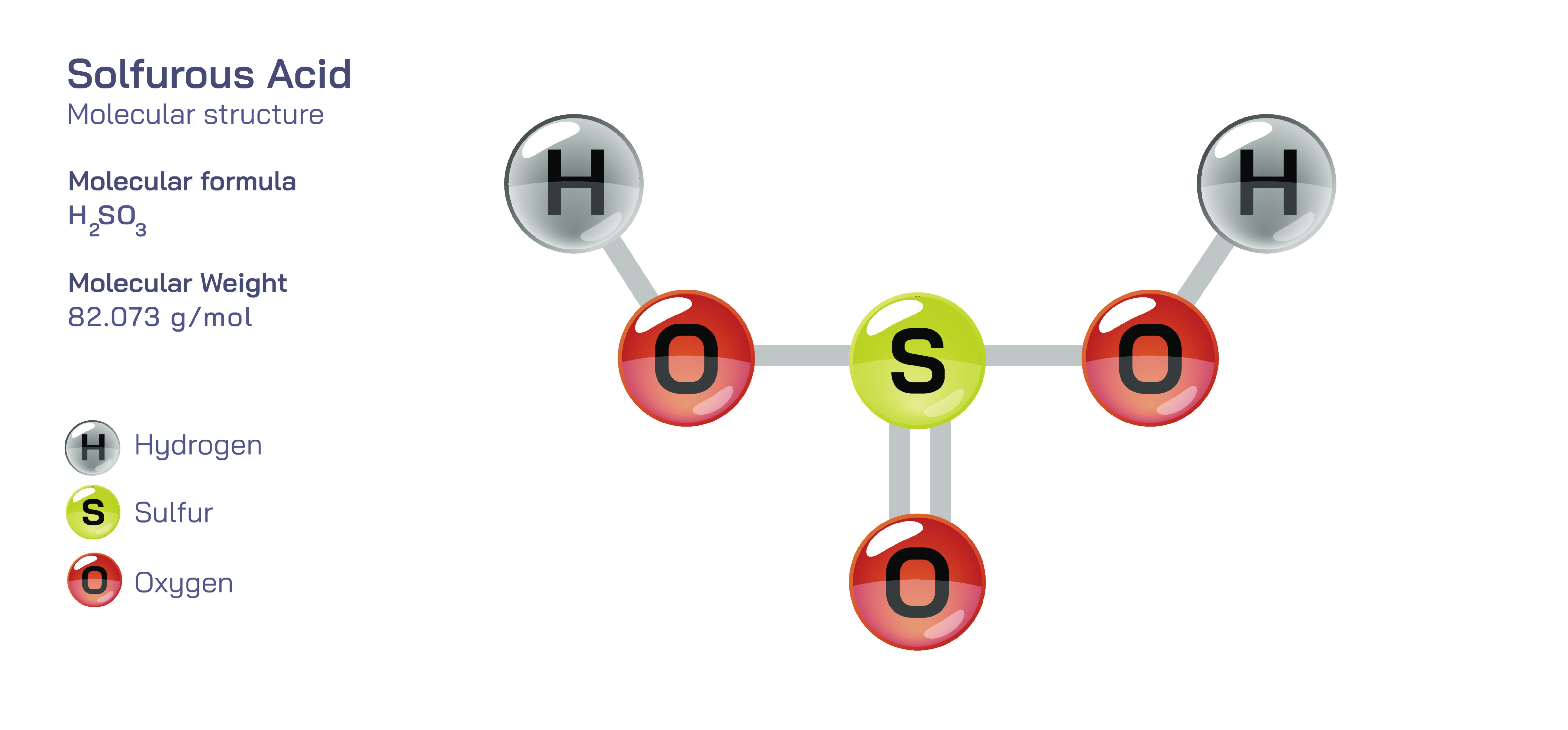
Sulfurous Acid – Molecular Structure, Formation in Aqueous Systems, Chemical Behavior, and Educational Significance in Understanding Acid–Base and Redox Chemistry
Sulfurous acid occupies an important conceptual space in chemistry because it allows learners to connect structure, bonding, acid–base theory, and redox transformations in a way that illustrates the dynamic nature of certain inorganic molecular species. Although it is commonly represented by the formula H₂SO₃, sulfurous acid is not an isolatable pure liquid under normal conditions, and this characteristic alone becomes a valuable teaching point: unlike many mineral acids that can be obtained in concentrated form, sulfurous acid exists only in solution and only when sulfur dioxide dissolves in water. The formation of sulfurous acid demonstrates how gas–liquid interactions can generate weak acids through reversible hydration of dissolved gases, and students gain a deeper understanding of molecular identity by recognizing that not every chemical formula corresponds to a stable and bottle-ready substance. The first step in understanding sulfurous acid from a structural perspective involves visualizing sulfur dioxide entering water and bonding with water molecules to form a hydrated product in which sulfur remains centrally bonded to oxygen atoms while hydrogen atoms attach to oxygen atoms rather than directly to sulfur. This structural arrangement introduces the polyatomic sulfite core, and the addition of hydrogen converts the sulfite ion into the conjugate acid, highlighting the role of protonation in modifying chemical identity. Through this formation pathway, students learn that sulfurous acid is not a permanent structure but rather an equilibrium species whose existence depends on molecular interactions occurring continually in aqueous surroundings.
