Oxygen – Molecular Structure, Bonding Behavior, Electron Configuration, Chemical Reactivity, Biological Importance, Atmospheric Role, and Deep Educational Insight into the Element That Sustains Life and Drives Energy Transformations
Oxygen holds a unique position in chemistry and biology because it is both a fundamental building block of matter and the molecule that sustains almost every form of complex life on Earth. Although many people think of oxygen simply as "the gas we breathe," its true scientific importance reaches far deeper into molecular bonding, metabolism, atmospheric stability, combustion processes, energy conversion, and evolutionary biology. Chemically, oxygen most commonly exists as a diatomic molecule (O₂) in the atmosphere, meaning two oxygen atoms bond together to form a stable pair. This molecular structure is not accidental; it arises from the electron configuration of the oxygen atom and its tendency to achieve stability through covalent bonding. Each oxygen atom has six valence electrons and needs two more to complete its octet. By sharing two electrons with another oxygen atom through a double covalent bond, both achieve an energetically stable configuration with eight electrons in their outer shells. The result is a linear O=O structure with equal sharing of electrons, and this bond arrangement explains why molecular oxygen is relatively stable yet still capable of powerful chemical reactivity.
On an electronic level, molecular oxygen has one of the most intriguing bonding patterns in chemistry because its ground-state form is a diradical, meaning it contains two unpaired electrons in separate antibonding orbitals. These electrons exist with parallel spins, giving oxygen paramagnetic properties—an unusual behavior for a stable diatomic molecule. This characteristic can be visually confirmed through laboratory demonstrations in which liquid oxygen is attracted toward a strong magnetic field. The paramagnetic nature of oxygen arises not from exotic external influences but from fundamental electron orbital arrangements predicted by molecular orbital theory. These orbital configurations, showing that oxygen possesses two unpaired electrons even in its stable form, teach students that Lewis structures alone cannot fully explain every molecular property. Instead, oxygen becomes a clear example of how quantum orbital models complement classical chemical bonding descriptions and deepen understanding of molecular behavior.
Despite its stability, molecular oxygen is chemically reactive because it strongly attracts electrons. This electron-seeking characteristic makes oxygen a powerful oxidizing agent. When oxygen accepts electrons from another substance, that substance becomes oxidized, leading to a release of energy in biological systems, industrial processes, and combustion reactions. Rusting of iron, burning of fuels, metabolic respiration, and cellular energy production all involve oxygen serving as the final electron acceptor. In living organisms, oxygen’s electron affinity is the driving force behind cellular respiration, the biochemical pathway in which nutrients such as glucose are broken down to produce ATP, the energy currency of life. Without oxygen acting as the terminal electron acceptor in the electron transport chain, aerobic organisms would be unable to extract large amounts of energy from food, and multicellular complexity would not be possible. In this sense, oxygen is not only a gas that organisms inhale; it is the essential molecular participant that enables life to convert chemical energy stored in nutrients into a usable form.
The presence of oxygen in Earth’s atmosphere has a profound evolutionary and planetary story. For billions of years, Earth’s early atmosphere contained almost no free oxygen; it was dominated by gases such as methane, ammonia, nitrogen, and carbon dioxide. It was not until ancient cyanobacteria evolved the ability to perform photosynthesis—splitting water molecules using sunlight and releasing oxygen as a by-product—that atmospheric oxygen began to accumulate. This transition triggered what scientists call the Great Oxygenation Event, radically altering Earth’s chemistry and life forms. The appearance of oxygen caused the extinction of many anaerobic organisms that could not tolerate oxidative environments, while simultaneously enabling the evolution of aerobic species with high-energy metabolic systems. Geological records show that oxygen began binding to iron in the oceans, forming massive banded iron deposits that still exist today. Only after iron was saturated did oxygen start collecting in the atmosphere, paving the way for ozone formation, which protected the Earth’s surface from harmful ultraviolet radiation and allowed complex land-based organisms to develop. In this way, the molecular structure of oxygen—not just its presence—shaped the trajectory of life and environmental stability on Earth.
Oxygen also plays a fundamental role in combustion chemistry, one of the most widely observed and technologically applied chemical processes. Combustion involves the rapid reaction of a fuel with oxygen, releasing heat and light. Whether it is the burning of wood in a fireplace, the ignition of gasoline in a car engine, or the conversion of chemical propellants in rocket propulsion, oxygen acts as the electron acceptor that drives these exothermic reactions. The concept of oxidation—once misunderstood historically as "the substance that adds oxygen"—was later redefined scientifically as loss of electrons, yet oxygen remains the most familiar oxidizing agent because of its ubiquity and reactivity. This duality makes oxygen a teaching bridge between historical chemical models and modern electron-transfer frameworks. Many fundamental science lessons involving fire safety, fuel efficiency, combustion engineering, and atmospheric control originate from the molecular behavior of oxygen and its double-bonded diatomic structure.
