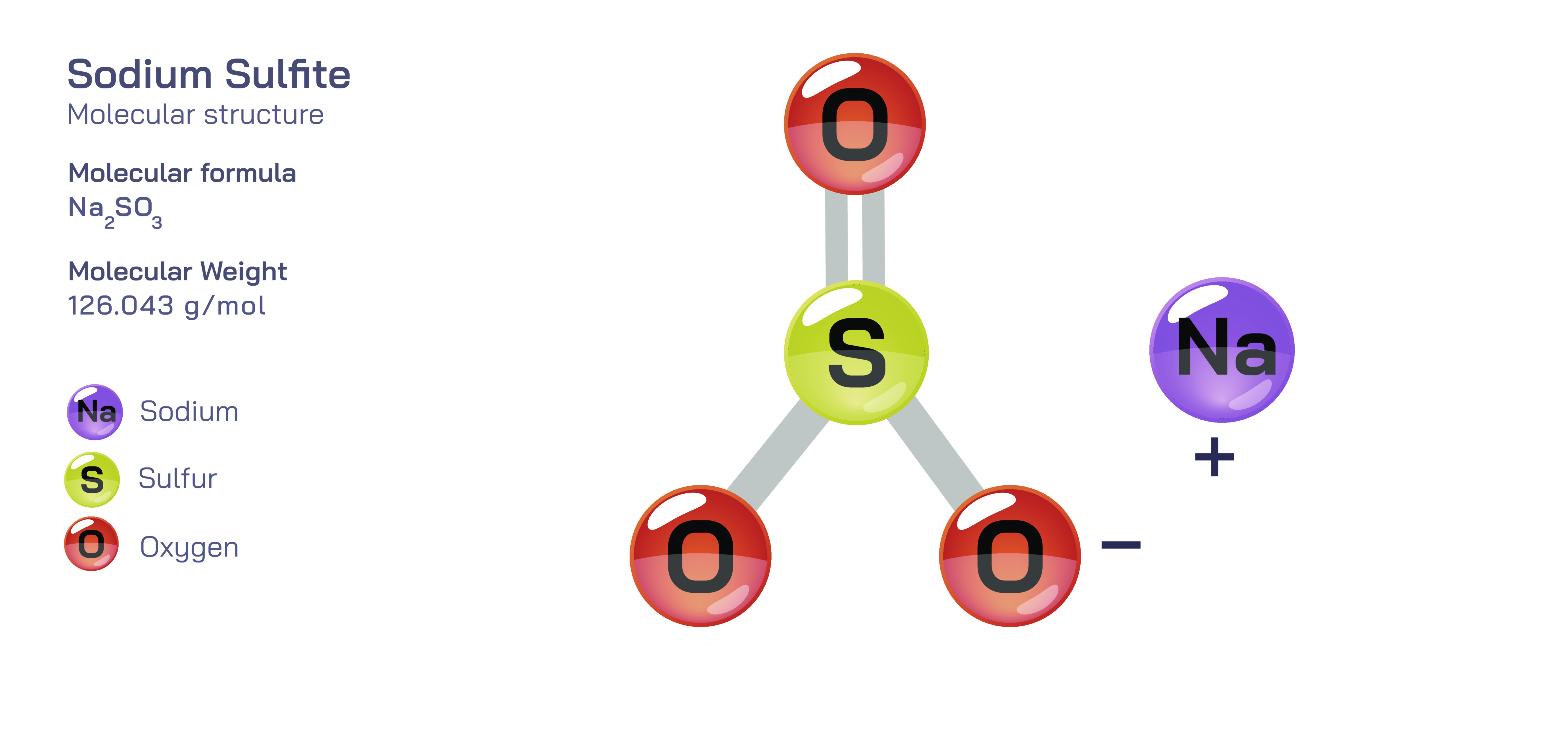
Sodium Sulfite – Molecular Structure, Ionic Composition, Aqueous Behavior, and Educational Significance in Chemistry
Sodium sulfite is an inorganic compound that offers a rich platform for explaining many foundational chemical concepts, especially those involving ionic structure, oxidation states, redox reactions, and aqueous chemical behavior. Represented by the chemical formula Na₂SO₃, sodium sulfite consists of two sodium ions and one sulfite ion, and this balance between cations and anions provides an early opportunity for students to understand the principle of charge neutrality in ionic compounds. With each sodium ion carrying a single positive charge and the sulfite ion possessing a total negative charge of two, the numerical relationship between ions becomes a key example of how electrical forces dictate the stoichiometry of salts. When visualizing the structural components, learners can imagine sodium atoms donating electrons to achieve a stable electronic configuration while sulfur and oxygen share bonded electrons to form the negatively charged sulfite group. This interplay between electron transfer and electron sharing governs the arrangement of atoms in the compound and helps frame the broader distinction between ionic and covalent bonding. While the sodium–sulfite attraction is fully ionic, the bonds inside the sulfite ion remain covalent, which makes sodium sulfite an ideal case to illustrate the coexistence of different bonding types within a single compound.
