Sulfur Trioxide – Molecular Structure, Bonding Arrangement, Electron Geometry, Reactivity, Acid Formation, Industrial Importance, and Educational Insight into Molecular and Environmental Chemistry
Sulfur trioxide, represented by the chemical formula SO₃, is one of the most significant compounds in modern chemistry because it sits at the crossroads of molecular structure, acid–base chemistry, industrial processes, environmental science, and atmospheric reactions. Although composed of only one sulfur atom and three oxygen atoms, SO₃ demonstrates how subtle differences in electron geometry, resonance, and molecular reactivity give rise to far-reaching chemical behavior. Studying sulfur trioxide at the molecular level offers insight not just into the behavior of this compound itself, but also into the broader logic of inorganic chemistry, illustrating how structure determines function in both laboratory settings and global environmental systems.
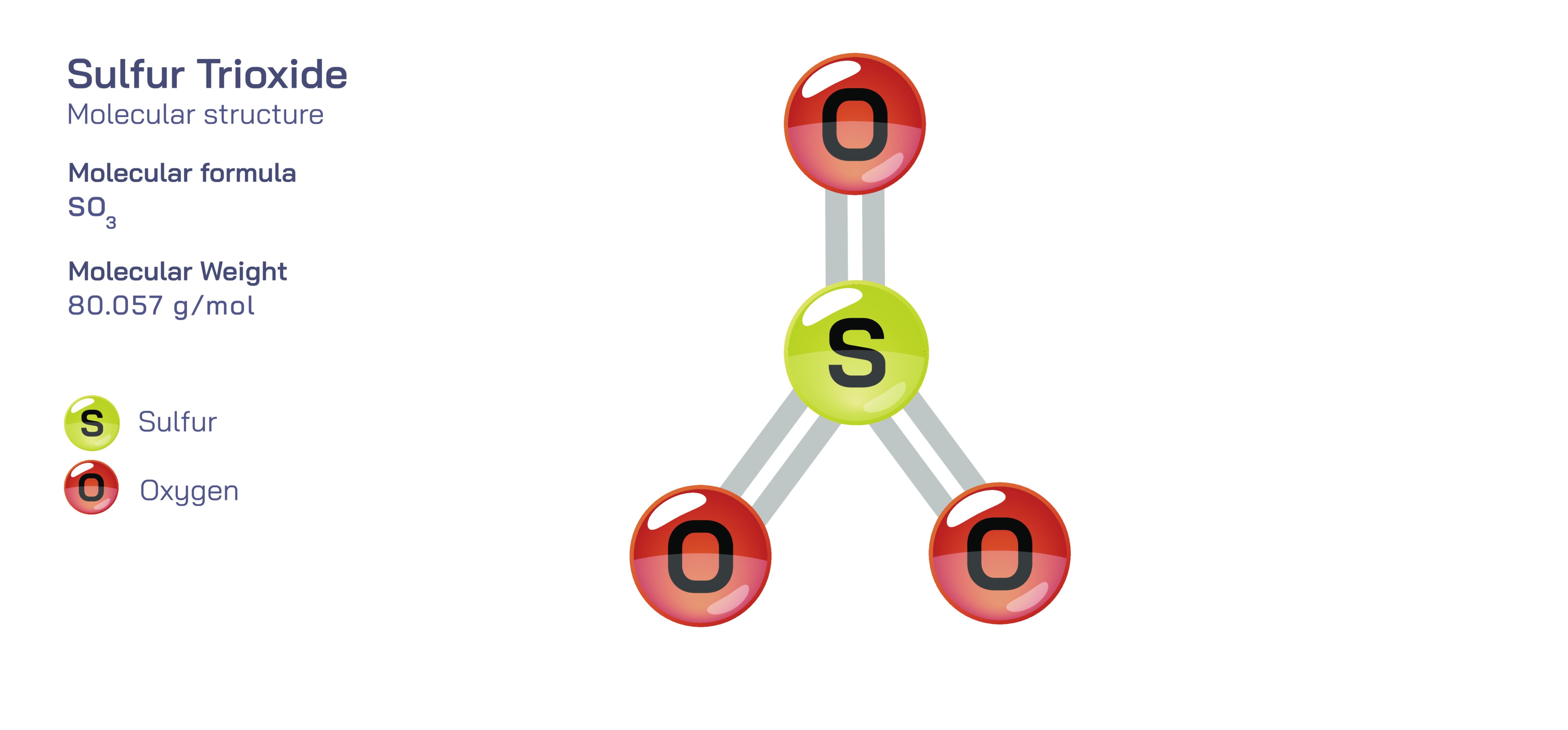
At the core of SO₃’s identity is its geometric structure. Sulfur trioxide adopts a trigonal planar molecular geometry, with the sulfur atom positioned at the center and the three oxygen atoms distributed evenly around it. The O–S–O bond angles are approximately 120°, reflecting equal repulsion between electron domains. There are three bonding electron groups and no lone pairs on the central sulfur atom, making the geometry symmetric and flat. This arrangement is consistent with valence shell electron pair repulsion (VSEPR) theory, which predicts that atoms and electron groups adopt configurations that minimize repulsion. The symmetry of the trigonal planar structure is not simply aesthetic—it directly influences the molecule’s polarity, reactivity, and role in acid formation.