Although oxygen is indispensable for life, there is a fine balance between its beneficial and harmful properties. Under normal physiological conditions, oxygen participates in controlled metabolism, yet excessive or unregulated oxygen reactions can generate reactive oxygen species (ROS) such as superoxide, hydrogen peroxide, and hydroxyl radicals. These highly reactive molecules can damage proteins, DNA, and lipids, leading to oxidative stress and aging at the cellular level. Biological organisms have therefore evolved antioxidant enzymes such as catalase, peroxidase, and superoxide dismutase to neutralize ROS and maintain equilibrium. This aspect of oxygen chemistry reflects a recurring scientific theme: the same element that enables life can threaten it when its reactions spiral beyond biological control. Understanding oxygen therefore teaches students that chemical species are not inherently "good" or "bad"; their effect depends on regulation, context, and concentration.
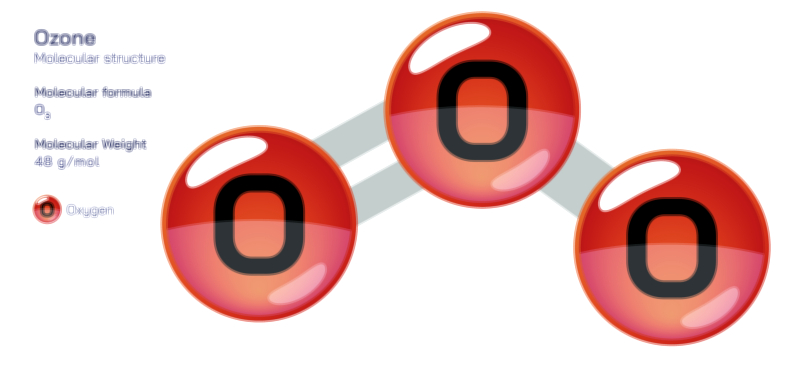
Oxygen exists not only as molecular oxygen (O₂) but also in other forms with distinct structures and roles. Ozone (O₃), for example, consists of three oxygen atoms bonded in a bent geometry with resonance-stabilized electron distribution. Ozone is highly reactive chemically and forms in the upper atmosphere through ultraviolet interactions with O₂, where it absorbs UV radiation and protects living organisms from genetic damage. At ground level, however, ozone becomes a toxic pollutant generated through photochemical smog formation. Another related form is singlet oxygen, an excited state of O₂ in which electrons become paired rather than unpaired, making the molecule highly reactive and relevant in photodynamic therapy, plant defense systems, and atmospheric chemistry. These oxygen variants illustrate how small changes in electron arrangement dramatically alter molecular reactivity and environmental or biological impact.
Oxygen also helps shape water chemistry, geological cycles, and environmental balance. Its role in oxidative weathering alters rocks and minerals over millions of years, influencing soil composition and nutrient availability for plant life. In aquatic environments, dissolved oxygen concentration determines the survival of fish and microbial species, making oxygenation essential for ecological balance. Hypoxic or anoxic waters—formed through pollution, eutrophication, or stagnation—can trigger mass die-offs and disrupt food chains, showing how oxygen availability governs not only individual organisms but entire ecosystems.
Industrial and technological applications of oxygen reflect its pervasive relevance. Oxygen is used in steel production, welding and cutting torches, wastewater treatment, medical ventilation systems, space exploration life support, and chemical synthesis pathways. In medicine, oxygen therapy sustains individuals with impaired breathing, yet must be administered cautiously because excessive oxygen exposure at high pressure can cause oxidative damage—particularly in premature infants or deep-sea divers. These contrasting scenarios reinforce the educational understanding that oxygen is a powerful chemical agent whose benefits arise from balance rather than abundance.
Studying oxygen reveals one of the most important scientific principles: a molecule’s structure determines its function. The double-bonded diatomic geometry, paramagnetic electronic structure, and high electron affinity explain why oxygen stabilizes life, accelerates combustion, shapes ecosystems, and influences evolutionary pathways. Oxygen is not simply a component of air but a molecular cornerstone whose properties ripple across biology, physics, chemistry, geology, and medicine.
Understanding oxygen helps learners see the world differently. It teaches that breathing is not just inhalation but a complex electron-transfer process; that life depends on delicate biochemical control; that the environment relies on molecular balance; and that scientific progress grows from understanding how atoms combine, bond, and behave under different conditions. Through the study of oxygen, students gain not only knowledge of one molecule but a broader scientific mindset—one that connects structure to function, chemistry to life, and microscopic interactions to the macroscopic patterns that define our planet.