Examining the molecular structure of sulfurous acid deepens students’ comprehension of bonding variability within oxyacids and challenges them to interpret compound geometry as a consequence of electron arrangement. In the most widely accepted structural representation, sulfur sits at the nucleus of the molecule bonded to three oxygen atoms, one of which forms a double bond, while the other two carry hydrogen atoms bonded through oxygen–hydrogen linkages. The geometry around the sulfur atom is trigonal pyramidal due to the presence of a lone electron pair, and this deviation from perfect symmetry provides an accessible illustration of how electron repulsion and lone pairs alter idealized molecular shapes. The hydrogen atoms are not attached directly to sulfur but instead bond to oxygen, reinforcing the concept that acidic hydrogen in oxyacids is associated with an OH functional group rather than the central atom. This placement also helps students understand why sulfurous acid acts as a proton donor: hydrogen atoms attached to oxygen remain polarized, and this polarization facilitates their release in aqueous solutions. The resonance character within the SO₃ framework illustrates how negative charge within the conjugate base can delocalize across multiple oxygen atoms, and this delocalization stabilizes the sulfite ion once deprotonation occurs. Even though resonance structures are theoretical constructs rather than measurable structural states, they help learners appreciate how shared electron distribution stabilizes ionic forms and influences acid strength.
As students explore the chemical behavior of sulfurous acid, they learn that its acidic nature is moderate and that ionization proceeds in multiple steps. When dissolved in water, sulfurous acid releases hydrogen ions gradually rather than completely, making it a typical example of a weak diprotic acid. The first dissociation step produces the bisulfite ion, and the second dissociation—less extensive—yields the sulfite ion. Observing this two-stage deprotonation helps students understand how diprotic acids do not lose both protons simultaneously and how each dissociation has its own equilibrium constant. Sulfurous acid becomes a convenient case for explaining the concept of conjugate acid–base pairs, because each ion generated through dissociation is related structurally and functionally to the acid from which it derives. The equilibrium nature of sulfurous acid also provides an easy entry into discussions about Le Châtelier’s principle, because changing the pressure of dissolved sulfur dioxide shifts the concentration of sulfurous acid in predictable ways. When more sulfur dioxide is introduced into water, the equilibrium shifts toward increased formation of sulfurous acid, whereas sparging or heating the solution drives sulfur dioxide out of the solvent, shifting the equilibrium back toward the gaseous side. These reversible structural changes engage students in the concept that acids can exist as momentary states rather than fixed chemical entities.
Beyond acid–base reactions, sulfurous acid also holds a key position in redox chemistry because of its ability to act as a reducing agent. The sulfur atom in sulfurous acid is in an intermediate oxidation state, making it capable of undergoing oxidation to sulfate under appropriate conditions. This redox activity explains why sulfurous acid and its salts are frequently encountered in reactions that involve bleaching, oxygen removal, and both industrial and biological antioxidant pathways. When sulfurous acid reacts with oxidizing agents, the products normally include sulfate ions along with protons, supporting exploration of oxidation number assignments and electron-transfer tracking in classroom settings. Because sulfurous acid has a reducing role, students observe its transformation through reactions with colored oxidizing agents, where color changes indicate the transfer of electrons. This type of experiment helps bridge theoretical deduction with real-time observation and reinforces the principle that atoms shift oxidation state when they donate or accept electrons. These ideas become even more meaningful when students see that the behavior of sulfurous acid mirrors that of sulfite salts, linking acid chemistry to ionic equilibria and showing how molecular behavior persists even after dissociation.
In educational laboratory sessions, sulfurous acid emerges not as a stock reagent stored in containers but as an active system formed in situ from sulfur dioxide, which itself may be generated during experiments involving combustion or the decomposition of sulfur-containing reducing agents. This feature reinforces an important scientific concept: not all substances encountered in chemistry are isolated in pure form, and some exist only in dynamic equilibrium with other species. For example, when sulfur dioxide is bubbled into water and the pH decreases measurably, learners experience a practical demonstration of acid formation from reactive gas absorption. The reversibility of this reaction creates an excellent opportunity to connect solution chemistry with environmental processes, particularly acid rain formation. While sulfuric acid often receives more attention in environmental discussions, sulfurous acid represents an early stage in atmospheric oxidation, demonstrating how dissolved gases in moisture initiate the acidification process before further oxidation converts sulfite species into sulfate. By integrating environmental chemistry into molecular study, sulfurous acid helps contextualize theoretical concepts within real planetary processes.
The educational importance of sulfurous acid grows even further when students recognize that the molecule bridges multiple domains of chemistry rather than fitting neatly into just one category. On a structural level, it introduces polyatomic geometry, hydrogen placement, and electron-pair effects. On an acid–base level, it provides a model for diprotic acids and reversible proton transfer. On an equilibrium level, it illustrates the dependence of product stability on external conditions such as temperature and pressure. On a redox level, it demonstrates oxidation state interconversion and electron-transfer reactions that underpin analytical and industrial processes. The value of sulfurous acid lies not in its physical storage or laboratory simplicity but in its intellectual versatility, helping students connect principles that might otherwise appear unrelated. As learners revisit the concept of sulfurous acid across different subfields, repeated exposure reinforces cognitive patterns that gradually transform scattered knowledge into a unified understanding rooted in the behavior of matter.
Ultimately, sulfurous acid offers a deeply instructive example of how molecular identity exists as a balance of structure, bonding, and environmental context rather than a static symbol on paper. Every aspect of its behavior—from its dependence on dissolved sulfur dioxide to its geometry, strength as an acid, dissociation patterns, and reactivity—stems directly from the arrangement of atoms and electrons within the molecule. Teaching sulfurous acid therefore provides an invaluable lesson in the holistic nature of chemistry: the structure determines the properties, the properties determine the reactions, and the reactions determine the role a substance plays in nature and technology. By examining sulfurous acid as both a molecular species and an educational tool, students begin to appreciate that chemistry is not simply the study of isolated compounds but rather the study of how chemical systems evolve in response to their surroundings and how every transformation reflects the fundamental patterns governing the behavior of atoms and molecules in the physical world.