The bonding in sulfur trioxide reveals one of the most instructive examples of resonance in inorganic chemistry. Lewis structures for SO₃ are often drawn showing sulfur double-bonded to all three oxygen atoms, but this picture appears at odds with the octet rule. Because sulfur is in the third period of the periodic table, it can stabilize more than eight electrons around itself by distributing electrons over expanded bonding interactions. However, instead of treating each S–O bond as a full double bond or a fixed mixture of single and double bonds, the most accurate description is that the electrons are delocalized across the entire molecule, forming a resonance hybrid with three equivalent S–O bonds. Experimental measurements support this: all S–O bonds in sulfur trioxide have the same intermediate length and strength. This delocalized bonding pattern gives the molecule its extraordinary stability and explains its strong tendency to act as a reactive electrophile toward other substances, including water.
Despite the high electronegativity difference between sulfur and oxygen, the molecule as a whole is nonpolar. Each S–O bond is polar, but in the trigonal planar arrangement, the three dipoles cancel out perfectly. This cancellation yields a molecule with no permanent dipole moment, even though its internal bonds are individually polar. This concept is critical for students learning the difference between bond polarity and molecular polarity, and SO₃ serves as a clear example of how molecular shape determines overall polarity. This symmetry affects how SO₃ dissolves, how it interacts with electric fields, and why it behaves differently from similarly bonded but asymmetrical molecules.
The reactivity of sulfur trioxide in the presence of water is one of its most important and defining characteristics. When SO₃ contacts water, it reacts extremely vigorously to form sulfuric acid (H₂SO₄). Unlike the more gradual hydration of sulfur dioxide to sulfurous acid, the hydration of sulfur trioxide is so energetically favorable that it cannot simply be mixed directly with water on an industrial scale due to heat release and the formation of acid mist. The reaction is not a passive dissolution; it is a chemical transformation that produces an acid with two replaceable protons and strong oxidative properties. This single reaction provides an educational link between molecular chemistry and industrial work, showing how the hydration of an oxide becomes the foundation for sulfuric acid production.
Sulfur trioxide’s reactivity and importance extend beyond laboratory chemistry because it plays an essential role in the manufacture of sulfuric acid, one of the highest-volume chemicals produced worldwide and a cornerstone of industrial civilization. The Contact Process, which converts sulfur dioxide to sulfur trioxide using a catalyst such as vanadium(V) oxide, builds directly on the molecular behavior of SO₃. Once formed, the gas is absorbed not directly into water but into concentrated sulfuric acid to form oleum, which can later be diluted safely. Every step of this production sequence—from the oxidation of SO₂ to the controlled absorption of SO₃—is driven by the molecular structure and reactivity of sulfur trioxide. This production chain is not merely industrial trivia; it is one of the clearest real-world demonstrations of how a molecule’s chemical nature shapes large-scale technological processes.
SO₃ also plays a major role in environmental and atmospheric chemistry, illustrating how human activity and molecular interactions intersect. When fossil fuels containing sulfur impurities are burned, SO₂ is released and can be oxidized in the atmosphere to SO₃. Once formed, sulfur trioxide reacts with atmospheric moisture to produce sulfuric acid aerosol droplets, contributing to acid rain and impacting ecosystems, buildings, and public health. The atmospheric transformation of SO₃ parallels what occurs in industrial sulfuric acid production, reinforcing the lesson that the same molecular process behaves differently depending on environmental context. Understanding SO₃ at the structural level therefore provides crucial insight into climate chemistry and environmental protection.
In controlled environments, sulfur trioxide is also used as a sulfonating agent in organic synthesis, contributing to the production of detergents, dyes, pharmaceutical compounds, and polymers. The strong electrophilic character of SO₃ allows it to attach a sulfonyl group (–SO₃H or –SO₃⁻) to organic molecules, dramatically modifying their solubility, acidity, and industrial value. This reactivity highlights how a molecule that behaves aggressively in water can, with proper handling, become a powerful tool in synthetic chemistry, emphasizing that chemical utility and chemical hazard can coexist depending on context.
For educational purposes, sulfur trioxide combines many foundational concepts in one molecule:
It demonstrates trigonal planar geometry and VSEPR theory in action.
It shows how third-period elements can expand valence capacity beyond the octet rule.
It reinforces resonance stabilization and electron delocalization in polyatomic species.
It teaches the relationship between geometry and molecular polarity.
It highlights the transformation of oxides to acids and the dynamics of acid–base reactions.
It connects molecular chemistry with industrial chemistry and global atmospheric processes.
Through sulfur trioxide, students learn to think about chemistry as an interconnected system rather than isolated facts. They see how structure shapes reactivity, how reactivity shapes application, and how application shapes industrial and environmental outcomes. The molecule functions not only as an object of study but as a lens through which to understand why chemistry has such profound influence on technology, ecosystems, and society.
Ultimately, sulfur trioxide illustrates one of the central truths of molecular science:
the microscopic arrangement of atoms governs both the behavior of matter and the impact it has on the world.
By studying SO₃ deeply, chemistry learners gain the analytical framework needed for advanced understanding in inorganic chemistry, industrial processes, atmospheric science, and environmental stewardship.