From a structural perspective, the sulfite ion itself has notable geometric and electronic features that provide educational value. Unlike the sulfate ion, which has a perfectly symmetrical tetrahedral shape, the sulfite ion maintains a trigonal pyramidal geometry created by three oxygen atoms surrounding a central sulfur atom along with a non-bonding electron pair. This molecular arrangement has important implications for electron distribution and polarization, and it helps teachers demonstrate the effect of lone electron pairs on molecular shape. Students who explore molecular geometry models can visualize how the presence of unshared electrons modifies the spatial positioning of atoms and influences molecular polarity. The sulfite ion also behaves as a resonance-stabilized species, meaning that the distribution of electrons can be represented through multiple structural depictions, a concept that deepens understanding of electron delocalization within polyatomic ions. Although resonance itself is not something directly observable, its impact becomes clear when we consider the stability and reactivity of sulfite-containing compounds, and sodium sulfite becomes a perfect subject for introducing this advanced idea without overwhelming beginners.
Sodium sulfite also offers an excellent gateway into the study of aqueous chemistry because of the distinctive behavior of the sulfite ion when dissolved in water. In solution, sodium sulfite dissociates into free sodium and sulfite ions, and because the sodium ions remain chemically inactive, most observable behavior in water arises from the sulfite species. The sulfite ion acts as a moderately strong base and interacts readily with water molecules, forming bisulfite ions and hydroxide ions while increasing the solution’s alkalinity. This property allows students to clearly observe the relationship between anion identity and pH changes, emphasizing that not all ionic compounds remain pH-neutral upon dissolution. Beyond its basic character, sulfite offers another important educational angle: its ability to serve as a reducing agent in aqueous environments. This redox capability is one of its defining chemical features, tied directly to the oxidation state of the sulfur atom and its tendency to undergo oxidation to sulfate when exposed to oxygen or certain oxidizing agents. Watching sodium sulfite transform through oxidation provides an accessible way to study electron transfer and the role that reducing agents play in stabilizing or altering the oxidative state of other substances.
In laboratory experiments, sodium sulfite is frequently used to demonstrate oxidative–reductive transformations because it undergoes chemical changes that are visually or analytically easy to track. When introduced to solutions containing oxidizing agents, sodium sulfite can gradually transform into sodium sulfate, allowing students to link observable changes to the underlying electron-transfer process. In analytical chemistry exercises, sulfite’s reducing properties are sometimes exploited to remove dissolved oxygen from aqueous samples, demonstrating the practical use of chemical reagents to control environmental conditions in experimental settings. Additionally, sodium sulfite’s interaction with acids introduces students to gas-evolving reactions, as sulfurous acid formed in situ decomposes to release sulfur dioxide gas. This behavior reinforces concepts related to acid–base chemistry, decomposition reactions, and the relationship between ionic identity and gaseous products. The visible bubbling and distinct sulfur dioxide odor produced during these reactions illustrate how chemistry often provides both sensory and theoretical clues to determine what transformations occur during experiments.
Industrial use of sodium sulfite offers further insight into how the intrinsic chemical nature of a compound translates into real-world relevance. Its reducing capability supports applications such as food preservation, where it inhibits oxidative spoilage by reacting with and neutralizing oxygen-containing compounds. In photographic development solutions, sodium sulfite protects photosensitive chemicals from oxidation, illustrating how chemical stability directly influences materials and processes beyond the classroom. In water treatment and environmental remediation, sodium sulfite can remove residual chlorine through reductive dechlorination, preventing damage to industrial systems and aquatic ecosystems. These practical applications demonstrate how the interaction between sodium ions and sulfite ions leads to predictable dissolved behavior, making sodium sulfite not only a standardized compound in educational chemistry but also an important industrial reactant. When students study these uses, they recognize that every chemical property—from molecular geometry to redox activity—has implications that scale far beyond theoretical study.
One of the most compelling educational aspects of sodium sulfite is its ability to bridge multiple domains of chemistry in a seamless and interconnected way. At the introductory level, it teaches ionic bonding, polyatomic ions, and dissociation in water. As students progress, sodium sulfite becomes a tool for exploring molecular geometry, resonance, and acid–base hydrolysis. At more advanced levels, its redox behavior opens discussions about oxidation states, reaction predictions, and industrial treatment strategies. Throughout this learning arc, sodium sulfite remains consistent in structure yet versatile in educational value, making it a compound that aligns naturally with many milestones in chemical understanding. When instructors choose sodium sulfite for demonstrations or experiments, students witness the logical unfolding of chemical principles—from atomic structures and electron organization to full-scale chemical reactions and applications—reinforcing the notion that chemistry is not a collection of isolated topics but a unified scientific framework.
In summary, sodium sulfite serves as a rich, multidimensional subject in chemistry education because its properties clearly reflect its molecular structure and ionic composition. The trigonal pyramidal geometry of the sulfite ion, the ionic attraction between sodium and the polyatomic anion, the compound’s behavior in water, its basicity, and its redox activity all combine to create an easily accessible yet deeply instructive chemical profile. Studying sodium sulfite trains learners to connect atomic-level reasoning with observable physical and chemical behavior, building a foundation for advanced scientific analysis and laboratory competence. As students grow more comfortable interpreting the relationships between composition, structure, and reactivity, sodium sulfite becomes more than a compound to memorize—it becomes a vivid demonstration of how chemistry explains the material world, how theoretical laws guide experimental outcomes, and how molecular interactions form the basis of technologies and processes that society depends on every day.